Abstract
Background:
Patients worldwide use lancing devices to perform self-monitoring of blood glucose (SMBG). Continued innovation remains important to minimize pain associated with finger sticking to facilitate patient adherence to SMBG regimens. The original One Touch Delica lancer (OTD) reduced the pain of testing. This study investigated a completely re-engineered lancing system, called One Touch Delica Plus (OTDP), intended to further minimize pain while improving the patient experience.
Methods:
One hundred and three subjects (type 1 or 2 diabetes) experienced finger sticks with both lancing systems. Device depth and blood volume (BV) generation were determined during visit 1. These parameters were used during pain assessments in visit 2. Patient feedback was also recorded using surveys.
Results:
Finger-stick testing was less painful using OTDP compared to original OTD irrespective of whether 30 (−48.2 mm, −30.4 mm, P < 0.0001) or 33 gauge (−42.6 mm, 30.5 mm, P = 0.0004) lancets were used. All 103 subjects generated the required BV (≥0.4 µL) per protocol. At equivalent depth penetration, OTDP generated higher average BVs than OTD using 30 (1.58 µL, 1.10 µL) or 33 gauge lancets (1.39 µL, 0.98 µL). Subjects responded that testing with OTDP was virtually pain free (78%) and that testing with OTDP was less painful than using their current lancing systems (79%) or original OTD (67%).
Conclusion:
OTDP demonstrated lower pain than the original OTD lancing system and participants agreed that testing with OTDP was virtually pain free and less painful than using their current lancing systems.
Introduction
Routine self-monitoring of blood glucose (SMBG) using a blood glucose meter and a home lancing device to produce a capillary blood sample remains standard of care for most patients for guiding diabetes management decisions and maintaining good glycemic control.1,2 However, poor adherence to SMBG is a significant issue with 60% of patients with type 1 diabetes (T1D) and 67% of those with type 2 diabetes (T2D) practicing SMBG less frequently than recommended by guidelines. 3 In Sweden, where both meters and strips are provided free, <50% of T1D patients adhere to guidance recommending SMBG ≥4 times per day and, notably, pain or discomfort from finger-stick lancing accounted for 14% of patient responses explaining suboptimal SMBG. 4 A study comparing pain and discomfort performing insulin injections and SMBG in 109 pediatric patients observed that 70% of kids agreed that it was important SMBG did not hurt. 5 One response from manufacturers has been to offer so-called “alternate site testing” using the palm or forearm to provide a blood sample but with mixed acceptance. A study using OneTouch UltraSoft devices concluded that patients found it more difficult to lance the palm, draw a sample, and apply enough blood to test strips compared to using fingers. 6 Therefore, continuing lancing innovation is critical to support patients to perform optimal SMBG. Heinemann and Boecker provided a detailed review of lancing technologies concluding that providing sophisticated lancing devices that allow the highest level of comfort is key for improving user acceptance. 7 We have previously shown that patients routinely express sufficient blood to fill their chosen test strip when using current lancing systems (including the OneTouch Delica, OTD) and that generating a large enough blood drop to confidently fill a test strip is important. 8 Original OTD was specifically designed to minimize vibration, lateral motion, and repetitive penetration and was shown to be less painful than most lancing systems with 80% of subjects in the study describing it as virtually pain free. 9 This current study sought to evaluate a completely new and improved version of OTD, called OneTouch Delica Plus (OTDP) (Figure 1). OTDP is a completely re-engineered replacement for the original OTD system and this study was intended to compare the performance of OTDP to OTD in terms of pain and blood sampling and also to ascertain patient feedback on OTDP compared to their current lancing system.
