Abstract
Background:
About 25% of adults >70 years suffer from type 2 diabetes. Due to the heterogeneity of the geriatric population, guidelines emphasize the need to individualize glycemic goals and simplify treatment strategies with the main focus of avoiding hypoglycemia. The aim of this study was to assess glycemic control in patients with type 2 diabetes in geriatric care facilities based on their individual health status.
Methods:
170 medical records of older adults with type 2 diabetes in geriatric care facilities were retrospectively assessed (64.7% female, age 80 ± 9 years; glycated hemoglobin 6.8% ± 3.6% [51 ± 16 mmol/mol]; body mass index 27.9 ± 5.8 kg/m2). Based on the individual health status, patients were allocated to three groups (healthy n = 27, complex n = 86, and poor n = 57).
Results:
The overall blood glucose (BG) value was highest in the poor health group with 188 ± 47 mg/dL (poor) vs 167 ± 42 mg/dL (complex) vs 150 ± 34 mg/dL (healthy). BG values of 1.6% (poor) vs 2.8% (complex) vs 1.5% (healthy) of patients were below 90 mg/dL. 36.8% (poor) vs 23.4% (complex) vs 18.5% (healthy) of patients received insulin as the main diabetes therapy, but of these only 14.3% (poor) vs 20% (complex) vs 40% (healthy) were treated with basal insulin.
Conclusions:
Overall, BG values were higher in the poor and complex health group. There were a few low BG values in all groups. Although recommended by international guidelines, basal insulin therapy with its low complexity and low hypoglycemic risk is still underused, especially in the poor health group. Therefore, simplification of diabetes therapy should be considered further.
Introduction
In the United States, the prevalence of type 2 diabetes mellitus (T2DM) is about 26% among adults aged 65 years and older and up to 34% in the long-term care population. 1 Older adults with T2DM are more likely to suffer from a functional disability, hypertension, coronary heart disease, or stroke compared with those with no T2DM. 2 The high percentage of people suffering from T2DM is also associated with rising diabetes costs. 3 To provide optimal quality of diabetes care and to slow down a costly progression of diabetes, a systematic review on this topic concludes with the recommendation to incorporate clinical practice guidelines into policies of health-care facilities. 4 International diabetes guidelines also recommend to implement individual care plans4,5 which include treatment targets that are relevant for the health status of older adults. These targets/plans should also take into account the heterogeneity of older adults in terms of comorbidities and functional impairments.2,6,7 Older adults with T2DM in good general health should have treatment targets comparable to younger adults with T2DM, whereas older adults with T2DM and poor health should have more relaxed treatment goals. The American Diabetes Association (ADA) recommends fasting blood glucose (FBG)/premeal levels of 90-130 mg/dL for older adults with T2DM in good general health (few coexisting chronic diseases, intact cognitive/functional status), 90-150 mg/dL FBG for those with a complex health status (multiple coexisting chronic diseases or 2+ instrumental ADL impairments or mild-to-moderate cognitive impairment), and 100-180 mg/dL FBG for older adults with T2DM with poor health (long-term care or end-stage chronic diseases or moderate-to-severe cognitive impairment or 2+ ADL dependencies). 5 One common goal of all guidelines is to avoid hypoglycemia due to the severe consequences, especially in older adults with T2DM (eg, loss of consciousness, falls leading to fractures or death).5,8-11 Therefore, diabetes medication with low hypoglycemic risk is recommended,9,12-14 and persistent hyperglycemic values, leading to dehydration, susceptibility to infections, or a worsening of cognition, should also be avoided. 15 Diabetes medication accounts for one-fifth of all emergency department visits for adverse drug events in older adults, indicating frequent overtreatment with diabetes medication in this age group. 16
Furthermore, there is still a lack of clinical trial data for older adults with T2DM. This lack is mostly due to the heterogeneity of the older patient population and their frequent exclusion from clinical research. The ADA recommends to collect clinical trial data of older adults with T2DM in geriatric care facilities in order to plan strategies to improve diabetes management in this population. 1
Although diabetes guidelines recommend different glycemic targets for different health statuses of older adults with T2DM, no study has yet evaluated diabetes management according to different health status. Therefore, the aim of this study was to assess retrospectively diabetes management, glycemic control, and safety issues of older adults with T2DM in geriatric care facilities, taking into account the different health statuses of this population. This assessment can serve as a basis to further simplify diabetes therapy in older adults with T2DM in the future.
