Abstract
Background:
Globally millions of people with diabetes still prick their fingers to measure blood glucose. The aim of this study was to comprehensively evaluate and to compare three lancing devices set at the minimum (“1”) and at the maximum (“5”) lancing depth with respect to blood volume (BV) and pain related to lancing.
Methods:
Lancing devices tested were A—Glucoject Dual PLUS, B—droplet (both: HTL-Strefa S.A., Poland), and C—Microlet Next (Ascensia Diabetes Care, Switzerland), all used with personal lancets of three sizes 28G, 30G, and 33G. BVs were measured with calibrated capillaries. Pain related to lancing was expressed as a derivative of pain rating with visual analog scale.
Results:
In 90 participants with diabetes, 360 lancing procedures were performed. Overall, BV and pain were higher for “maximum” compared to “minimum” lancing depth (for both P < .001). Pain differed between devices (P ≤ .001), overall was higher for device A compared to B or C; in paired comparisons differences were significant for the following settings: A > B for 28G/1 and 33G/1, B > C for 30G/1, and A > C for 28G/1, 30G/1, and 33G/1. In aggregated comparison we did not prove a significant effect of lancet size on either BV nor pain (P = .1109, P = .4966, respectively).
Conclusions:
BV depended mainly on lancing depth. Pain depended on lancing depth and to some degree on device type. The results may serve as a source of comparative data of lancing devices performance for studies in which other lancing devices and/or lancets would be tested.
The study was registered at ClinicalTrials.gov: NCT03479619
Introduction
Lancing devices are still in common use among all individuals with diabetes who perform self-monitoring of blood glucose (SMBG) and, although continuous glucose monitoring (CGM) has an important role in treatment of many patients with diabetes, lancing fingertips cannot be dismissed yet. 1 The continuing SMBG and lancing devices popularity is due to CGM systems’ limited accessibility and costs. Moreover, lancing devices and SMBG are used by many CGM systems’ users, eg, during rapid fluctuations in the blood glucose (BG) concentration, for calibration of certain real-time systems and because not all CGM systems are approved for making treatment decisions. Thus, although we are approaching “the End of Fingersticks” 2 this process is gradual with SMBG remaining, parallel to CGM, up-to-date. As majority of adults with type 1 diabetes (globally more than 40 million people) still need to frequently prick their fingers, and many individuals with type 2 diabetes also perform SMBG, the global population of lancing devices’ regular users remains significant.1,3
Obtaining a proper blood sample for SMBG may be challenging due to improper blood amount or due to pain related to lancing.4,5 To perform a reliable measurement, many contemporary BG meters require blood samples as small as 0.3-0.6 µL; however, it was postulated that samples of 1-2 µL are more practical and easier for handling. 5 Despite the fact that lancing devices remain an important element of diabetes management, there is still no clear consensus on which parameters influence blood volume (BV) and pain, eg, how lancet size and lancing depth affect blood sample volume or pain or whether there is a correlation between pain and BV obtained during lancing.6-8 Several valuable studies assessing patients’ routine lancing practices or comparing lancing devices have been published; however, these publications lack information about lancing performance in non-patient-adapted conditions.6,9-11 Most lancing device-related studies were scheduled in such a way that first the minimum lancing depth necessary to get a predetermined minimum blood sample was established, and then tested lancing devices were compared between themselves with these individually established lancing settings.6,7,9-11 Such study designs do not enable an assessment or comparisons between different types of lancing devices set at the same lancing depth, or between lancing devices of the same type used with lancets of different size (gauge) or set for different lancing depth, which are situations met by patients when they start using a new type of a lancing device.
Taking the above into consideration, the aim of this study was to evaluate in a comprehensive way and to compare three conventional lancing devices set at the minimum and at the maximum lancing depth used with lancets of three sizes, with respect to BV obtained and pain related to lancing.
Investigational Devices and Methods
In this randomized, single-blind, single-center, one-visit study, three lancing devices set at minimum and maximum lancing depth used with personal lancets of three sizes were tested with respect to BV and the intensity of pain related to puncture. Lancing devices used in the study were Glucoject Dual PLUS (HTL-Strefa S.A., Adamówek, Poland)—device A, droplet (HTL-Strefa S.A., Adamówek, Poland)—device B, and Microlet Next (Ascensia Diabetes Care Holdings AG, Basel, Switzerland)—device C (Supplemental Figure 1). All the three devices have five grades for setting the lancing depth; each device type was tested only for its minimum (“1”) and maximum (“5”) lancing depth. For each device the lancing depth setting indicator “1” represents a different absolute lancing depth in millimeters (A, 1.60 mm, B, 0.85 mm, C, 1.25 mm), and for each device the lancing depth setting indicator “5” represents a different lancing depth in mm (A, 2.40 mm, B, 2.05 mm, C, 2.75 mm) (Supplemental Table 1). Devices were used with personal lancets droplet of three sizes: 28G, 30G, and 33G (HTL-Strefa S.A., Adamówek, Poland, Supplemental Figure 2).