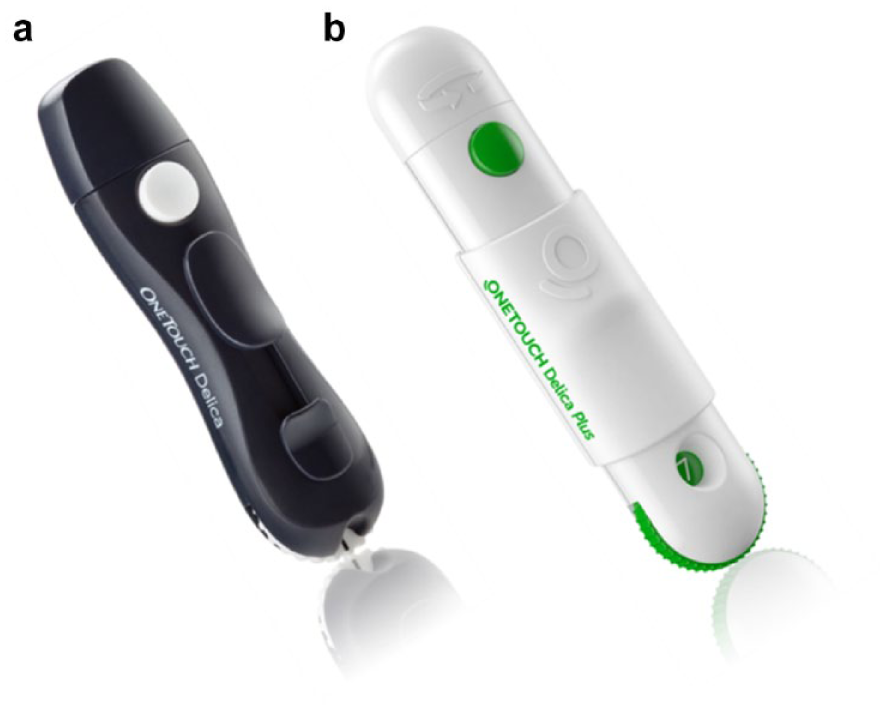
(a) One Touch Delica and (b) One Touch Delica Plus.
Methods
This single-center, open, multivisit, randomized clinical study was intended to compare the performance of original OTD to OTDP with respect to pain perception and blood volume (BV) generation. The study was approved by the relevant investigational review board and all subjects provided written informed consent. Subjects were over 18 years old, had T1D or T2D for at least 6 months, and were currently performing SMBG at least once per day for at least 6 months. The study was conducted over two visits at one center by Radiant Research Inc (FL, USA). During the first visit, 104 subjects were consented, evaluated against inclusion/exclusion criteria, and demographic information was collected. Subjects were randomized to two groups: subjects who used OTD and OTDP with standard 30 gauge lancets and subjects using OTD and OTDP with thinner 33 gauge lancets. For both groups using either lancet gauge, visit 1 consisted of a maximum of 8 lancing events to determine the depth setting necessary to achieve ≥0.4 µL of blood on the skin. A randomization schedule dictating the order of testing (device type, right or left hand, which finger and which side of the finger) was followed to minimize bias. BV expressed on the skin in microliters was determined using precision-bore glass capillary tubes (Drummond 1-000-0020 microcaps). Subjects themselves performed the lancing event after the lancer was prepared by a facilitator. Facilitators performed a standardized gentle milking of the finger to express blood on the skin to ensure consistency across all subjects. Success equated to achieving ≥0.4 µL of blood on two of three lancing events. If after two or three attempts, two successful samples were not obtained, depth was increased by 1 depth setting (OTD has 7 depth settings and OTDP has 13 depth settings). For both groups, visit 2 required each subject to perform four finger sticks with OTD and four with OTDP at the depth determined during visit 1. A randomization schedule dictating the order of testing (device, hand, finger, and side of finger) was followed to minimize bias. Following each finger stick, each subject scored perceived pain using a 150 mm visual analogue scale (VAS, Figure 2) with usual pain (pain experienced at home with their current lancer) as the center point. Subjects then completed a survey on their experiences testing using OTD and OTDP and how each system compared to their current lancing system they were using at home.
