Abstract
Background:
Gestational diabetes mellitus (GDM) is an increasing health issue among pregnant women worldwide. Treatment of hyperglycemia during pregnancy improves outcomes for both mothers and infants. Effectively performing and reviewing self-monitoring of blood glucose is time-consuming for patients and care providers. In the modern era, most people having access to smartphones create opportunities for use of phone-based technologies to improve patient care in chronic diseases. This review aims to investigate the awareness and use of the smartphone application (app) with respect to management of GDM among pregnant women.
Materials and Methods:
Various relevant studies (n = 522) from 3 databases named Pub Med, Cochrane Library, and Google Scholar were included. For this, the study involved designing of a 5-stage review framework, which included research question identification, identification of articles, article selection, data collection, and result reporting.
Results:
Initial search criteria used a combination of keywords, by which we found out 522 literatures from 3 databases. After screening the titles and abstracts, 249 articles were excluded due to duplicate literatures and 252 articles were excluded due to the following reasons: not relevant (n = 172), editorial (n = 43), not in English (n = 7), and abstract only (n = 30). Furthermore, 10 articles were excluded because apps such as MobiGuide, pregnant + app, and GDm health were not mentioned in these articles. A total of 11 articles were included for the final analysis.
Conclusion:
The mobile apps described in the present study (pregnant +, MobiGuide, and GDm health) provided personalized health care services, patient care improvement, and enhanced patient’s compliance toward blood glucose monitoring and treatment.
Introduction
Gestational diabetes mellitus (GDM) is an increasing health issue among pregnant women worldwide, affecting 5.8%-12.9% of pregnancies 1 and having a prevalence rate ranging from 4.6% to 14% in urban areas and from 1.7% to 13.2% in rural areas of India. 2 Insulin requirements are increased during pregnancy because of the presence of insulin antagonists such as human placental lactogen and cortisol that, in turn, promote lypolysis and decrease the utilization of glucose. In the presence of insulin resistance, uptake of blood glucose (BG) is prevented and the blood sugar level continues to remain at higher levels. Another factor for increased insulin requirements during pregnancy includes increased production of insulinase by the placenta. Genetic defects of beta cells affecting the action of insulin, diseases of the exocrine cells of pancreas, endocrinopathies, drugs, and genetic syndromes can also be the causative factors for glucose intolerance with varying capacity in different subjects. The primary reason behind 80% of GDM cases is dysfunction of beta cells causing increase in the resistance to insulin. 3