Inclusion criteria were diabetes diagnosed >2 years ago, age 18-50 years, and performing 3-8 BG measurements per day. Patients taking anticoagulant medication or nonsteroidal anti-inflammatory drugs and those with diagnosed bleeding disorders were excluded.
Lancing procedures were performed in all participants by the same investigator. The 18 different configurations of lancing device type (A, B, C), lancet size (28G, 30G, 33G), and puncture depth ( “1” or “5”) were randomly assigned to consecutive participants and their 4 lanced fingers (middle and ring fingers of both hands). For each finger a new device and a new lancet were used. During the lancing procedures a participant was separated from the investigator by a curtain and did not see neither the device nor her/his own hands. The investigator punctured the lateral surface of the fingertip; after the puncture, a light pressure was applied on the finger (distal, along the arterioles, as it is patients’ custom practice). After the puncture, blood was collected into calibrated glass capillaries. When no blood was visible after the first lancing, lancing device was cocked again, and lancing was repeated to the maximum of three attempts. Collected BV (in µL) was calculated with an accuracy of 0.17 µL based on the amount of blood collected in capillary expressed in millimeters. After each finger puncture the participant marked the intensity of pain related to lancing on a visual analog scale (Supplemental Figure 3). To standardize the assessment of pain perception, pain related to lancing was expressed as normalized-VAS (nVAS), where nVAS = VAS/everyday-VAS (“everyday-VAS” was the patient’s rating of pain she/he perceived during everyday finger pricking and “VAS” was the rating of pain related to lancing this patient’s particular finger during the study). Skin type of patients’ fingers was assessed visually and using palpation by the same investigator, and classified as thin, normal, thick, very thick, or callous skin.
The study protocol was approved by the Bioethics Committee of the Medical University of Lodz (decision Nr RNN/257/17/KE, update KE/140/18) and all participants signed informed consent before starting study procedures. The study was registered at ClinicalTrials.gov under identifier NCT03479619.
The Statistical Analysis
Categorical variables were presented as numbers and percentages (n, %). These parameters were compared by using Pearson’s chi-square test, Yates’ corrected chi-square test, and Fisher’s exact test depending on numbers within 2 × 2 table. Continuous variables were presented as medians with interquartile range (lower quartile—Q1, upper quartile—Q3) and minimum–maximum values, and were compared using the Mann–Whitney U-test or Kruskal–Wallis test by ranks (one-way analysis of variance on ranks). Post hoc comparisons were done using Dunn’s test. Correlations were evaluated using nonparametric Spearman’s rank correlation test. The Statistica 12.5 PL package (Statsoft, Tulsa, OK, USA) was used for the analysis. A P value of <.05 was considered as statistically significant. The studied group size was determined as N = 90 participants (determination of sample size is described in Supplemental Materials).
Results