Methods
The retrospective assessment of medical records was performed in a population of older adults with T2DM in five acute and six long-term care facilities of the Geriatric Health Centers of the city of Graz, Austria (GGZ). To assess an equal number of patient records in acute and long-term care facilities, we considered records of all older adults who stayed at acute geriatric wards between September and November 2014, and records of all older adults in long-term care facilities between May 2014 and April 2015. The retrospective assessment was approved by the local ethics committee of the Medical University of Graz (EK-Nr. 27-471 ex 14/15) and was performed in accordance with the Declaration of Helsinki and the principles of Good Clinical Practice.
Medical records of all patients aged ≥50 years (due to the admission criteria of the geriatric facilities) with a documented diagnosis of T2DM were included in the retrospective assessment. Data from patients were excluded if they had stayed in the facility for less than 1 week or if medical data were incomplete. All data were examined regarding diabetes management, glycemic control, and the safety of diabetes therapy during the stay. Analysis parameters were chosen following the United Kingdom National Diabetes Inpatient Audit (NDIA). 17 Standardized electronically documented data were automatically extracted for analysis, and paper-based data were captured in the clinical data management system OpenClinica (OpenClinica, LLC, Waltham, MA, United States). All assessed facilities used physician-based routine care for diabetes management with no diabetes guidelines and no diabetes-related trainings for health-care professionals implemented in any assessed facility.
To take into account the heterogeneity of the assessed older adults, all patients were allocated to three subgroups (healthy, complex, poor health) depending on their health status.
In Austria, care dependency is assessed and expressed as care time needed as hours per month. We assigned older adults in need of care of 65-119 hours/month (Pflegestufe 1 or 2) to the group “healthy”, those with 120-179 hours/month (Pflegestufe 3 or 4) to the group “complex”, and older adults in need of extraordinary care of 180 hours/month or more (Pflegestufe 5-7) to the group of “poor” health. 18 In the acute geriatric care setting, older adults were allocated by a geriatrician considering comorbidities of the included patients. Chronic diseases with severe adverse effects on health (dementia, cardiac insufficiency) had a value of two points each and diseases with considerable effects on health (cardiac infarction, coronary heart disease, impaired kidney function, osteoporosis, other relevant diagnoses [eg, malignancy] and >85 years of age) had a value of one point each. Patients with a total of zero to two points were allocated to the group “healthy”, those with three to four points to the group “complex”, and those with five or more points were allocated to the “poor” health group. The degree of care was not available for patients in the acute geriatric facilities because they are admitted due to an acute medical condition, and thus, the degree of care is not recorded. To be able to include data from both types of facilities, we decided to perform the described assignment.
Statistical Analysis
Patient data were analyzed retrospectively in terms of gender, age, body mass index, serum creatinine, comorbidities, mean glycated hemoglobin (HbA1c) values, mean blood glucose (BG) values at different times of day, main diabetes therapy, mean number of BG measurements according to diabetes therapy, number of low (≤90 mg/dL, ≤72 mg/dL) and high (≥300 mg/dL) BG values, and emergency department visits. All data were analyzed according to health status and per patient stay. An exploratory descriptive statistical analysis was performed. Numerical data are presented as arithmetic mean ± standard deviations. For categorical data, proportions were used to describe the outcome data. We used the Kruskal–Wallis rank sum test (metric variables) to determine significant differences (for different levels of significance α = 0.05, α = 0.01, and α = 0.001) of mean BG values and BG values within certain ranges among the three groups of older adults. We used the Mann–Whitney U test to further determine statistically significant differences among particular groups (healthy vs complex, complex vs poor, and healthy vs poor). All statistical analyses were performed with R (v. 3.1) and RStudio (v. 1.0.136). 19
Results
In the assessed geriatric health-care facilities, the documented diabetes prevalence in 2014 was 17.6%. In the (pre-)defined time frames of our assessment, a total of 180 older adults with a diagnosis of T2DM had stayed in the assessed facilities. Of these, 10 patients had to be excluded because they had stayed in the facility for less than 1 week (n = 9) or had an incomplete medical record (n = 1). Altogether, 170 medical records of older adults with T2DM were analyzed (100 records of older adults in acute geriatric wards and 70 records of older adults in long-term care facilities). Twenty-seven older adults were allocated to the “healthy” group, 86 to the “complex”, and 57 to the “poor” health group. Patient characteristics are presented in Table 1.
