Abstract
Background:
Closed loop (CL) automated insulin delivery systems are demonstrated to be safe and effective in regulating glucose levels and reducing cognitive burden in people with type 1 diabetes (T1D). However, given the limited market options and the do-it-yourself nature of most systems, it can be difficult for potential users to shape their expectations fitting them into daily lives and management routines. As such, we examined the potential feasibility of a virtual reality (VR) intervention.
Methods:
A four-part VR intervention was created to expose adults with T1D to expected CL system barriers: body image, perceived hassles of using CL, deskilling fears, and unwanted social attention. Goals of the pilot were to assess feasibility and expose patients to CL. Surveys were conducted pre- and postparticipating in the VR experience.
Results:
A total of 20 adults with T1D completed the pilot. Average time to complete the experience was 14.1 minutes (8.8-39.9). Reported VR sickness was low. Willingness to use VR was maintained in 90% (n = 18) and did not change expectations of CL in 95% (n = 19). Virtual reality changed perceived hassles of CL in 25% (n = 5) with four concerned over alarms and one connectivity issues: positive diabetes technology attitudes, confidence in managing hypoglycemia, overall perceptions of appearance, and positive affect maintained after the VR intervention. Negative affect significantly decreased after exposure and perceptions of being overweight trended toward significance.
Conclusion:
This pilot VR intervention demonstrated high potential in addressing expected barriers to uptake and usage of CL systems without decreasing enthusiasm or changing expectations of CL.
Introduction
Closed loop (CL) automated insulin delivery systems represent a major advancement in type 1 diabetes (T1D) management, as they are designed to automatically and precisely regulate glucose levels and reduce cognitive burden, and in this study, we examined the potential role and efficacy of virtual reality (VR) in addressing potential barriers to CL use and uptake. Closed loop systems are composed of four parts: (1) insulin pump, (2) continuous glucose monitor (CGM), (3) the mathematical algorithm predicting insulin needs, and (4) an operating system running the algorithm and communicating with devices. There is much reported research on known barriers to insulin pumps and CGM’s, some reported barriers from clinical trials on the algorithm and operating system, and none of Food and Drug Administration (FDA) approved on-market full CL systems.1-5 In clinical trials, CL systems have been demonstrated to be safe and effective in increasing time in target range, decreasing mean glucose levels, and improving health-related quality of life through clinical studies.6-11 Developments in CL technology have been rapid, with Medtronic releasing the first hybrid CL system MiniMed 670G in 2017, wherein the hybrid approach includes meal announcement with an estimated carbohydrate intake and a premeal bolus of insulin, and a number of other CL systems are in the pipeline for future release. Preset settings, alarms, and calibration are not modifiable at this time with this system, which research has shown to be a barrier to uptake. 12 In response, do-it-yourself (DIY) CL systems that are open source algorithms paired with the existing insulin pumps have emerged from T1D communities as a more customizable option, and both hybrid and DIY CL systems have shown similar improvements in clinical settings13,14 and with real-world use.15-18 However, in order for any CL systems to be successful long term, adults and youth alike need to invest in using them and their component parts. 19 It can be difficult for potential users to shape their expectations for how the one FDA approved hybrid CL system and varied DIY CL systems might fit into their daily lives and management routines. As such, interventions focusing on addressing known barriers from uptake with component parts and uptake of CL systems in randomized clinical trials (RCTs) are needed to increase the uptake of CL systems and their component parts.