Characteristics of the study group (N = 90) are presented in Table 1. Skin type distribution of the lanced fingers was the following: thin skin 11 fingers (3.1%), normal skin 177 fingers (49.2%), thick skin 129 fingers (35.8%), and very thick skin 43 fingers (11.9%). Results for 360 devices (120 of each type) used with 360 personal lancets (120 of each size) were included into the final analyses. Each of the 18 possible configurations of 3 lancing devices, 3 lancet sizes, and 2 depth settings were used 20 times except for 2 configurations (C/33/5 was used 21 times, C/28/5 was used 19 times). Patients, whose fingers were lanced with devices A, B, or C, did not differ significantly with respect to sex distribution, age, diabetes duration, body mass index, HbA1c, skin type of lanced fingers, or present smoking status.
Study Group Characteristics.
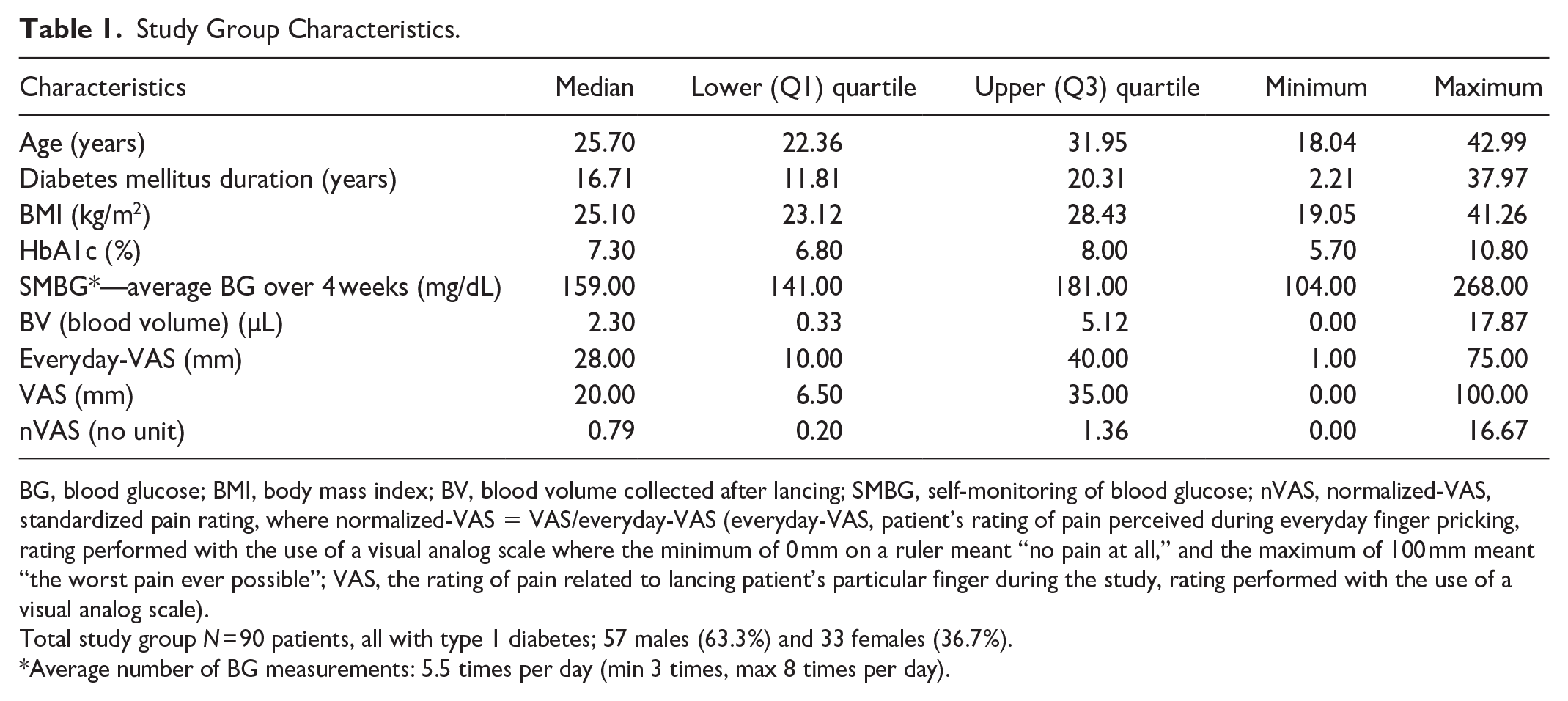
BG, blood glucose; BMI, body mass index; BV, blood volume collected after lancing; SMBG, self-monitoring of blood glucose; nVAS, normalized-VAS, standardized pain rating, where normalized-VAS = VAS/everyday-VAS (everyday-VAS, patient’s rating of pain perceived during everyday finger pricking, rating performed with the use of a visual analog scale where the minimum of 0 mm on a ruler meant “no pain at all,” and the maximum of 100 mm meant “the worst pain ever possible”; VAS, the rating of pain related to lancing patient’s particular finger during the study, rating performed with the use of a visual analog scale).
Total study group N = 90 patients, all with type 1 diabetes; 57 males (63.3%) and 33 females (36.7%).
Average number of BG measurements: 5.5 times per day (min 3 times, max 8 times per day).
Sample volumes and pain intensity (nVAS) were higher in an aggregated subgroup of patients with thin and normal skin compared to the aggregated subgroup of patients with thick or very thick skin (P = .0199 and P = .0498, respectively). Sex and right- or left-handedness were not correlated with BV or pain intensity (nVAS).
General Devices Performance
During 321 lancing procedures lancing device was cocked and released once (default situation). During 39 lancing procedures (when no blood was visible after the first lancing device release) lancing device was triggered more than once: 35 times—twice (9 devices A, 14 devices B, and 12 devices C), 4 times—3 times (1 device B and 3 devices C). During 30 lancing procedures during which the device needed to be triggered twice, a device was set at lancing depth “1” and in five cases at lancing depth “5.” All four lancing procedures when the device was triggered three times were with lancing depth “1.” A result of the last lancing attempt was included into the statistical analyses, and for the total number of 360 lancing procedures in 318 cases (88.3%) a measurable BV was obtained (ie, 0.17 µL or more).
Blood Sample Volumes
Aggregated comparison did not show significant differences between lancing devices with respect to BV (Table 2). In comparison of particular devices’ settings, only for the thickest lancet size (28G) used with devices set at “1” significant differences were found in paired comparisons: BV was higher for device A compared to B (P = .0283) and for A compared to C (P = .0026). Aggregated comparison did not show significant differences with respect to BV between lancets of different sizes (Table 2). In aggregated comparison lancing depth setting “5” yielded higher BV than “1.” In paired comparisons it was shown that this difference between lancing depth settings was significant for several configurations of devices B and C (for B/28G, P = .0072, for B/33G P = .0128, for C/28G P = .002, and for C/33G, P = .011), and for none with device A. BVs collected after lancing with particular configurations of device type/lancet size/lancing depth are presented in Figure 1.
Aggregated Characteristics of Lancing Devices, Lancet Sizes, and Lancing Depths with Comparisons of Collected BV and Intensity of Pain Related to Lancing (nVAS).