Clinical Characteristics of Older Adults and Main Diabetes Therapy According to Health Status.
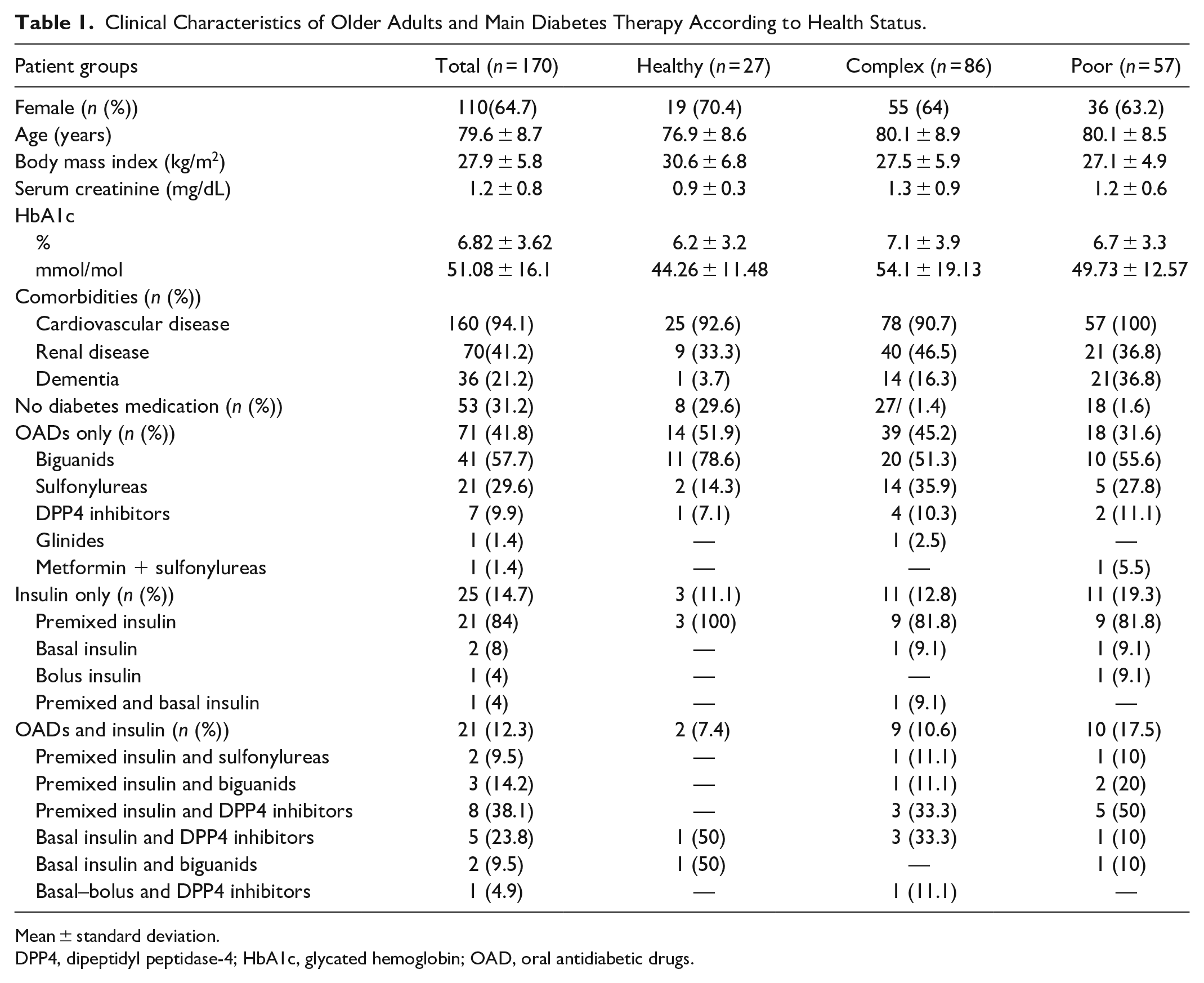
Mean ± standard deviation.
DPP4, dipeptidyl peptidase-4; HbA1c, glycated hemoglobin; OAD, oral antidiabetic drugs.