Women with GDM have more chances to experience complications such as preterm birth, future type 2 diabetes mellitus (T2DM), high blood pressure, and preeclampsia. GDM mothers’ babies also have higher risk of developing excess birth weight, respiratory distress disease, and low blood sugar level, and there are chances that shoulder dystocia, birth injuries, and hyperbilirubinemia may occur in them.3,4 Controlling the blood sugar levels is the fundamental approach for the management of GDM.5,6 Healthy eating, physical activity, and weight management help patients to have stable blood sugar levels without medical treatment. 7 The treatment prescribed for 85% of GDM-diagnosed women includes lifestyle changes, insulin therapy, or administration of metformin orally. 7 To assess the difference in complications among 958 GDM women, some researchers conducted a study in which the women were divided into 2 groups: in the first one, only standard care was provided and in the other one, standard care plus dietary intervention were administered and self-monitoring of BG (intervention group) was done. Findings from the protocols clearly revealed a significant decrease in associated risks for the intervention group, which gave the following results: gestational hypertension or preeclampsia (13.6% vs 8.6%, P = .01), cesarean delivery, shoulder dystocia (33.8% vs 26.9%), and macrosomia (14.3% vs 5.9%). 8
Mobile phones have certain advantages such as portability, constant internet connectivity, easy usage of applications (apps), etc. to provide personalized interventions for GDM patients,9-11 as pregnant women are more concerned with the possible complications affecting their fetus due to GDM.12-14 GDM apps are easily available on online stores and are user-friendly, helping the patients to avoid anxiety and stress that are otherwise the common signs of pregnant women with GDM. In a short period of time from diagnosis to delivery (12-16 weeks), they can collect disease-related information and information on lifestyle modification and learn BG monitoring and self-administration of insulin and other medications with the help of such apps. 15 Because of the listed merits, the apps can be of interest to view social media sites and the related apps. Systematic reviews are essential to summarize evidence relating to the efficacy and safety of healthcare interventions accurately and reliably, in order to serve the targeted group of patients. 16 Recently, in 2019, Wu et al conducted a systematic review and meta-analysis to find out the efficacy of mobile phone apps for lifestyle modification in patients with diabetes. Twenty-six out of 2663 articles were included for compilation of review process. Overall effect on glycated hemoglobin (HbA1c) was statistically insignificant (P = .67), which led to conclude that there was a strong evidence for the efficacy of mobile phone apps for lifestyle modification in type 2 diabetes. 17 The primary objective of the present review is to find out the most accurate apps developed for GDM patients systematically and to look for the recent evidences for confirming the efficacy of mobile phone apps as interventions to promote adherence to medication regimen and to have selective improvement in therapeutic outcome among pregnant women with GDM, while the secondary aim focuses on understanding the different outcomes of these mobile apps through various studies. It also emphasizes the acceptance of mobile apps developed for GDM patients by the physicians and assesses the usefulness of apps in improving the management of such cases.