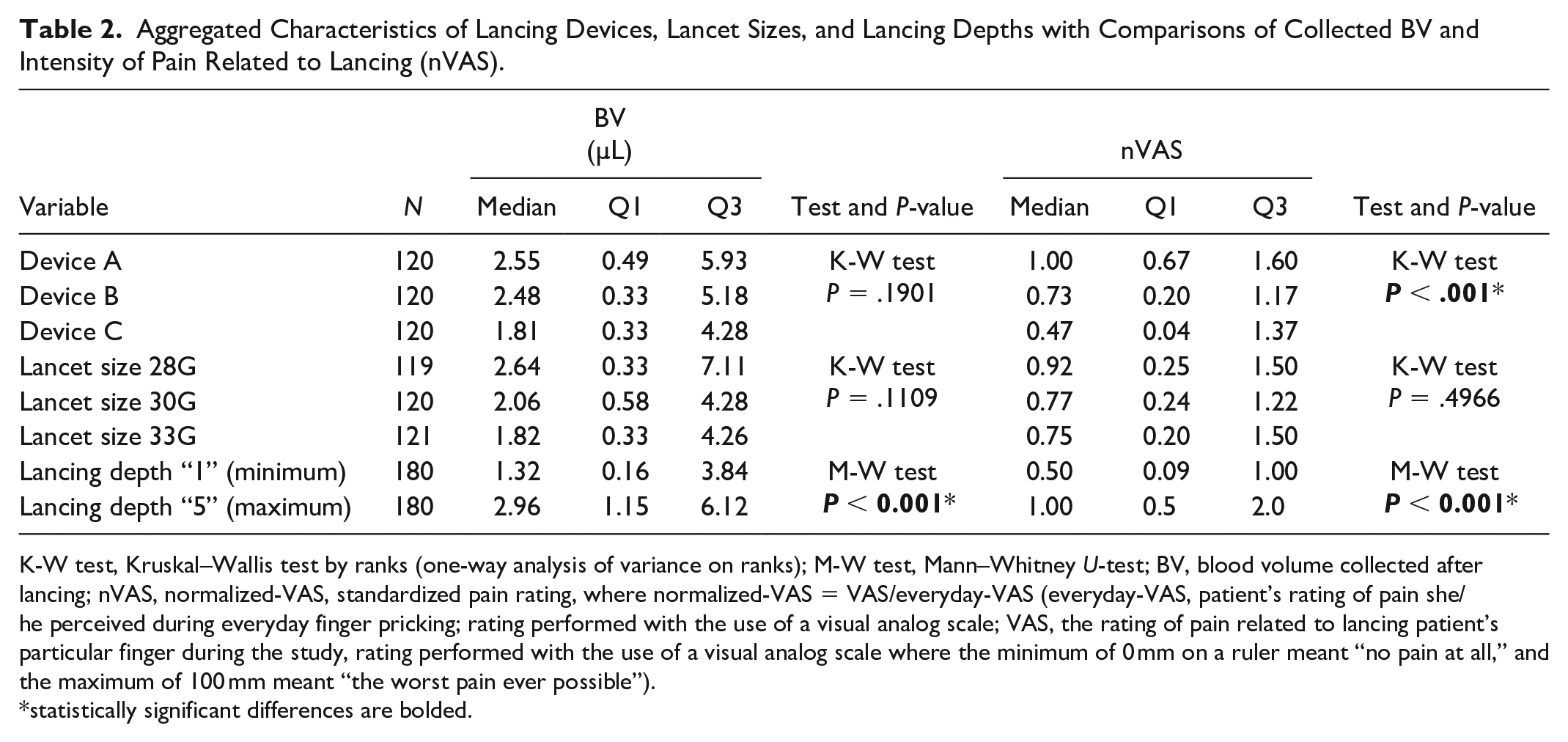
K-W test, Kruskal–Wallis test by ranks (one-way analysis of variance on ranks); M-W test, Mann–Whitney U-test; BV, blood volume collected after lancing; nVAS, normalized-VAS, standardized pain rating, where normalized-VAS = VAS/everyday-VAS (everyday-VAS, patient’s rating of pain she/he perceived during everyday finger pricking; rating performed with the use of a visual analog scale; VAS, the rating of pain related to lancing patient’s particular finger during the study, rating performed with the use of a visual analog scale where the minimum of 0 mm on a ruler meant “no pain at all,” and the maximum of 100 mm meant “the worst pain ever possible”).
statistically significant differences are bolded.
