Abstract

Introduction
Continuous quality assessment of blood glucose monitors (BGMs) is not only of clinical importance, but also a normative argument for evidence-based decision making, in particular for Conformité Européene (CE) marking in Europe. The International Organization for Standardization’s (ISO) directive 15197 1 provides harmonized procedures and guidelines to ensure international quality standards for BGMs. Here we present a postmarketing evaluation of the CE-marked TD4216 BGM’s system accuracy.
Methods
System accuracy was assessed in accordance with the requirements stipulated in ISO 15197 at the Institute of Diabetes Karlsburg, Germany, in August 2019. Reference measurements were performed with a Cobas c 111 (Roche Diagnostics GmbH, Germany). Two BMG TD4216 and 1,100 GDH-FAD sensor strips from three lots (WG19B112-CEE, WG19B122-CEE, and WG19B123-CEE) were supplied by Tai Doc Technology Corp. (New Taipei City, Taiwan). One hundred and nine test subjects were enrolled and their blood samples taken to obtain 100 evaluable data sets. Study personnel collected the capillary blood sample from the subject’s disinfected fingertip. Measurements of the blood glucose concentration were performed in duplicate with 2 m BGM using sensor strips from the same vial, repeated for all three lots, and compared with duplicate (prior and after BGM testing) reference values. Environmental conditions met the manufacturer’s requirements (21.3°C-24.1°C, 41%-58% rH). Determination of the hematocrit was performed after measurements using centrifuged samples and a nomogram alignment chart.
Results
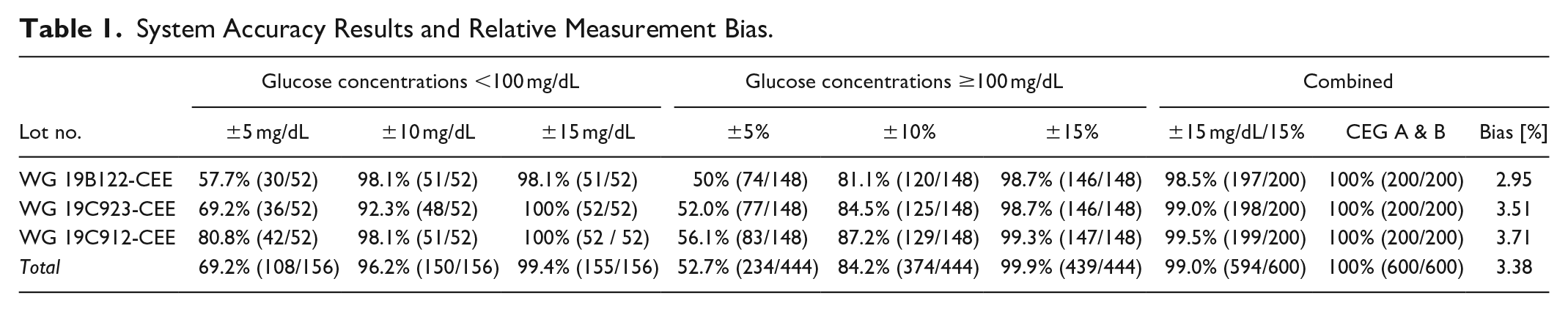
Glucose concentrations distributed in accordance with sampling requirements ranged from 40 mg/dL to 592 mg/dL. Results obtained with the BGM were found to be in compliance with all acceptance criteria stated in the directive. Between 98.1% and 100% of measurements were within ±15 mg/dL/±15% of reference measurements. Between 98.1% and 81.1% reside within ±10 mg/dL/±10%, and between 80.8% and 50.0% within ±5 mg/dL/±5% for glucose concentrations <100 mg/dL and ≥100 mg/dL, respectively (see Table 1). All measurements reside within zones A and B of the consensus error grid (CEG).
System Accuracy Results and Relative Measurement Bias.
Discussion
The “trueness” of measured glucose concentration is estimated by a comparison with a validated reference. Subject to the factual deviation of the BGM and reference values, system accuracy achieves a not to be underestimated relevance with regard to therapeutic decision making, medication and therapy. The requirements stipulated in the directive were fulfilled with 99% of measurements residing within the specified acceptance levels of ±15 mg/dL (±15%), and 100% of measurements in the clinically acceptable CEG zones A and B. A bias analysis revealed negligible differences between sensor strip lots. As all measurements were performed in controlled environments by trained personnel however, the results may not be entirely representative given that measurements performed by laymen often are less accurate. 2
Conclusion
The BGM TD4216 fulfils the criteria for system accuracy. More than 95% of measured capillary blood glucose concentrations did not deviate by more than ±15 mg/dL or ±15% of the reference for glucose concentrations <100 mg/dL and ≥100 mg/dL, respectively. All measurements reside within the clinically acceptable CEG zones A and B.
Footnotes
Acknowledgements
TaiDoc was permitted to review and comment on the manuscript; final decision on content was retained by the authors.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institute of Diabetes Karlsburg, Germany, which carries out studies evaluating blood glucose meter systems on behalf of various companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by TaiDoc Technology Corp., New Taipei City, Taiwan.