Diabetes Management
The majority of all older adults (41.8%, n = 71) were treated with oral antidiabetic drugs (OADs) only, 31.2% (n = 53) had no diabetes medication, 14.7% (n = 25) were treated with insulin, and 12.3% (n = 21) with a combination of OADs and insulin (Table 1). In the OAD group, the majority of patients were treated with biguanides, followed by sulfonylureas and dipeptidyl peptidase-4 (DPP4) inhibitors. The majority of those older adults treated with insulin were administered premixed insulin, followed by basal insulin therapy, bolus insulin therapy, basal–bolus insulin therapy, or a combination of premixed and basal insulin (Figure 1). A glycemic target range was mentioned in only five out of all medical records (2.9%). On average, BG measurements were taken on 3.5 ± 2.2 days per week in all patients; 59.3% (healthy), 47.7% (complex), and 61.4% (poor) of the assessed records had an appropriate BG monitoring frequency on at least 1 day during stay when compared with the United Kingdom NDIA recommendations. 16
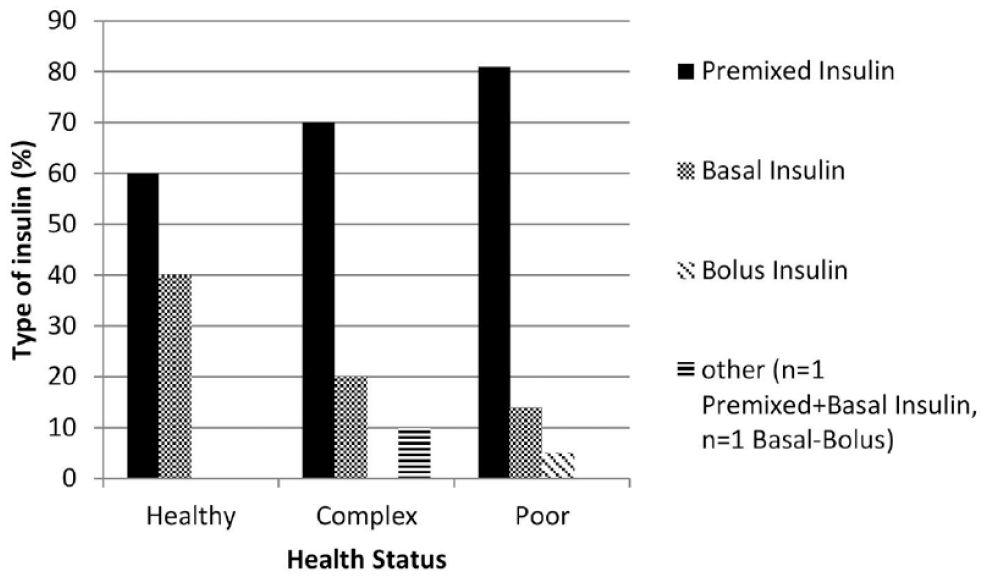
Different types of insulins according to health status.
Glycemic Control
We evaluated a total of 4644 BG values that were documented in the 170 assessed records (healthy n = 552, complex n = 2324, poor n = 1768). The mean overall BG value was 171 ± 45 mg/dL for all groups (acute geriatric care vs long-term care facilities: 162 ± 43 mg/dL vs 186 ± 43 mg/dL) and lowest in the healthy group with 150 ± 34 mg/dL vs 167 ± 42 mg/dL in the complex vs 188 ± 47 mg/dL in the poor health group. BG values were lowest in the morning in all groups and highest at bedtime (healthy, complex) or in the afternoon (poor) (Figure 2); 37.4% (healthy) vs 44.6% (complex) vs 64.4% (poor) of all FBGs and 26.3% (healthy) vs 33.9% (complex) vs 39% (poor) of all BG values were in the target ranges that are recommended by the ADA 2 (Table 2). Conducting hypothesis tests resulted in no significant differences in BG value target ranges among the three groups. However, when comparing BG values, significant differences among the target groups were shown for the overall BG value (P < .001), as well as the midday (P < .05), afternoon (P < .01), and night BG value (P < .05). HbA1c measurements were available for 77.8% (healthy), 62.8% (complex), and 57.9% (poor) of all older adults; the mean HbA1c values were 6.2% ± 3.2% (44.26 ± 11.48 mmol/mol), 7.1% ± 3.9% (54.1 ± 19.13 mmol/mol), and 6.7% ± 3.3% (49.73 ± 12.57 mmol/mol), respectively.