Virtual reality is an effective and feasible strategy to expose individuals to vivid virtual worlds not otherwise accessible in the real or “physical” world, 20 and these worlds allow individuals to experience visceral environmental cues 21 paired with emotion producing experiences, built to enhance behavior change. 22 Virtual reality has been successful in positively impacting implicit biases and discrimination by increasing empathy23-25 as well as improving public speaking by overcoming anxiety. 26 Furthermore, in the healthcare setting, VR has been used to address issues such as weight management, 27 body image disturbance, 28 and chronic pediatric pain, 29 demonstrating high translational potential in effecting health behavior change related to optimizing uptake of CL systems. Prior studies have shown that the most common perceived barriers to using diabetes devices are (1) worry that the device will physically change the way the body looks and feels; (2) perceived added hassles of wearing CL; (3) fear of deskilling with foundational diabetes management tasks; and (4) worry that the device will draw attention in social situations.1,30 Although some of the research behind these barriers are specific to CGM and insulin pump use, these themes came from a large international qualitative study of expectations, worries, and hopes for CL systems from users who used component parts, some who had participated in the RCT’s of CL systems, and others who had not used component parts 31 ; we expect many barriers for FDA approved on-market CL systems and DIY CL systems to be similar since they use combinations of the same component parts. Each of these critical intervention points provides an opportunity for targeted intervention with VR: (1) virtual exposure to wearing a CL system, (2) setting realistic expectations for patients to optimize use of CL systems, (3) increasing situational confidence of using a CL system across contexts, and (4) improving problem solving and decision making skills. Thus, the primary purpose of this study was to determine the feasibility, acceptability, and preliminary efficacy of this VR intervention and to provide preliminary data that can be used in a future, large-scale randomized controlled trial.
In this study, we aimed to test the feasibility of a four scenario VR intervention on addressing expected barriers to CL systems (future FDA approved systems and DIY CL systems). We expected this VR experience to expose people with diabetes to common difficulties in using CL without diminishing their willingness to use CL. We hypothesized that there would be changes in perceived barriers to CL use, perceived hassles of using CL, and diabetes device attitudes; however, we did not hypothesize a direction of effect given the novelty of this type of VR exposure and because of the pilot nature of the project where we powered to detect differences.
Methods
Photon Biomedical, a medical VR company, helped to create a VR experience to simulate the use of a CL system in a work setting. This company was chosen as they were already engaged with healthcare systems and education. Videos demonstrating CL systems were provided to the makers of the VR program prior to building out the scene, and people with diabetes and diabetes medical professionals were involved in the creation process to ensure accurate portrayal of situations related to diabetes technology use. The VR experience consisted of four behavioral interventions addressing common barriers to CL device use: devices and body image, perceived hassles of using CL, the fear of becoming de-skilled, and unwanted social attention. The HTC Vive was used to run the VR experience.
Once within the VR setting, an introductory session was completed that taught participants how to use the VR system and allowed participants to personalize their avatar to better represent themselves. Then, the participant navigated through a normal workday on their new CL system. First, they went to the bathroom and looked at themselves in the mirror where they could see the CL system clearly on their torso followed by overhearing colleagues ask about “the new strange beeper” they are wearing, to address the devices and body image barrier. Then, the participant walked to their cubicle to start working when a connection failure alert noises and the participant has to rearrange their device to achieve better connection. Then, another alert sounds resulting in the need to change the insulin pump cartridge (Figure 1). These scenarios aimed to address the hassles of using CL barrier. Then, the participant went to lunch and had to choose whether to trust the system to treat a high blood sugar and the lunch carbohydrates or override the system to give extra insulin, to address the fear of becoming deskilled barrier as well as honing in on how much they trust the system. Finally, the participant went to get coffee in a cafe with coworkers when a CL system alert goes off and the participant is asked questions about their devices to address the unwanted social attention barrier. Each of these scenarios involves various active interactions and decisions made by the participant.

Closed loop alert example in virtual reality experience.
The goal of these interventions was to expose patients to CL and some of the hassles associated with use, agnostic of brand, and type of pump, or whether they are DIY or soon to be on-market; therefore, the VR CL system incorporated elements from all available systems both DIY and mainstream in an unbranded presentation. The insulin pump and alert sounds were similar to the commercially available 670G system, while the CL interphase appeared on the user’s iPhone, similar to DIY systems. The user was able to see both of these elements either in the mirror or by looking at their hand when alerts went off to view the phone. Questionnaires were conducted right before the VR experience and directly after the VR experience.
Twenty participants with type 1 diabetes ≥18 years old with a wide variety of previous diabetes device usage were recruited.
Measures
Participants reported their date of birth, date of diabetes diagnosis, sex, ethnic group or race, method of insulin delivery, current CGM usage, most recent Hemoglobin A1c, income, and level of education.