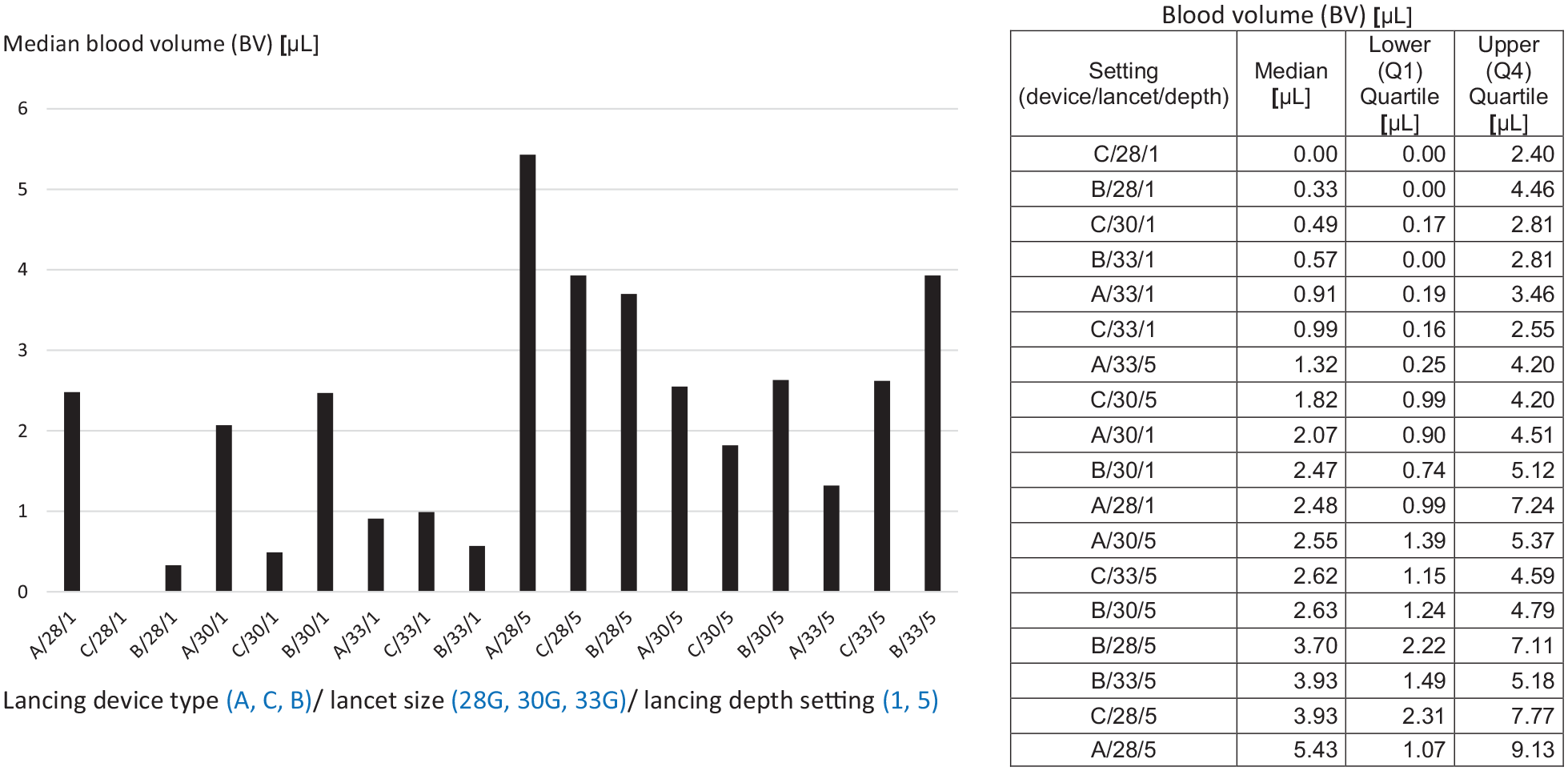
BV according to lancing device type /lancet size /lancing depth setting (eg, C/28/1 means lancing device C/lancet size 28G/lancing depth “1”). In this figure BV is presented according to (i) lancing depth setting (values for “1”—minimum are on the left, values for “5”—maximum are on the right), (ii) lancing device type (according to the average of lancing depth calculated for “1” and “5” in millimeters, ie, A [2 mm]—C [2 mm]—B [1.45 mm]), and (iii) descending lancet diameter (ie, ascending gauge No.). In the table values are presented in ascending order. There were n = 20 lancing events for each of the 18 possible configurations except for C/33/5, n = 21 and C/28/5 n = 19.
Samples of BV higher than 1 µL were yielded during 234 (65.0%) lancing procedures and 187 (51.9%) lancing procedures provided BV higher than 2 µL. No significant difference was found between percentages of samples over 1 µL for different devices (A—66.7%, B—69.2%, C—59.2%, P = .2396) or for different lancet sizes (28G—64.7%, 30G—71.7%, 33G—58.7%, P = .1067). Percentage of samples over 1 µL was higher for lancing depth “5” (76.7%) compared to lancing depth “1” (53.3%, P < .001). Percentages of samples over 2 µL were also higher for lancing depth “5” (63.3%) compared to lancing depth “1” (40.6%, P < .001).
Pain Intensity
Final results regarding pain intensity are presented as nVAS; nVAS correlated with directly assessed pain intensity (VAS) (rs = 0.801, P < .001, Supplemental Table 2). There was a positive correlation between BV and nVAS (rs = 0.3339, P = .003; Supplemental Figure 4).
An aggregated comparison between devices, lancet sizes, and lancing depth settings with respect to pain intensity are presented in Table 2. Aggregated comparison has shown that pain intensity differed between devices (Table 2) and was higher for device A compared to B (P = .0003) or C (P < .001), while the difference between B and C was not significant (P = .1848). In detailed paired comparisons, differences between devices were significant only for settings with the lancing depth “1”: A > B for 28G/1 (P = .0242) and 33G/1 (P = .0017), B > C for 30G/1 (P = .0044), and A > C for 28G/1 (P < .001), 30G/1 (P < .001), and 33G/1 (P < .001). Aggregated comparisons did not show significant differences between lancets of different sizes with respect to pain intensity (Table 2). In aggregated comparison it was shown that nVAS was higher for depth setting “5” compared to “1” (Table 2). In paired comparisons significant differences with more intense pain for “5” than for “1” were shown for several settings of devices B and C: B/28G (P = .0026), B/33G (P = .0133), C/28G (P = .0002), C/30G (P = .009), and C/33G (P < .0001). Values of pain intensity for particular configurations of device type/lancet size/lancing depth are presented in Figure 2.