Materials and Methods
Various relevant studies (n = 520) from 3 databases named Pub Med, Cochrane Library, and Google Scholar were included. For this, the study involved designing of a 5-stage review framework, which included research question identification, identification of articles, article selection, data collection, and result reporting.
Research Question Identification
The first and the most essential step of a review or research is identification of research question. In this review, we formulated 2 research questions as follows:
What are the main mobile apps that were accepted and deployed in hospital according to the published clinical studies for GDM?
What are the roles and outcomes of mobile phone apps in patients with GDM?
Identification of Articles
Several changes in clinical practice must be addressed during the process known as identification of recent articles. To identify the relevant studies, we selected 3 databases: Pub Med, Cochrane Library, and Google Scholar. We conducted an electronic search using 2 sets of keywords and their combination. Keywords used were: gestational diabetes mellitus and smart phone app, mobile app, m health. The combination of first 2 sets of keywords was searched in order to find out relevant studies. To explore the convenient results, titles and abstracts were also searched in Pub Med and Cochrane Library. The articles which the met inclusion criteria were finally included in the study.
Article Selection
According to the inclusion criteria, we extracted literatures for further analysis. We retrieved the articles by title, abstract, and full text. The search strategy flow diagram is depicted in Figure 1. The initial search was carried out in January 2019, which was followed up in August 2020 to identify newly published studies.
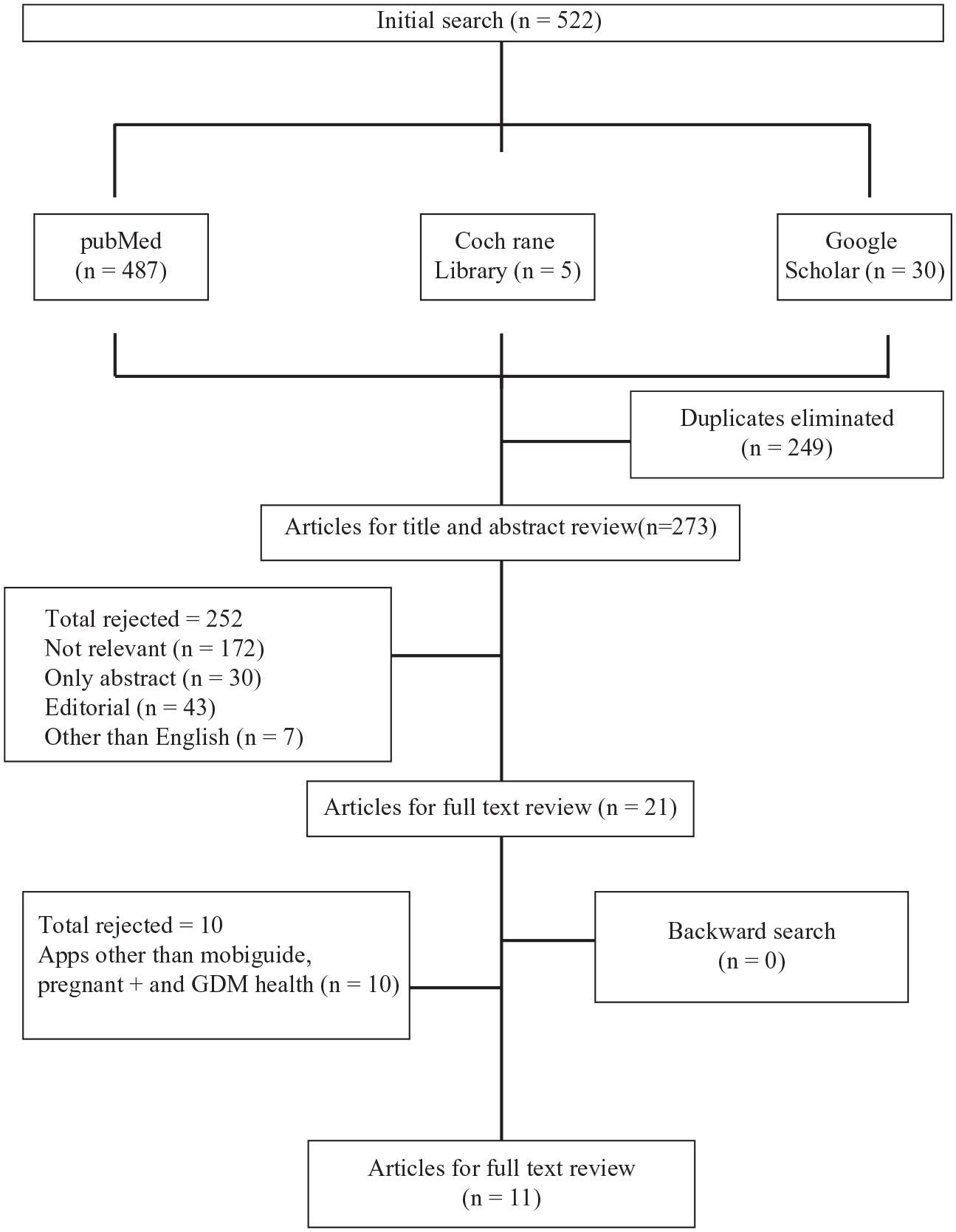
Search strategy flow diagram.
Data Collection
After full text review, data charting was carried out in Excel to answer the research questions and included the following contents: year of publication, authors’ names, study site, study design, sample size, and outcome of the study.
Result Reporting
To summarize data from all studies, information from data charting was used and results from various review results were compared.
Results and Discussion
Initial search criteria were based on a combination of keywords, by which we found out 522 literatures from 3 databases. After screening the titles and abstracts, 249 articles were excluded due to duplicate literatures and 252 articles were excluded due to following reasons: not relevant (n = 172), editorial (n = 43), not in English (n = 7), and abstract only (n = 30). Furthermore, 10 articles were excluded because apps such as MobiGuide, pregnant +, and GDm health were not mentioned in these articles. A total of 11 articles were included for the final analysis. We conducted an electronic search using 2 sets of keywords and their combination. Keywords used were the following: gestational diabetes mellitus and smart phone app, mobile app, m health. The selection process is shown in Figure 1. A summary of all these articles related to the mobile apps for GDM women is presented in Table 1.
Summary of the Mobile Applications.