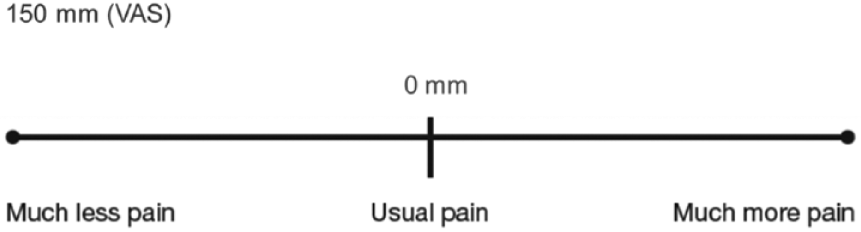
Pain assessments (VAS).
Statistical Analyses
Statistics are mainly descriptive for BV means, SDs, and medians. BV averages were calculated for each subject and the average of averages reported. BV proportions were also examined using a criterion of ≥0.4 µL average BV across subjects. For depth determination, BV, and pain, Wilcoxon’s signed-rank test was used to compare devices (and each device using either 30 or 33 gauge lancets). All statistical tests of hypothesis employed a level of significance of P < 0.05 and no adjustments were made for the number of tests performed. Survey responses were presented as favorable responses with response rates deemed statistically significant, P < 0.05.
Results
Subjects
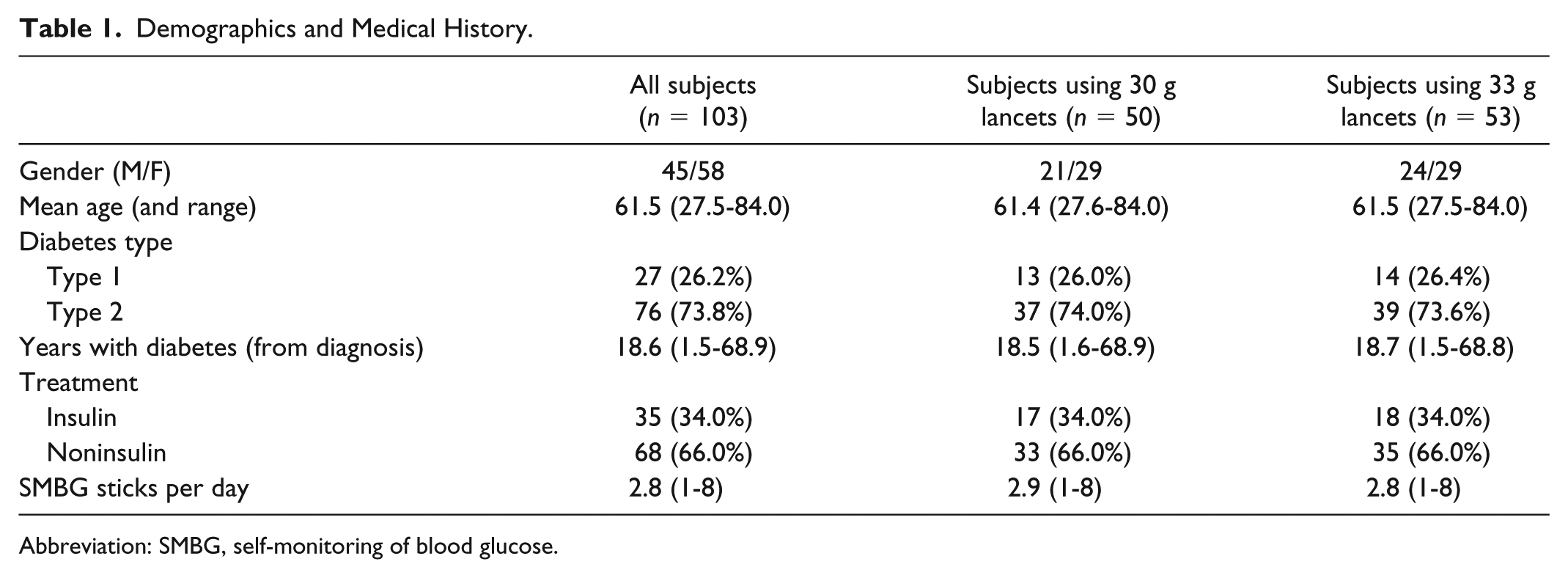
One hundred and four subjects were enrolled with 103 completing the study. One subject was lost to follow-up. Baseline characteristics are shown in Table 1. While 26.2% of the subjects had T1D, 73.8% had T2D, and among them, 34.0% of subjects were on insulin and 66.0% were noninsulin users. Mean SMBG test frequency was 2.8 tests per day across all subjects with 4.4 tests in T1D and 2.3 in T2D subjects.