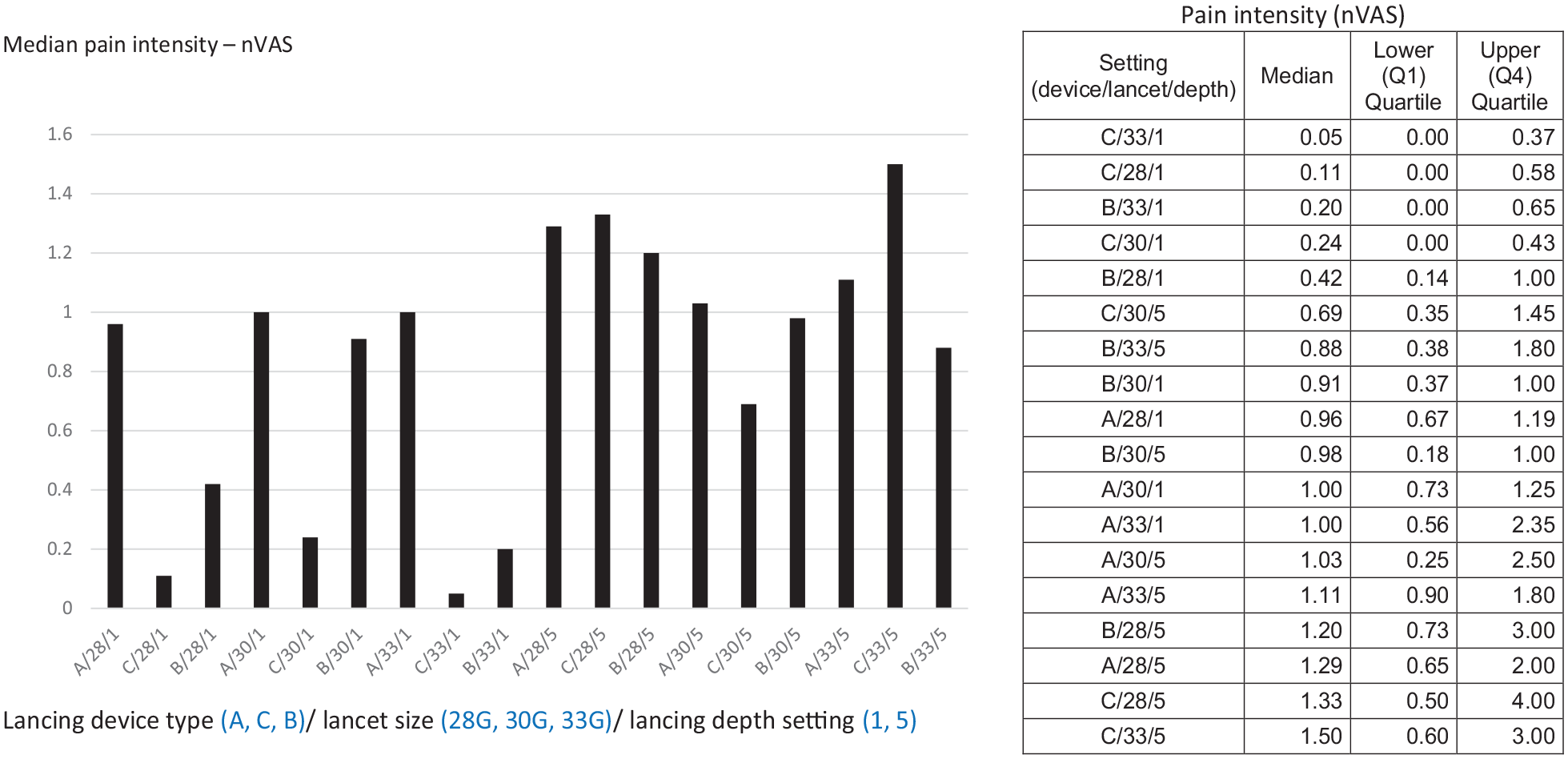
Intensity of pain related to lancing (nVAS) according to lancing device type/lancet size/lancing depth setting (eg, C/33/1 means lancing device C/lancet size 33G/lancing depth “1”). In this figure nVAS values are presented according to (i) lancing depth setting (values for “1”—minimum are on the left, values for “5”—maximum are on the right), (ii) lancing device type (according to the average of lancing depth calculated for “1” and “5” in millimeters, ie, A [2 mm]—C [2 mm]—B [1.45 mm]), and (iii) descending lancet diameter (ie, ascending gauge No). In the table values are presented in ascending order. There were n = 20 lancing events for each of the 18 possible configurations except for C/33/5, n = 21 and C/28/5 n = 19.