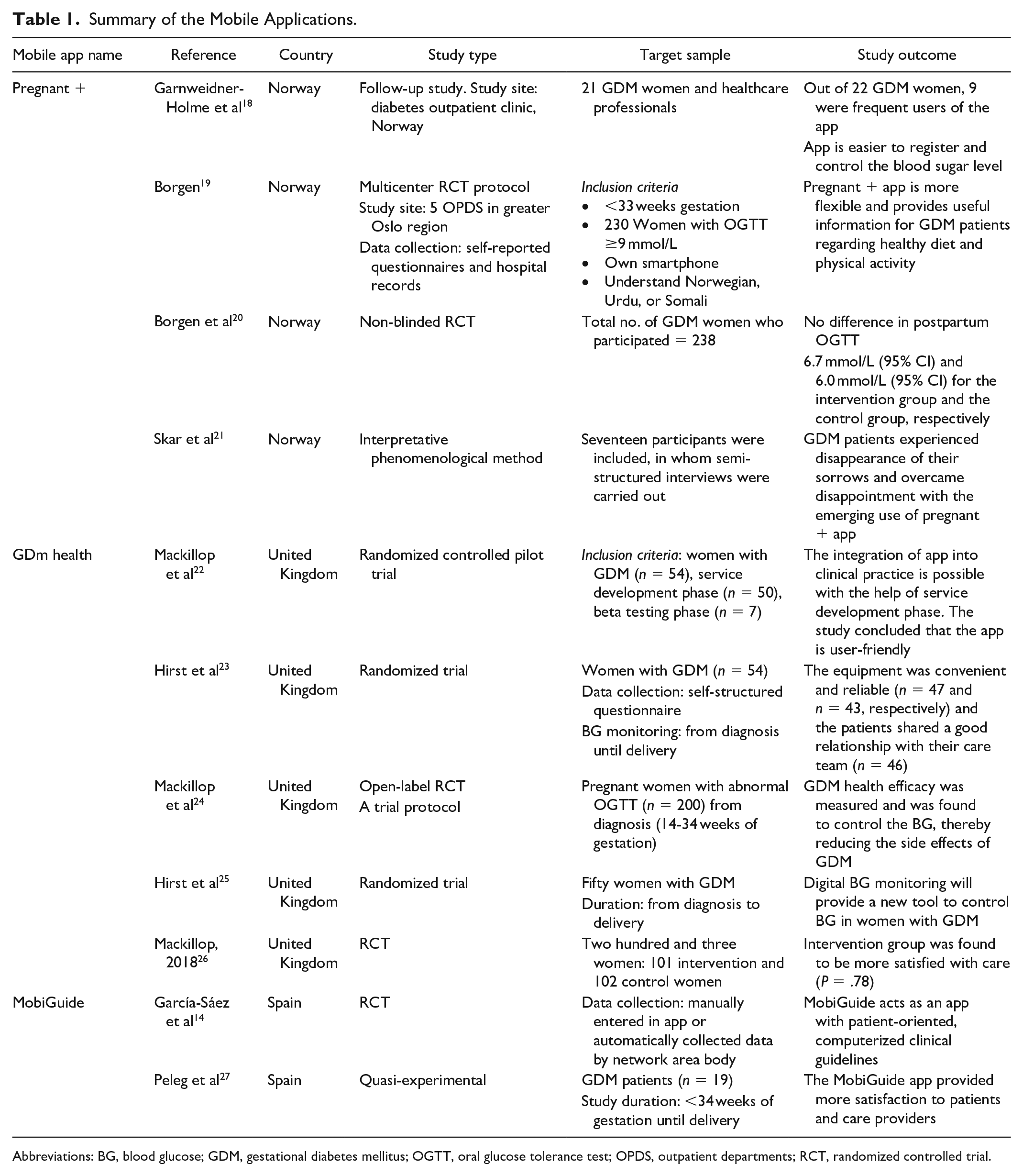
Abbreviations: BG, blood glucose; GDM, gestational diabetes mellitus; OGTT, oral glucose tolerance test; OPDS, outpatient departments; RCT, randomized controlled trial.
Mobile apps have played an important role in the field of GDM research. Various mobile apps accepted by the healthcare professionals, such as pregnant + (Norway), MobiGuide (Spain), and GDm health (United Kingdom), are used for the management of GDM. It has been shown that effective management and prevention of risk factors associated with GDM can be achieved by identifying women with GDM and strictly controlling their BG levels.