Demographics and Medical History.
Abbreviation: SMBG, self-monitoring of blood glucose.
Characterization of Pain Perception With OTD and OTDP
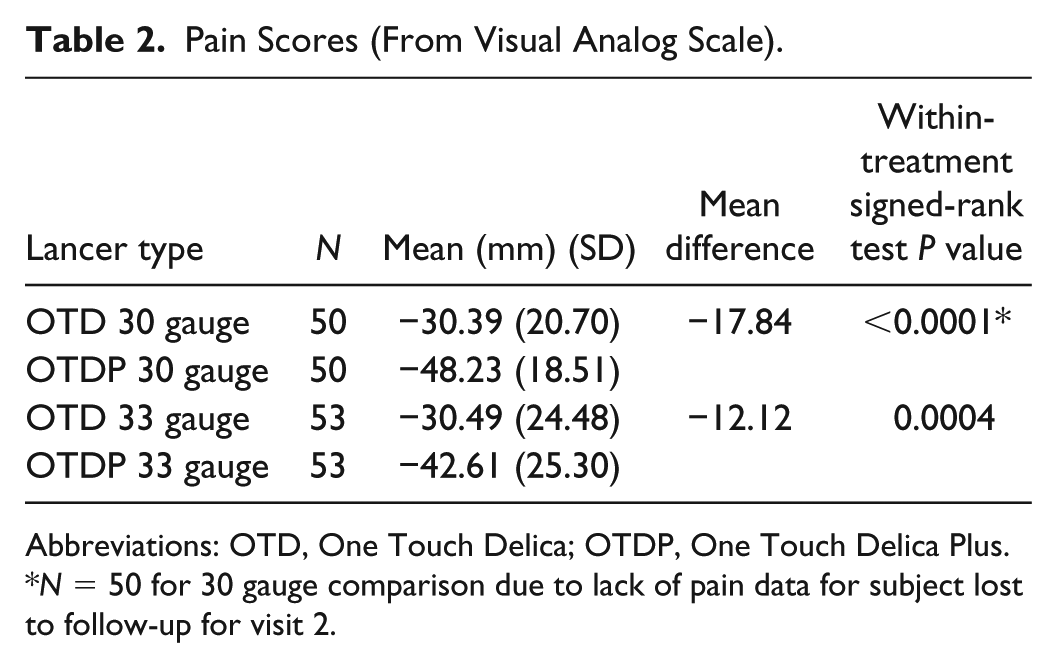
Pain assessments were completed during visit 2 with each subject experienced a total of 8 finger sticks at the respective depth for each specific lancer (capable of achieving ≥0.4 µL of blood) that had been predetermined during site visit 1. During visit 2, 4 finger sticks were performed by the subjects themselves with OTD and OTDP to provide 4 pain scores per lancer. Mean pain scores for OTDP subjects using 30 gauge lancets were significantly lower (−48.2 mm, P < 0.0001) than mean pain scores for OTD subjects in using 30 gauge lancets (−30.4 mm) with respect to the VAS (where 0 mm on the VAS was equivalent to the usual pain experienced by the subject when using their current lancing system at home). Mean pain scores for OTDP subjects using 33 gauge lancets were also significantly lower (−42.6 mm, P = 0.0004) than the pain scores in OTD subjects using 33 gauge lancets (−30.5 mm) (Table 2, Figure 3).
Pain Scores (From Visual Analog Scale).
Abbreviations: OTD, One Touch Delica; OTDP, One Touch Delica Plus.
N = 50 for 30 gauge comparison due to lack of pain data for subject lost to follow-up for visit 2.

Pain scores for One Touch Delica and One Touch Delica Plus.