Diabetes technology attitudes 32 : Attitudes toward diabetes technology were assessed with five items rated on a five-point Likert scale, previously validated 32 with five indicating positive attitudes and one indicating negative attitudes. Questions included items such as “diabetes technology has made my life easier” and “Diabetes technology takes more time and work than it is worth.” Scores were summed to create a total score ranging from 1 to 25. Higher scores indicate more positive diabetes technology attitudes.
Hypoglycemia confidence 33 : Confidence in managing hypoglycemia was assessed using nine items including questions such as “How confident are you that you can avoid serious problems due to hypoglycemia?” and “How confident are you that you can stay safe from serious problems with hypoglycemia while you are sleeping?” Questions were rated on a five-point Likert scale with higher scores indicating increased confidence in hypoglycemia management. Responses were summed to create a total score ranging from 9 to 36. Higher scores indicate more confidence in managing hypoglycemia.
Positive and negative affect 34 : Mood was measured using the 20-item positive and negative affect schedule (PANAS) scale. Participants were asked to indicate the extent to which they have felt moods such as “excited,” “scared,” “nervous,” or “jittery” over the past week on a five-point Likert scale with one indicating “very slightly or not at all” and five indicating “extremely.” Positive and negative items were scored separately to create two sums each ranging from 10 to 15. Higher scores indicate higher affect.
Multidimensional body-self relations questionnaire appearance scale (MBSRQ-AS) 35 : The MBSRQ-AS questionnaire was used to determine participants attitudes toward their body image. All items were rated on a five-point Likert scale. The appearance evaluation consists of seven items including “I like my looks just the way they are” and “most people would consider me good-looking.” Higher scores indicated more satisfaction with personal appearance. The appearance orientation evaluation, a measure of the extent of cognitive and behavioral investment in appearance, consisted of 12 items including “I check my appearance in a mirror whenever I can” and “It is important that I always look good.” Higher scores indicated placing more importance on how they look. The body satisfaction evaluation consisted of nine items where they rated how dissatisfied or satisfied they are with various aspects of their body including “Face,” “Mid torso,” and “muscle tone.” Higher scores indicated increased content with areas of their body. The overweight preoccupation evaluation consisted of four items including “I am very conscious of even small changes in my weight” and “I have tried to lose weight by fasting or going on crash diets.” Higher scores indicate being more occupied about being overweight. Finally, self-classified weight was evaluated with two items including “From looking at me, most other people would think I am:” with one indicating very underweight and five indicating very overweight. Scores for each evaluation were summed.
Virtual reality sickness questionnaire (VRSQ) 36 : The VRSQ was utilized to measure VR sickness after VR intervention. Participants rated how strongly they felt various symptoms including “fatigue,” “drowsiness,” and “dizziness” on a scale from zero to six, with zero indicating none, and six indicating severe. There were a total of 13 items, and scores were summed. Higher scores indicate more VR sickness.
Virtual reality experience data: During the VR experience, participants had the option of choosing to ignore CL system alerts or address it in various ways. If alerts were ignored, the CL system would alert again, resulting in another set of options on how to respond. This resulted in different numbers of decision points for participants. Participants with more decision points ignored alerts, chose to not trust the CL system, or were uncomfortable with addressing devices in social situations. The total time to complete the VR experience was also collected.
Open-ended questions: Participants were asked several questions at the end of the VR experience on whether it changed their expectations of CL, their willingness to use CL, or their perception of the hassles of using CL. Participants were then asked to explain their response. Participants were also asked to rate “How realistic did the virtual reality scenarios feel?” on a scale from 0 (not at all) to 100 (very realistic) followed by an open-ended explanation.
Analysis
Descriptive statistics were run on all demographic variables for the sample. Following this, a Pearson product-moment correlation coefficient was computed to assess the relationship between demographics and study variables before and after exposure to the VR intervention. A correlation table was created with all study variables to examine correlations between measures, given that no direction of effect was hypothesized a priori. Total scores for each measure from before VR exposure and directly after VR exposure were compared via independent t-tests. Cohen’s D effect size was calculated to determine the measure of magnitude. Given the small sample size and pilot nature of the data, moderation analyses were not possible, however, the sample was split into groupings by age and the results were compared between young adults (18-29) and older adults (30-45) to determine whether age correlates with VR sickness, amount of time to finish the VR experience, or other outcomes.