Pregnant + App
Pregnant + is a mobile app that was developed to provide an automated transfer of measured BG into the smartphone. It is an easy way to analyze the BG level for pregnant women with DM. Information included in the apps is on healthy diet, physical activity, and general information regarding GDM. BG records cannot be sent automatically to healthcare professionals due to some security reasons. BG record print out facility is available at clinics. A group discussion is conducted among researchers and healthcare professionals. Content of the mobile app should be more specific to GDM, and optimal BG levels can be highly individual. In a study conducted on 21 pregnant women with GDM, 9 were frequent users of apps. Results of the app were analyzed between June 2014 and January 2015. This study was conducted to analyze the functions of the app. Most of the patients involved in the study were able to understand clearly the general information (91%), BG level registration option (100%), information on healthy diet (91%), and physical activity advices (91%). Some other options were also available in the mobile app like how to take appointments for medical advice and where to input body weight values. Participants were satisfied with the pregnant + app as compared with the standard treatment alone.
Garnweidner et al conducted a qualitative study based on healthcare professionals’ attitude toward and experiences of using a culture-sensitive pregnant+ app for women with GDM. Most of the study participants were midwives (n = 6) and the remaining were nurses specialized in diabetes care (n = 3). From the study, they found that the mobile app (pregnant + app) as an appropriate tool for the care and follow-up of women with GDM. 18
In 2017, a study was done by Borgen et al to find out the importance of smartphone app (pregnant + app) in women with GDM. They recruited 230 study patients from 5 outpatient departments in the greater Oslo region. Patients were divided into 2 groups: one of the groups received standard care and pregnant + app and the control group received only standard care. The main outcome of the study was the glucose level measured at two hours OGTT at three months postpartum. The secondary outcome measured consisted of assessment of complications for both child and mother, the mode of delivery, how to prevent illness and regain quality health, knowledge about GDM, birth weight, and quality of life (QOL). 19
Skar et al conducted a study in 2018 wherein the analysis involved interpretative phenomenological method. Seventeen participants were included; among them, semi-structured interviews were carried out. GDM patients experienced disappearance of their sorrows and overcame their disappointment with the emerging use of pregnant + app. The study concluded that the use of pregnant + app contributed to BG management among GDM women. The study findings also indicated that the implementation of pregnant + app is only possible with closer collaboration between patients and healthcare professionals. 21
Borgen et al conducted a multicenter, non-blinded, randomized controlled trial in 2019 at 5 diabetes outpatient clinics in the Oslo region to assess the effect of pregnant + app in postpartum OGTT among women with GDM. The control group received usual care only, and for the intervention group, usual care and pregnant + app were used. The study had 238 participants. There was no difference in the postpartum level of OGTT among pregnant women (intervention group = 6.7 mmol/L, control group = 6.0 mmol/L). The study concluded that pregnant + app had no effect on two-hour glucose levels at routine postpartum OGTT. 20
MobiGuide
ModiGuide is a personalized evidence-based decision supporting system for GDM patients and their caregivers. Data collected from patients, hospital prescriptions, and mobile sensors are transferred and stored as personal health record in the mobile app. It provides personal advices and real-time GDM patient-centered recommendations through artificial intelligence.
Garcia Saez et al 2004 described the development of MobiGuide. The GDM guidelines have been used to make the mobiguide application. Healthcare professionals and patients are benefited by MobiGuide. MobiGuide app features include therapy advices, monitoring parameters, prescription by doctors, and clinical assessment for patients. The physicians can check patient compliance and BG control through mobile app; accordingly, they can make changes in diet and exercise, insulin prescription, etc. 14
Peleg et al conducted a study to understand the importance of personalized patient guidance system (MobiGuide system). The study concluded that the patients had high compliance with MobiGuide for the measurement of BG, ketonuria, and BP. 27
GDm Health
Mackillop et al developed a real-time smartphone solution for the management of women with GDM, known as GDm health. It consisted of a website and mobile app. GDm health offered two-way communication process. Physicians provided advices regarding medications and lifestyle changes by analyzing data available from the website. Patients’ BG levels were checked with the help of a glucometer. Bluetooth facility was attached with the glucometer. The readings were immediately transferred to the website. The women involved in the study used system for approximately 13.1 weeks. The results showed that the compliance of GDM app by women was excellent.