Characterization of BV and Depth With OTD and OTDP
Descriptive statistics of subject BV are shown in Table 3. Mean BV in OTD subjects was 1.44 µL using 30 gauge lancets (n = 50) and 1.36 µL using thinner 33 gauge lancets (n = 53). Mean BV using OTDP was 1.22 µL (n = 51) using 30 gauge and 1.15 µL using 33 gauge lancets. OTD has 7 depth settings and OTDP has 13 depth settings; therefore, each lancer penetrates the skin at a different depth for a given setting. The average depth setting (sufficient to generate at least 0.4 µL of blood) for OTD was 1.22 (±0.54) in subjects using 30 gauge lancets and 1.49 (±0.85) in subjects using 33 gauge lancets. In contrast, the average depth setting used with OTDP was 2.40 (±0.77) in subjects using 30 gauge lancets and 2.55 (±0.82) in subjects using 33 gauge lancets.
BV and Depth Penetration.
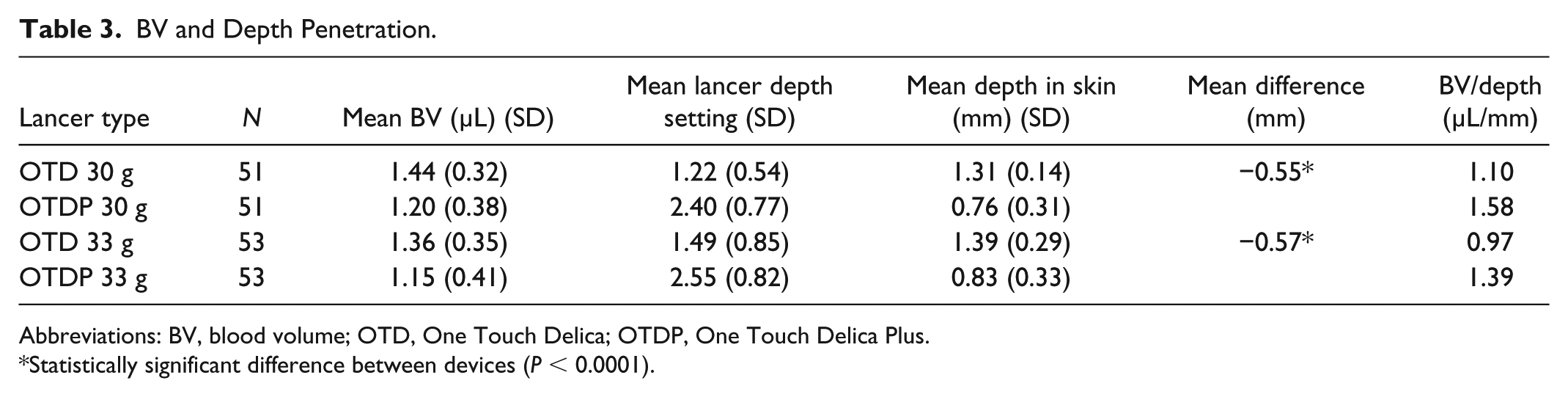
Abbreviations: BV, blood volume; OTD, One Touch Delica; OTDP, One Touch Delica Plus.
Statistically significant difference between devices (P < 0.0001).
To account for the differing number of depth settings, we, therefore, reanalyzed the BV data for each lancer based on the actual skin puncture depth (in millimeters) for each respective setting, to allow a direct comparison of BV generation between OTD and OTDP. After adjusting for actual depth penetration, mean BV in OTD subjects was 1.10 (µL/mm) using 30 gauge lancets and 0.98 (µL/mm) using 33 gauge lancets. Mean BV using OTDP was 1.58 (µL/mm) using 30 gauge and 1.39 (µL/mm) using 33 gauge lancets (Table 3). Equating for actual depth penetration the BV generated by OTDP is higher than OTD.