Results
Demographics are shown in Table 1. Twenty participants completed the VR experience with surveys before and after exposure (age 28.4 ± 8.0, 50% female). A total of 15% of participants reported they were Asian or pacific islander, and 85% reported white, non-Hispanic race/ethnicity. The median length of diabetes duration was 13.2 years (Interquartile range [IQR] 10.2-21.8).
Patient Characteristics.
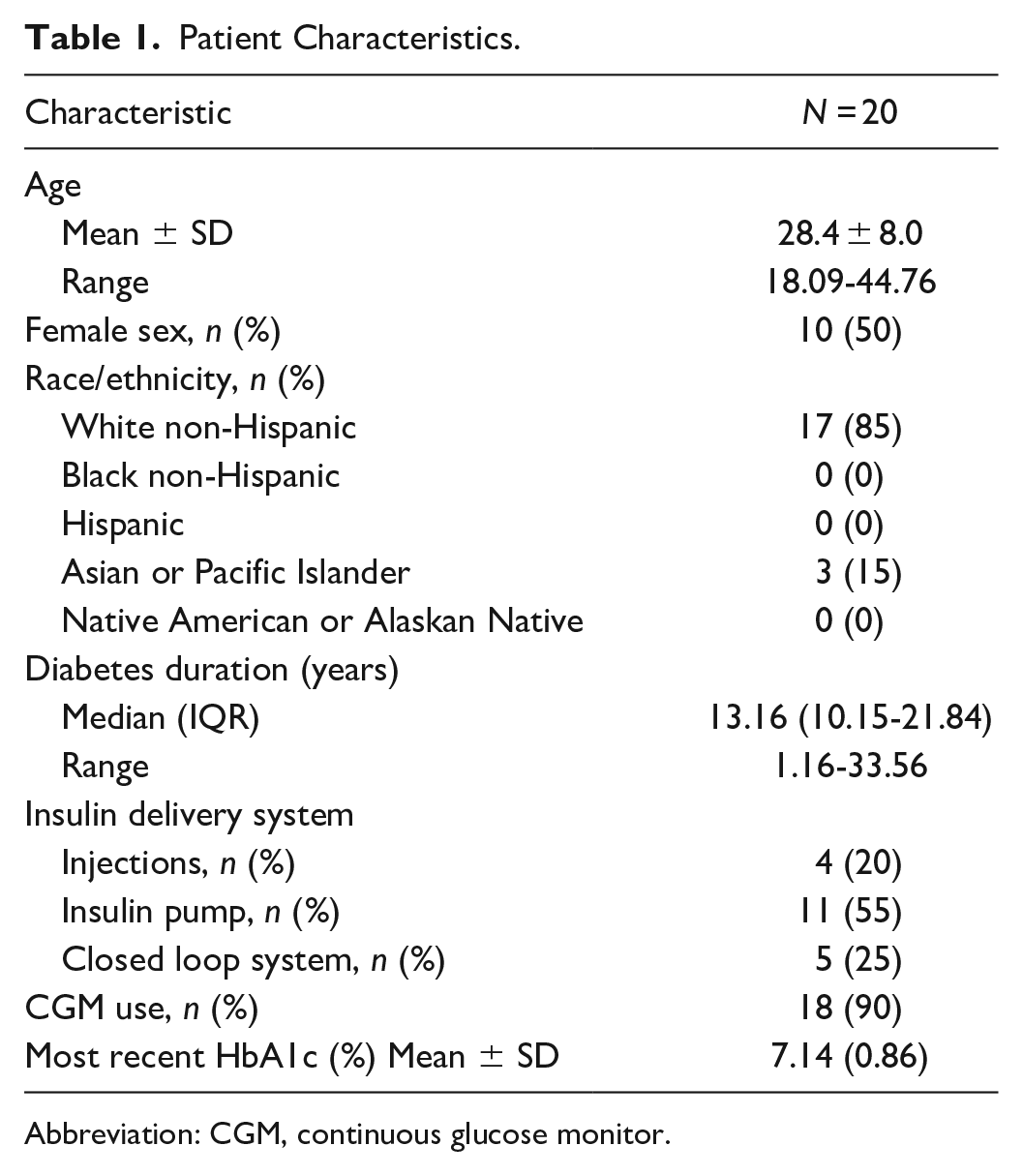
Abbreviation: CGM, continuous glucose monitor.