Discussion
In this study BV and lancing pain related to use of three lancing devices, two lancing depth settings (minimum and maximum), and personal lancets of three sizes were evaluated and compared. To our knowledge, this is the first study that enabled a comprehensive assessment of the impact of lancing device type, lancing depth, and lancet size on BV and pain related to lancing without earlier adaptation of lancing device depth setting or lancet size to a particular person.
Lancing Device Type: BV and Pain
In the total comparison, the BV obtained with the three lancing devices was not different; however, for one setting (the thickest lancet size, ie, 28G and lancing depth “1”) volumes obtained with device A were bigger than samples obtained with devices B or C. This may be attributed to the highest absolute lancing depth at lancing depth setting “1” for lancing device A (1.60 mm) compared to device B (0.85 mm) and C (1.25 mm).
In the total comparison the intensity of pain related to punctures (nVAS) was the highest for device A, higher than for B or C. In detailed paired comparisons significant differences were confirmed for certain configurations with the minimum (“1”) lancing depth setting but not for the maximum (“5”) lancing depth setting. It is possible that just when the lancet tip penetrates deeper (under the upper dermal blood vessels plexus, which can be typically reached with lancing depths up to 2 mm5) and reaches the nerve layers, pain is not only more intense but also “more stable” and less distinguishable between different lancing depths (in millimeters), while during pricking the more superficial layers of the skin, the pain sensations may be more subtle and easier for staging. Due to this, differences in pain intensity between devices might be observed only for the minimum (and not for maximum) lancing depth. A probable explanation why device A was the most painful is the highest absolute (in millimeters) lancing depth for the setting “1” on device A compared to B or C (like for the BV). However, contrary to such interpretation, for one configuration (30G/1) significantly higher pain intensity was also observed for device B compared to C, even though nominal lancing depth “1” in millimeters is lower for device B compared to C. Several studies have shown that pain related to lancing may be attributed to the lancing device type or its version.6,9-11 This relation is not surprising as devices may differ with respect to their construction, which can determine jittery movement of the lancet or its hard stop at the deepest point of penetration, both contributing to generation of pain related to lancing. Devices used in the present study are constructed with a pair of springs and use spring-powered (ie, mechanical), linear motion actuation technology. They differ subtly in construction details and materials from which its parts were fabricated. We did not find significant differences between the devices with respect to BV; however, pain related to lancing was in the general assessment the most intense for the device A. This was shown even though if we “artificially” calculate the average for the used lancing depths (ie, “1” and “5”) for each device, we find that such values are the same for devices A and C (ie, 2 mm) and lower for B (1.45 mm). This, together with the observation that for certain, above- mentioned configuration pain was significantly more intense for device B than C, suggests that not only lancing depth in millimeters but also other lancing devices’ characteristics might have affected lancing pain and caused differences in pain intensity between devices.
Lancing Depth: BV and Pain
In the total comparison BV was bigger for the maximum lancing depth setting compared to the minimum. In a more detailed analysis it was found that this difference was significant only for devices B and C (and not with device A), which can be related to the higher absolute (in millimeters) difference between minimum and maximum lancing depth setting for these two devices (B: Δ = 1.20 mm, C: Δ = 1.50 mm) compared to device A (Δ = 0.80 mm). Also, the significant difference in pain intensity between lancing at minimum and maximum depth setting shown for the total group was, in further analyses, attributed mainly to devices B and C (not A). Our results with respect to pain assessment have shown that lancing depth setting influenced intensity of pain related to lancing. Higher BV obtained with higher lancing depth and more intense pain related to higher lancing depth can be attributed to the skin anatomy. A lancet has to penetrate the skin deeply enough to reach the upper dermal blood vessels plexus and to cut certain number of capillaries to provide a sufficient blood sample; however, deeper lancet penetration also increases the chance that it triggers the nerve layer located beneath and increases pain sensation. Data from some studies have also shown these correlations. 7 A relatively low but significant positive correlation between the BV and intensity of pain related to lancing that we observed may be viewed as a consequence of the above observations. The fact that this latter relation was not shown in the study authored by Grady et al may be due to a different study design. In that prior study the lancing depth was preadapted to ascertain a predefined minimum BV, which was mimicking patient’s practices with devices they already “know” and which they are familiar with. 6 Our protocol was mimicking a situation of using a new device that a patient is not familiar with and the observed BV–pain correlation may be attributed to the fact that lancing depth was not individually preadapted but randomized.
Lancet Size: BV and Pain
Within a whole group no significant differences in BV or pain could be attributed to lancet size. This observation is interesting, as manufacturers diversify lancet sizes, often between 33G (approximately 0.2 mm), 30G (approximately 0.3 mm), and 28G (0.35 mm). In one study a trend for smaller BV with smaller lancet diameters was found; however, it was observed, when differences in lancets’ diameters were significant, eg, between diameters 0.8 and 0.3 mm or 0.8 and 0.4 mm. 7 Although we did not show that commonly used lancet sizes 33G, 30G, and 28G differ significantly with respect to BV or pain, a weak trend toward bigger BV with increasing lancet diameter could be noted (Table 2), so this observation deserves further studies.