Subject Feedback on Testing With OTD and OTDP
Table 4 shows that 81% of subjects agreed OTDP was gentle to use, and 78% agreed testing with OTDP was virtually pain free. Subjects responded that testing with OTDP was more comfortable (75%), less painful (67%), and more gentle (64%) than testing with OTD. Table 5 shows that 79% of subjects agreed that testing with OTDP was less painful compared to the other lancers they had used. Furthermore, subjects agreed that OTDP was more gentle (79%) and more comfortable (71%) to use than lancers they had tested with in the past.
Subject Feedback on OTDP.

Abbreviations: OTD, One Touch Delica; OTDP, One Touch Delica Plus.
One hundred and three subjects responded to a 5-point scale for each survey statement: strongly agree, agree, neither agree nor disagree, disagree, or strongly disagree.
n = 80, after excluding neutral responses.
Subject Feedback on OTDP Compared to Current Lancing Systems.

Abbreviations: OTD, One Touch Delica; OTDP, One Touch Delica Plus.
One hundred and three subjects responded to a 5-point scale for each survey statement: Strongly agree, agree, neither agree nor disagree, disagree, or strongly disagree.
Safety
No unanticipated device-related adverse effects were reported.
Discussion
The study demonstrated that people with T1D and T2D experienced less pain performing finger sticks with OTDP compared to original OTD and that OTDP provided sufficient blood for blood glucose monitoring. In clinical practice, patients can select thinner (33 gauge) or thicker (30 gauge) lancets with most lancing systems and that choice may depend on their individual sampling needs, the condition of their skin, or their individual tolerance or perception of pain. Our study also demonstrated that people with diabetes experienced less pain performing finger sticks with OTDP using either 33 or 30 gauge lancets.
It is well accepted that minimizing barriers to performing SMBG could theoretically lead to increased patient compliance to SMBG regimens. However, there remains some debate with respect to the impact of pain per se on lack of adherence to SMBG. Some authors have described pain as a primary reason for patients not performing SMBG, 10 while others have shown from large surveys that around 14% of patients stated that pain was the main reason for not performing frequent SMBG. 4 Clearly there is insufficient literature on this topic but there is enough evidence to acknowledge that developing new lower pain lancing devices are necessary and will be welcomed by many patients, particularly those testing more frequently.
An in-depth review article on lancing described various lancing device features (technical improvements) and human factors that contribute to patients’ perception of pain. 7 The effectiveness of the new OTDP system in lowering pain perception may result from a number of these factors. First, controlling the needle trajectory (straightness and vibration) as the lancet fires into the skin to the desired depth could minimize soft tissue damage (and associated pain perception) within the upper layers of the skin. Some evidence for improved vibration control from supplementary bench testing was presented for OTDP given that providing in vivo data visualizing vibration or reduced tissue disruption is not feasible (Figure 4). Other manufacturers have also recognized straightness and vibration as contributing factors to a better user experience (and potentially reduced pain) by providing rail-guided or cam-driven systems, enabling smoother lancet movement to dampen vibration.7,11 Notably, our study population contained 103 subjects using the latest lancing devices, including 26 (25.2%) using LifeScan systems, 17 (16.5%) using Roche systems, 16 (15.5%) using Nipro systems, 12 (11.7%) using Bayer, and 6 (5.8%) using Freestyle systems with the remaining lancing systems from a variety of smaller manufacturers.
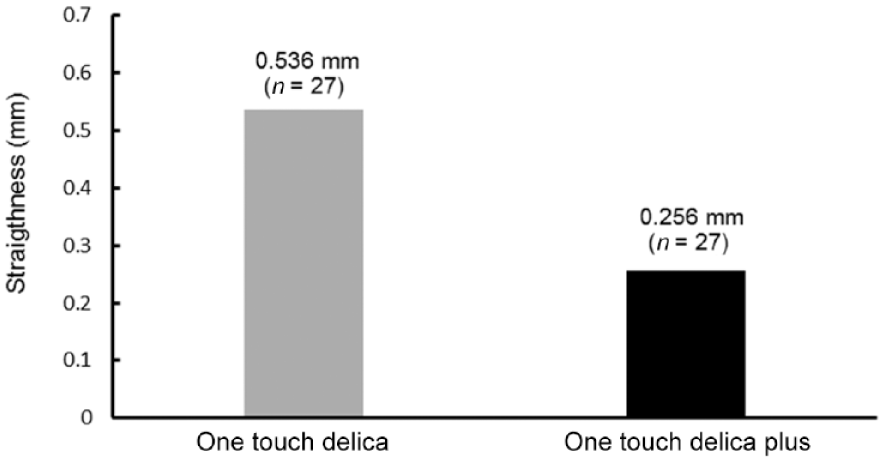
Vibration control (straightness of needle trajectory)—OTD compared to OTDP.