As shown in Table 2, age was significantly correlated with MBSRQ-AS self-classified weight both before and after the VR experience, with older individuals indicating that they weigh more (P = .05). Length of diagnosis significantly correlated with MBSRQ-AS self-classified weight only after the VR intervention experience (P = .05). Participants of the male sex were more likely to rate positive affect higher than females before VR exposure (P = .01). The current use of CGM significantly correlated with willingness to use CL such that the current CGM use decreased one’s willingness to use CL (P = .05). Also, the current use of CGM positively correlated with higher self-classified weight (P = .05). HbA1c showed no significant correlation with any measures.
Pearson’s r Correlation Analysis.
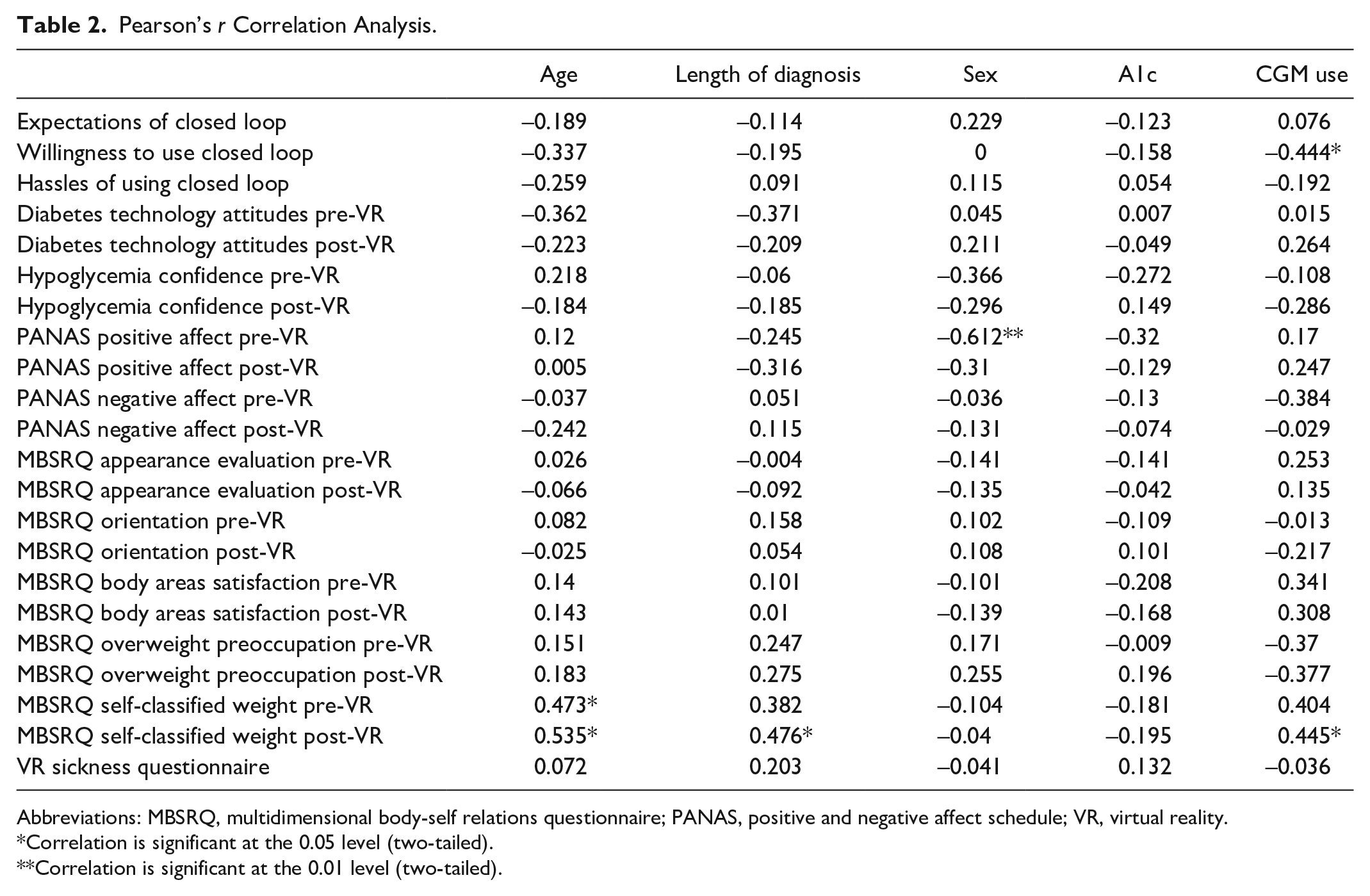
Abbreviations: MBSRQ, multidimensional body-self relations questionnaire; PANAS, positive and negative affect schedule; VR, virtual reality.
