Abstract

Keywords
A recent letter in Journal of Diabetes Science and Technology from Grino and colleagues 1 has commented on the different time in hypoglycemia reported for the study populations in three RCTs using traditional continuous glucose monitoring (CGM) or flash glucose monitoring (FGM).2-4 The authors repeat previous assertions that sensor glucose monitoring data in clinical trials should not be reported without accompanying blood glucose monitoring (BGM) data. 5 Grino and colleagues then report their own data comparing FGM and BGM in a population of 9 elderly patients with type 2 diabetes on multiple daily injections of insulin, 6 in which a proportion of sensor readings below 70 mg/dL were associated with BGM readings above 70 mg/dL and not in hypoglycemia. The authors conclude that FGM is unsuitable for detection of hypoglycemia in elderly patients. This is incorrect and is not part of FDA pre-market approval as implied by the authors.
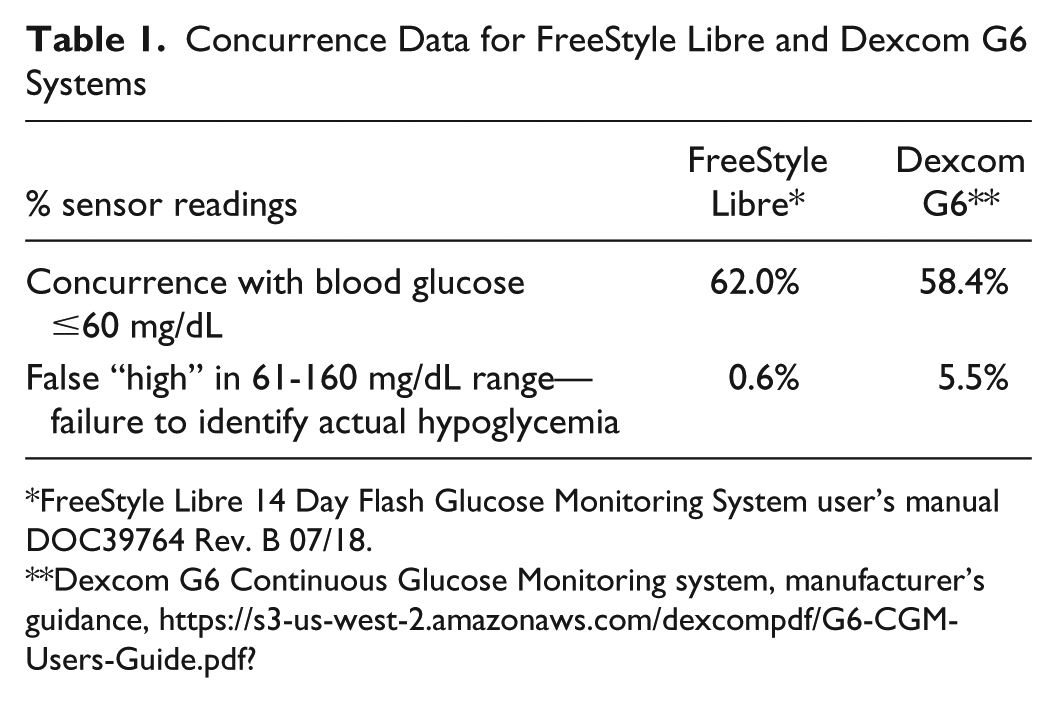
In real-world situations, it is critical that potential episodes of hypoglycemia be identified and managed before there are harmful consequences for people with diabetes using insulin. Potential harm is not associated with false low sensor readings, when actual blood glucose is euglycemic, but with false high sensor readings when blood glucose is below 70 mg/dL. From a clinical perspective, we must avoid a situation in which sensor systems report that glucose is above 70 mg/dL when capillary blood glucose is in fact below 70 mg/dL and the patient may not have time to prevent a potentially harmful hypoglycemia. On these points, the concurrence data for different devices is informative.7,8 These data indicate for each range of sensor readings whether paired laboratory reference blood glucose values are in the same glucose range or in a glucose range above or below the sensor readings. The FreeStyle Libre FGM system has 62.0% concurrence with reference blood glucose levels below 60 mg/dL, and the Dexcom G6 CGM system is 58.4% concurrent with reference blood glucose below 60 mg/dL. When FGM readings are within a range 60-160 mg/dL, reference blood glucose is lower than 60 mg/dL only for 0.6% of all system readings. The concurrence data in this range are important to note as it is when all of the “false” high sensor readings occur. For the CGM system, as many as 5.5% of sensor readings are shown in the 60-160 mg/dL range when reference blood glucose values are below 60 mg/dL. These rates of sensor readings that fail to identify actual low blood glucose are clinically significant as they expose the user to undetected hypoglycemia. It is interesting that these data indicate that the CGM system is more than 9-fold more likely to underreport low glucose compared to the FGM system. These data are summarized in Table 1, and together they confirm that the performance of the FGM system is aligned with the safe and effective reporting of low glucose events for people with diabetes on insulin therapy.
Concurrence Data for FreeStyle Libre and Dexcom G6 Systems
FreeStyle Libre 14 Day Flash Glucose Monitoring System user’s manual DOC39764 Rev. B 07/18.
Dexcom G6 Continuous Glucose Monitoring system, manufacturer’s guidance, https://s3-us-west-2.amazonaws.com/dexcompdf/G6-CGM-Users-Guide.pdf?
Footnotes
Abbreviations
BGM, blood glucose monitoring; FGM, flash glucose monitoring; RCTs, randomized controlled trials.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alexander Seibold is full time employees of Abbott Diabetes Care.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Abbott GmbH & Co KG.