A second factor affecting pain perception is the noise emitted by different lancers, which accompanies firing of the device, which may unsettle people with diabetes. To mitigate this, OTDP has been engineered to emit the lowest possible noise to limit the negative connotations and any distress that the firing mechanism might elicit from patients. Supplementary evidence showing reduced noise for OTDP was gathered during bench testing showing that it was quieter than original OTD and quieter than two commonly used Roche Accucheck lancing systems (SoftClix Plus and FastClix) (Figure 5).
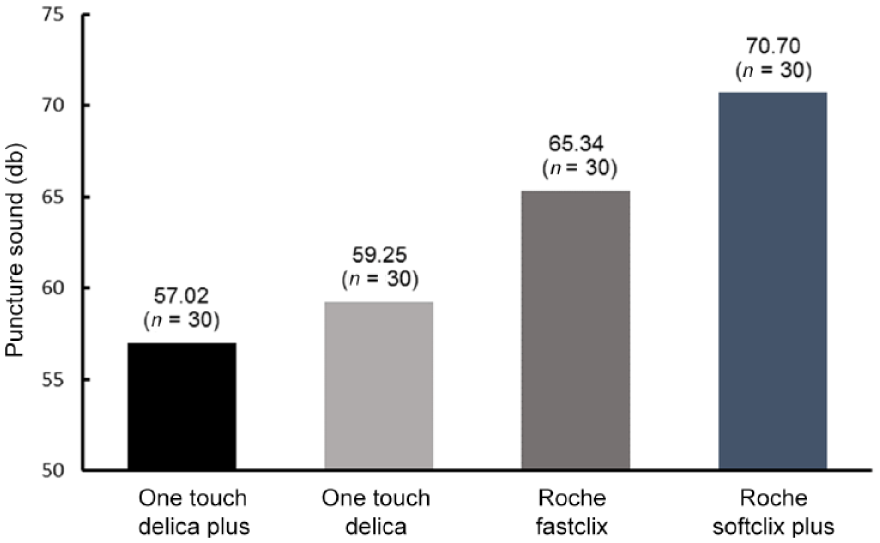
Puncture sound of selected lancing systems.
A third factor, the speed of the lancing device as it fires into the skin may also have some bearing upon the perception of pain by the patient during finger sticking. Intuitively, since there are no existing publications correlating lancing speed to pain sensation, it would seem plausible that lancing quickly is desirable to minimize the time the needle spends in contact with the skin. We included supplementary bench data on lancing speeds and found that OTDP, OTD, Roche SoftClix, and FastClix had similar lancing speeds of between 1.5 and 2 m/s (Figure 6). It is encouraging that these modern systems have broadly equivalent speeds and this factor may well contribute to reducing the pain experienced when lancing is performed.