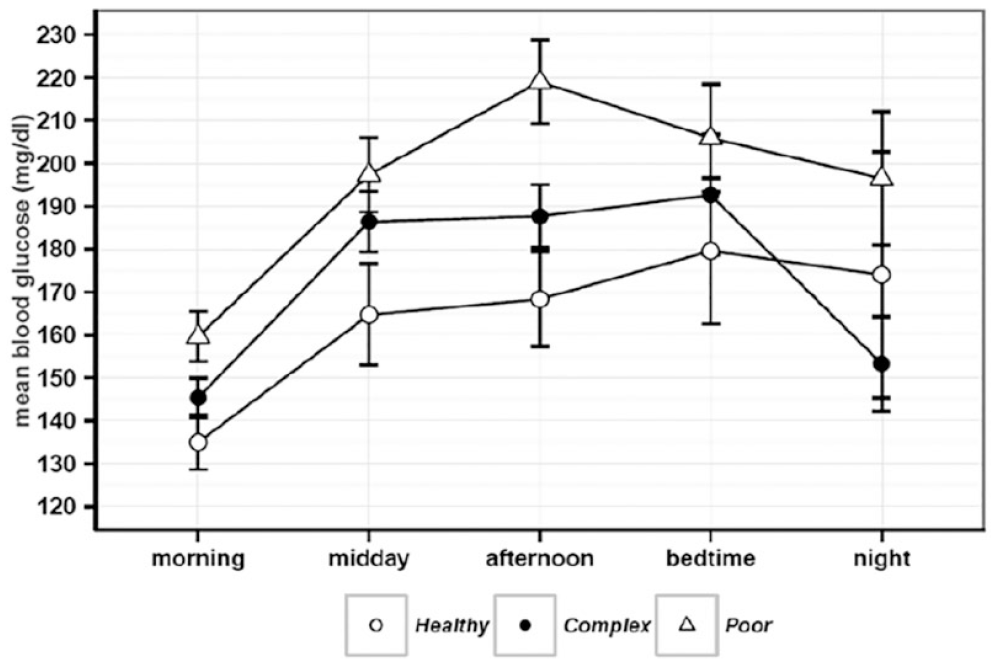
Overall mean blood glucose (BG) values (±standard error) at different times of the day according to health status (significant differences of BG values: midday (P < .05), afternoon (P < .01), night (P < .05)).
Overall BG Value in different Glycemic Ranges.
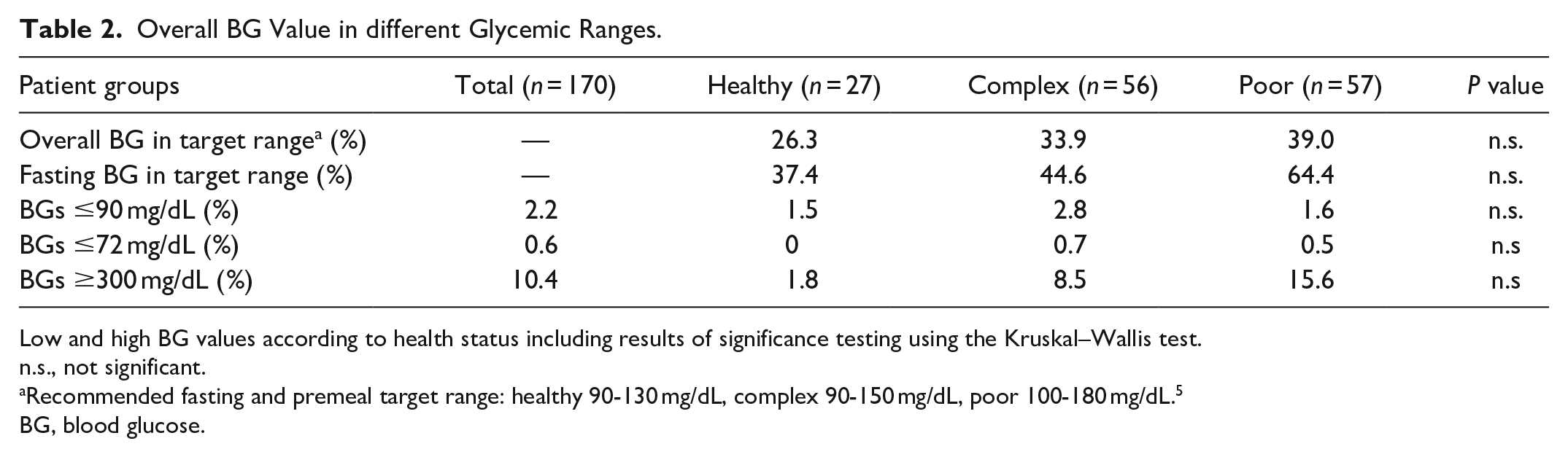
Low and high BG values according to health status including results of significance testing using the Kruskal–Wallis test.
n.s., not significant.