Correlation is significant at the 0.05 level (two-tailed).
Correlation is significant at the 0.01 level (two-tailed).
As shown in Table 3, examining t-tests, the PANAS negative affect significantly decreased after the VR intervention (P = .008). Multidimensional body-self relations questionnaire overweight preoccupation trended toward significance (P = .090) with an overall decreased preoccupation with being overweight. Overall levels of VR sickness (VRSQ) were low and not significantly different among younger vs older participants (P = .393). Positive diabetes technology attitudes, confidence in managing hypoglycemia, overall perceptions of appearance, and positive affect did not diminish after the VR intervention.
Survey Measures Before and After Virtual Reality Intervention Experience.
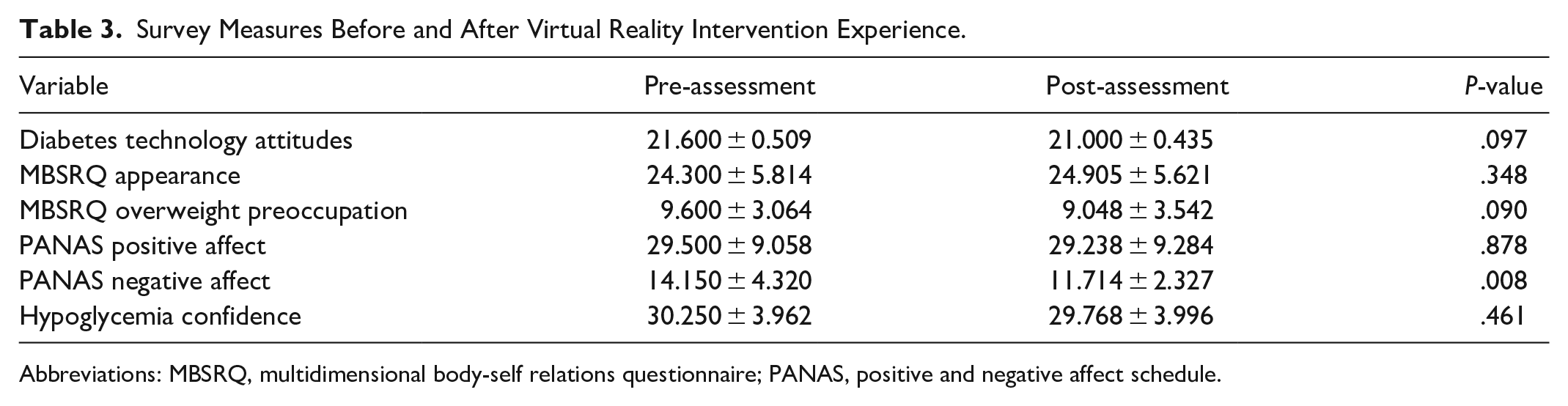
Abbreviations: MBSRQ, multidimensional body-self relations questionnaire; PANAS, positive and negative affect schedule.