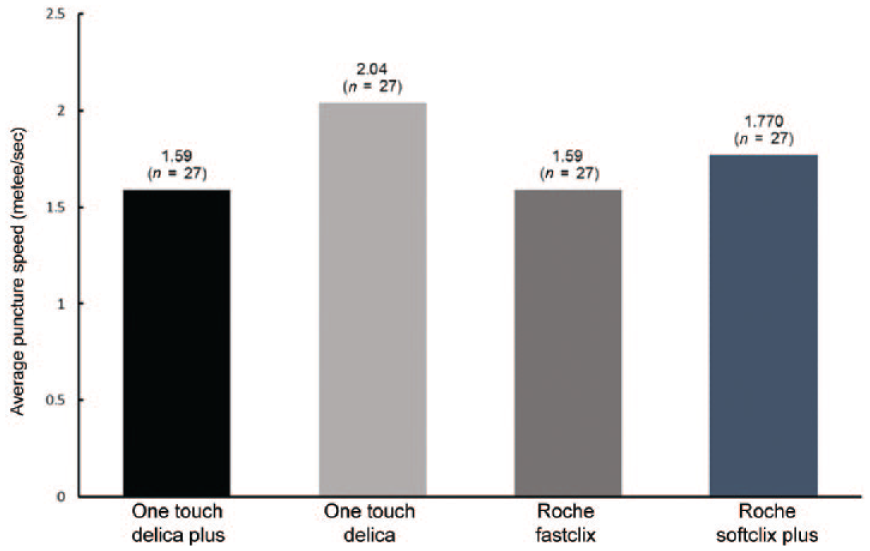
Puncture speed of selected lancing systems.
It is plausible that the evidence of reduced pain (from direct VAS measurements) using OTDP compared to original OTD was due to a combination of all three device factors, namely less vibration, less noise, and a high-speed lancing mechanism. Furthermore, OTDP provides 13 customizable depth settings, whereas original OTD provided only 7 depth choices. By comparison, Roche SoftClix/FastClix each provides 11 settings. Providing more depth settings allows patients to personalize a depth sufficient for them to generate blood at the lowest depth for SMBG. In addition, the 30 and 33 gauge lancets available for OTDP are siliconized to ensure the lancet travels through the skin with minimal friction to limit tissue damage and minimize pain.
In terms of how we evaluate the lancing process, it is helpful to separate lancing into two distinct events whereby we have the initial finger stick itself, followed by a manual “patient-specific” technique of liberating blood from the finger, eg by applying some form of pressure to the skin. Clearly, patients have their own techniques and preferences for the second step in order to liberate a blood drop that the patient felt was sufficient to fill their test strip. We previously demonstrated that patients expressed an average of 3.1 µL of blood after performing routine finger sticks with eight different lancers (including original OTD). 8 In that study, subjects used their own preferred lancing depth and were asked to manipulate their own fingers exactly as they would have done at home to create their desired blood drop. However, the present study was not intended to repeat or mirror that design given that we sought to standardize our procedure (at the lowest depth that could achieve 0.4 µL) to allow us to directly compare both lancing devices (OTD and OTDP). We also standardized the finger manipulation by asking a health care practioner to use the same consistent and gentle technique across all subjects. As a result our average BVs ranged from 1.15 to 1.44 µL but were still significantly above the 0.4 µL threshold required per protocol, which all patients achieved irrespective of the lancing device or lancet gauge used.
A study limitation exists with respect to the calculation of the blood-volume-to-lancer-depth-setting ratio given that at shallow lancing depths the relationship between BV and lancing depth may not be linear. For example, blood can only be obtained when lancing depth is larger than stratum corneum thickness at the lancing site and furthermore in vitro protrusion of a lancet tip may not reflect the actual depth penetration since skin tends to bulge when a lancet tip hits the skin surface.
Ultimately, acceptance and adherence to using any particular lancing system is dependent upon the patients’ experiences. In our feedback surveys, 79% of subjects responded that OTDP was less painful, more gentle (79%), and more comfortable (71%) than their current lancer. Our intention was also to improve upon the original OTD experience and, therefore, it was satisfying to observe that 75% of subjects felt OTDP was more comfortable than the original OTD and that 78% of subjects felt testing with OTDP was virtually pain free.
Conclusion
The new OTDP lancer delivers a lower pain experience than the original OTD lancer and was observed to be less painful than a wide range of current lancing systems. OTDP also provides a better patient experience, in terms of lower noise and less vibration during finger-stick testing and provides a wider selection of depth settings for individualized comfort. Improvements such as this in lancing technology continue to be important to assist patients to comply with the current test regimens recommended by their health-care professional.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG and HC are employees of LifeScan Scotland, Ltd. GL is an employee of APS America and AS is a consultant to APS America. LM is an employee of Synexus USA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by LifeScan, Inc.