In 2016, Hirst et al conducted a study to determine women’s satisfaction with the use of GDm health system and their attitudes toward their diabetes care. In this study, 52 pregnant women were recruited; their BG levels were monitored using GDm health from diagnosis until delivery. After delivery, the women completed a structured questionnaire assessing general satisfaction, equipment issues, and relationship with the diabetes care team. Then the responses were scored on a seven-point Likert-type scale. The study findings were: 91% women agreed that their care was satisfactory, 95.9% and 87.7% women agreed that the equipment was convenient and reliable, respectively, 85.7% women were satisfied that GDm health fitted into their lifestyle, and 93.8% agreed that they had a good relationship with their healthcare team. Results of this pilot study demonstrated that GDm health is acceptable and convenient for a large proportion of women. 25
Mackillop et al made a trial protocol for GDM patients. The objective was to compare the efficacy of standard clinical care for GDM patients with smartphone-based BG management. The GDM patients were randomly assigned to 2 groups. Women with 14-34 weeks of gestation were included in the study. The primary outcome of the study was BG control. BG checking was done from recruitment until delivery for both control and intervention groups. 22
Hirst et al conducted a study in 2015 to find the importance of digital BG monitoring. They included 49 GDM women in their study, of which 92% were satisfied with the app. The results suggested that the system was reliable, convenient, and fitting well with their lifestyle. They also checked the birth outcome of GDM women participants, but the data of only 24 women was available; of these, 29% delivered and gave birth to large-for-gestational-age (LGA) babies. 23
Mackillop et al (2018) was conducted a randomized controlled trial to determine mobile phone-based BG management to control GDM. The participants (203 GDM women) of the study were divided into 2 groups (101 intervention and 102 control GDM women). GDm health app was applied for the intervention group. The intervention group reported less preterm births, few cesarean deliveries, etc. There was no significant change in BG levels between the control and the intervention groups. Both the groups showed similarity in neonatal and maternal outcome. The P value was found to be not less than .01 (P = .23) while comparing the duration from recruitment to delivery; this period was 54 and 49 days, respectively, in the intervention group and the control group. The study concluded that BG monitoring was safe and effective. It explained the high-level data pickup using GDm health. Digital BG monitoring provides a practical solution to overcome the burden of GDM. 24 A summary of all these articles related to mobile apps for GDM women is presented in Table 1.
The result from this review will be providing a new hypothesis for future research/experimentation and improving the health care of GDM women. It will be a future reference for researchers from India to develop and implement GDM mobile apps in Indian languages, as such types of apps have not been developed yet in India.
Conclusion
This is the first study which has completed a comprehensive literature review of mobile phone app in pregnant women with GDM. The study involved the use of mobile apps such as pregnant + (Norway), MobiGuide (Spain), and GDm health (United Kingdom) that are well accepted by the healthcare professionals. It has been shown that effective management and prevention of risk associated with GDM can be achieved by identifying women with GDM and strictly controlling their BG levels. These mobile apps provide personalized healthcare services, patient care improvement, and enhanced patient’s compliance toward BG monitoring and treatment. Moreover, these findings highlight the need for better reporting of health-related app interventions. The use of health intervention apps is more appropriate, and they have attractive means of communication (sound, interactivity, pictures, and text) and functionality (BG level). The review showed that effective management and prevention of risk associated with GDM can be achieved by the use of mobile apps along with standard medical care. Collaborations among researchers, healthcare professionals, app developers, and policy makers could enhance the process of delivering and testing proof-based apps to improve health effects.
Footnotes
Acknowledgements
All authors significantly contributed to design, data interpretation, and analysis of the literatures.
Abbreviations
DM, diabetes mellitus; GDM, Gestational diabetes mellitus; BG, blood glucose; QOL, quality of life; LGA, Large for gestational age.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