Lancing Performance
A measurable BV (ie, 0.17 µL or more) was obtained after the first lancing of 307 fingers (85.3%) and during 88.3% of the total number of lancing procedures (including these when lancing was repeated). The success rate defined as “the percentage of lancing trials that yield a sufficient amount of blood with the first lancing attempt” cited in the literature is 90%-95%. 5 However, source data for these numbers for conventional lancing devices are not commonly available in medical literature, while success rate for one of the experimental devices (using an automatic puncturing and squeezing system) was 75%. 12 In our study the overall success rate expressed as BV of more than 1 µL was 65% and was significantly higher for the maximum (76.7%) compared to the minimum lacing depth (53.3%). However, this relatively small percentage of lancing procedures that yielded blood sample bigger than 1 µL should be interpreted with caution. Our study was not designed to mimic everyday lancing practices of patients and no individual “titration” of lancing depth or adaptation of lancing device to patients’ skin type was performed. It was designed to assess lancing devices’ performance at the start of their use by a patient, and depth setting necessary to get a predefined BV was not established. Moreover, participants of this study pricked their fingers frequently in their daily life (average 5.5 times per day) and 47.7% of them had thick or very thick finger skin. We have shown that BV was significantly lower for fingers with these types of skin. Such results suggest that in daily lancing practice lancing depth should be adapted accordingly not only personally, but also for a subject’s particular finger. Diabetes educators should provide patients with detailed knowledge aiming at both, obtaining a proper, not too small but also not too big blood sample, and at preventing unnecessary pain related to lancing.
An advantage of this study is that it was a single-blinded study and that in all study participants lancing procedures were performed by the same investigator, in similar conditions, with a new device and lancet, which together with lancing depth setting were randomly assigned to a particular finger. Per protocol limitation was that the tested devices’ nominal lancing depths were different, which did not allow a comparison of BV and pain between the specific settings of the puncture depth expressed in millimeters and only “lancing depth setting” as a categorical and not quantitative variable was compared. Moreover, the use of intermediate settings would enable to assess more precisely the influence of grading of lancing depth on BV and pain intensity. Another limitation was that repeated lancing (up to three times) was allowed, as this could affect the pain sensation. However, the majority of cases when lancing was repeated were for lancing depth “1,” so it probably did not affect the final results as for this setting the pain was overall lower than for the depth “5.” The study group characteristics limit the possibility to freely generalize our results. Only persons with relatively long diabetes history, who performed frequent SMBG, were included, and high percentage of them had thick or very thick skin. Other groups should be studied to assess the performance and pain intensity related to the study devices in other populations of subjects who measure BG less frequently and whose skin characteristics may be different, eg, elderly patients with type 2 diabetes. Further studies should also focus on standardized, broader comparisons between other lancing devices available on markets or being prepared for launch to provide final users with information that is of practical importance for them. Results of our study might serve as a source of values for such comparative analyses.
Conclusion
This study demonstrated that blood sample volume depended on lancing depth. Pain depended on lancing depth and to some degree on device type. The latter may be partly, but not only, attributed to different lancing depths in millimeters for the same depth setting indicator on different devices. Data obtained in our study may serve for comparative purposes, as a source of values of lancing devices’ performance parameters (success rate, BV, and pain intensity), for future studies in which other lancing devices and/or lancets would be tested.
Supplemental Material
MIANOWSKA_Suppl.TablesandFiguresandText_Revision1 – Supplemental material for Evaluation of Three Lancing Devices: What Do Blood Volume and Lancing Pain Depend On?
Supplemental material, MIANOWSKA_Suppl.TablesandFiguresandText_Revision1 for Evaluation of Three Lancing Devices: What Do Blood Volume and Lancing Pain Depend On? by Beata Mianowska, Wojciech Mlynarski, Irena Szadkowska and Agnieszka Szadkowska in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We thank all the patients who participated in the study for their cooperation. We thank the team of Centre for Innovation and Technology Transfer, Medical University of Lodz, Poland, namely Ireneusz Staroń, MSc and Marzena Bielińska, MSc for their invaluable help in administering the study and Elżbieta Czerniawska, MD, PhD for the help in recruiting participants.
Authors’ Note
The study was conducted according to the Medical University of Lodz Study Protocol No 010617 version 03, dated 2018-03-09 approved by the Bioethics Committee of the Medical University of Lodz (decision Nr RNN/257/17/KE, update KE/140/18).
Preliminary results of this study were presented as a poster during the Diabetes Poland (PTD, Polskie Towarzystwo Diabetologiczne) Annual Meeting, May 16-18, 2019, Lublin, Poland.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BM, WM, IS, and AS received remuneration from the Medical University of Lodz for preparing the protocol and carrying out this study. None of the authors declared another conflict of interest related to this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by HTL–STREFA S.A., Adamówek 7 Str., 95-035 Ozorków, Poland (![]() ). The funder was not involved in the collection of study data, in the data analysis, or publication decisions.
). The funder was not involved in the collection of study data, in the data analysis, or publication decisions.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