Recommended fasting and premeal target range: healthy 90-130 mg/dL, complex 90-150 mg/dL, poor 100-180 mg/dL. 5
BG, blood glucose.
Safety Issues
All in all, we found only 0.6% (n = 24) BG values <72 mg/dL in our assessment. Patients experienced these values as symptomatic hypoglycemic events in 0% (healthy) vs 47.1% (complex) vs 28.6% (poor). High BG values >300 mg/dL occurred in 10.4% of all BG values and most often in the poor health group (Table 2).
A total of 122 falls occurred during the assessment period, but none was associated with a BG value below 90 mg/dL. During the evaluation period, a total of 23 events required emergency department visits (healthy n = 5, complex n = 10, poor n = 8) for various reasons (eg, fall, exacerbation of pulmonary disease), but none was related to hypoglycemic or hyperglycemic episodes.
Discussion
In this study, we assessed glycemic management according to health status in older adults staying in acute geriatric and long-term care facilities. This is the first study that takes into account both health status and glycemic control of institutionalized older adults as recommended by international guidelines.5-7
Our results show satisfactory average BG values of 150 mg/dL (healthy), 167 mg/dL (complex), and 188 mg/dL (poor), and few low BG values in all health groups. We found higher overall BG values in the complex and poor health groups compared with the healthy group which is in accordance with guideline recommendations that advise relaxed glycemic targets for older adults with poor health.5,6,12 However, only 37.4%-64.4% of all FBGs were in the FBG target ranges that are recommended by the ADA. 5 This could indicate that the recommended tight FBG target ranges, in particular for older adults with T2DM in good health (recommended FBG target: 90-130 mg/dL), are rather difficult to reach in daily routine diabetes therapy.
We found that most of the insulinized older adults in the poor health group were treated with premixed insulin and one person had bolus insulin as the main diabetes therapy. Guidelines recommend a diabetes therapy regime that is as simple as possible and has a low hypoglycemia risk for older adults with T2DM.11,15 Long-acting insulins are the method of choice,6,9,10,12,20-24 whereas the routine use of rapid-acting insulin should be avoided in older adults with T2DM, due to the high risk of hypoglycemia.1,6,23 Contrary to guideline recommendations, only 40% (healthy), 20% (complex), and 14.4% (poor) of the older adults treated with insulin in our study used basal insulin. Results of other studies confirm our findings, showing rather complex diabetes therapy regimens applied to a large number of older adults with T2DM.25-30 Further research could investigate whether older adults with poor health are also able to reach the recommended FBG targets when using a less complex diabetes therapy regime, for example, basal insulin therapy as recommended by diabetes guidelines.
As with BG targets, guidelines recommend HbA1c targets depending on the health status of older adults with T2DM. For healthy older adults with T2DM, a similar HbA1c as for younger adults is recommended, whereas for those with complex or poor health higher values are acceptable.6,11,15,22 In our evaluation, even patients with poor health fulfilled HbA1c criteria for healthy/young adults with T2DM. The discrepancy between average BG values and average HbA1c values in our evaluation could be due to several reasons. One reason might be that HbA1c values were only available for about two-thirds of all studied patients, possibly reflecting mainly those with good glycemic control. Another reason could be anemia in old age 31 which leads to HbA1c values that are falsely low.
Many diabetes guidelines suggest to determine a BG measurement frequency on an individual basis13,32,33; only the American Medical Directors Association (AMDA) 6 and the NDIA 17 recommend standardized BG measurement frequencies. We chose the NDIA criteria for our study since the criteria used by the NDIA (at least one measurement/week for diet or metformin, at least two measurements/day for insulin, sulfonylureas, DPP4 inhibitors, glitazones, sodium-glucose cotransporter-2inhibitors or glucagon-like peptide 1 analogs and at least four measurements/day for basal bolus insulin) are less stringent compared with the AMDA recommendations (two measurements/day on 1-2 days/week for non-insulin, two measurements/day on 3-4 days/week for simple insulin regime with 1-2 injections/day, and at least four measurements/day for complex insulin regimes with three or more injections/day). Even when less stringent criteria were used, less than 50% of the older adults in the complex health group in our study had just 1 day with appropriate frequencies of BG measurements during their stay, compared with about 60% in the healthy and poor health groups. Therefore, it could be considered to adapt the recommendation and take into account current glucose control and hypoglycemia risk in older adults with T2DM, especially those staying in long-term care facilities. For older adults with stable and simple insulin therapy, the BG measurement frequency could be reduced, whereas patients with a high hypoglycemic risk need more frequent BG measurements. In the near future, electronic decision support systems can assist health-care professionals when recommending a safe BG measurement frequency according to the chosen health status, diabetes therapy, and individual needs of older adults. 34
It would have been interesting to obtain data of older adults using CGM to gain a deeper insight into BG variation of this population. However, in Austria, CGM is only paid under certain conditions that do not apply to older adults in geriatric care facilities, so none of the patients in our retrospective study used CGM.
Low BG values and symptomatic hypoglycemic events were rare in our study. Only 0.6% (n = 26) of all BG values were below 72 mg/dL. A comparable study revealed symptomatic hypoglycemic events in 9.1% of the older patients. 30 The reason for the low number of hypoglycemic events in our study could be the low average frequency of BG measurements. As stated before, there was an inadequate BG measurement frequency according to the NDIA criteria in 40%-50% of all studied persons. Older adults also show both less specific signs of hypoglycemia and lower awareness hypoglycemia symptoms, which makes it difficult to detect hypoglycemic events in this age group. 17 A recent study revealed that BG measurements had only been taken in half of older adults with T2DM experiencing episodes of acute confusion, falls, irritability, somnolence, and nonspecific unwellness, which could all be signs of hypoglycemia. 35 In our evaluation, only 28.6% (n = 2) of all BG values below 72 mg/dL in the poor health group were experienced as symptomatic hypoglycemic events, possibly indicating that in older adults with T2DM both health-care professionals and patients are less aware of hypoglycemia. This highlights the need for diabetes-related training for health-care professionals in geriatric care facilities.
BG values were lowest in the morning in all groups and highest at bedtime or in the afternoon, possibly indicating that high values in the afternoon/evening are tolerated to avoid hypoglycemia during nights.
No patient in our evaluation had to be sent to an emergency department due to low or high glycemic values, although other comparable studies revealed that emergency department visits due to hypoglycemia are common in older adults with T2DM, with frequencies varying from 1% to 8%.27,36,37
Conclusions
To summarize the results of our evaluation, we recommend individualization of diabetes therapy in older adults with T2DM that takes into account the individual health status to guarantee a safe diabetes therapy and to avoid hypoglycemic events. In the future, we address the issue of simplifying and finding the best fitting diabetes therapy and BG measurement frequency by developing and implementing electronic decision support systems especially focusing on geriatric needs to assist health-care professionals in the daily routine work.
Footnotes
Acknowledgements
The authors acknowledge the critical review of the manuscript and the editorial assistance of S. Mautner, Medical University of Graz, data management of B. Tschapeller, C. Krainer, Joanneum Research GmbH, data monitoring S. Bischof, Medical University of Graz. The authors thank the Geriatric Health Centers of the City of Graz for their contribution in providing data.
Author Contributions
AL, KL, AC, TW, BH, PB, and WS designed the study, interpreted data, and contributed to discussions. AL drafted the manuscript. GC and KD performed the statistical analysis. JP and KL supervised the project. TP interpreted data, contributed to discussions, supervised the project and is the guarantor of this work. All authors critically revised the article and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Austrian Research Promotion Agency (Grant number 844737).
Prior Presentation of Data
Parts of this study were presented at the International Congress of the European Union Geriatric Medicine Society (2016, Lisbon, Portugal), the Annual Meeting of the Austrian Diabetes Federation “Österreichische Diabetes Gesellschaft” (2016, Salzburg, Austria), the 11th International Conference on Advanced Technologies and Treatments in Diabetes (2018, Vienna, Austria), and the 24th Nordic Congress of Gerontology (2018, Oslo, Norway).