The average time it took to complete the VR scenarios was 14.1 minutes (range: 8.8-39.9), and no significant difference was observed between older (>30 years) and younger (<30 years) participants. A total of 90% of participants (n = 18) reported that VR did not change their willingness to use CL, and in open-ended text, the majority shared that they would be willing to use a CL system regardless of barriers and imperfections. A total of 45% of participants never silenced alarms, trusted the CL system, and directly addressed the CL system in social situations during the VR experience, and no significant difference was observed between older (>30 years) and younger (<30 years) participants. A total of 95% (n = 19) reported that VR did not change their expectations of CL. A total of 25% (n = 5) reported that VR changed their perceived hassles of using CL with four expressing concern over alarms and one over connectivity issues. On a scale from 0 to 100, participant rated how realistic the experience felt at an average of 57 (range 5-90, SD 23.7). Those that rated the realism of the experience below 50 mostly mentioned that they felt they were unable to properly communicate their opinions in the social situation experiences, difficulties with setting the proportions of their avatar, and the lack of diversity in surrounding characters.
Participants who were already using CL (MiniMed 670G and DIY systems) mentioned that the hassles experienced in the VR interventions were realistic, but that those hassles were worth the effort compared to manual diabetes management.
Discussion
Although few significant differences in survey responses before and after the VR experience were found, the main objective of the study to test feasibility was successful, as we found that using VR to address barriers and difficulties of using CL systems is feasible. The four intervention modules in combination with the instructional module took an average of 14 minutes to complete, which is short enough to maintain attention but long enough to be valuable to the user. There were no significant difference in VR sickness between younger (<30 years) and older (>30 years) participants, indicating that this kind of intervention can be used across ages.
Despite being exposed to several difficulties related to a CL system use including recurrent connection loss and social situations, the VR intervention did not decrease participant’s willingness to use CL or change expectations of using CL. This indicates that this type of intervention may be useful to expose potential CL users to common difficulties of using a CL system before use without losing interest in the system overall. Virtual reality exposure therapy has been shown to be effective in reducing anxiety 37 and many of the barriers addressed in this VR experience are a source of anxiety for potential CL system users (eg, unwanted social attention and alerts going off in public). Although we were unable to show a reduction of worry about these barriers in this pilot study, exposure to them also did not decrease overall willingness to try a CL system and further exposure to these situations may be beneficial to potential CL system users so that they can anticipate the barrier and work through it prior to using the system.
The VR experience may also be a useful tool to help clinicians teach potential CL users about the system before they accept financial responsibility for the system. Furthermore, the DIY CL community may utilize this as an at-home intervention to better understand the daily experience of using the system before testing it out. If this were adapted for use with more widely available at home VR systems, users would be able to experience the CL system without the hassle of gathering insulin pump and CGM materials and coding the interface before deciding to try a DIY system out.
The VR intervention experience may also be useful in teaching clinicians about what it is like to wear a CL system. Although not presented above, as surveys were not conducted, our research team utilized clinicians and trainees to beta-test the VR intervention for bugs, and each clinician noted afterward how different the experience was of “wearing” the CL system was than what they thought it would be, as well as the social aspects and annoyances that they did not expect. This may help clinicians determine which barriers might be more or less prominent for different people with diabetes, as well as expose them to some common ways to address these barriers. Furthermore, it may be helpful for clinicians to better understand some of the DIY CL specific difficulties which were touched upon in this experience, such as connection issues.
Limitations include that the barriers identified in this study come from clinical trials of CL systems, studies on component parts (ie, insulin pumps and CGM), and a large study where expectations for CL systems were assessed, but there are no full CL systems on market at this time. Thus, there are no current data on uptake or use of a fully automated CL system and barriers with an FDA approved on-market algorithm is not available. The only approximation would be clinic research done on the Medtronic 670G, a hybrid CL system, which has shown a decline in the use of auto-mode over time, with participants indicating hassles with calibrations and dislike of alerts. 12 This was also a pilot consisting of a small sample size resulting in low statistical power. A larger analysis should be conducted to determine whether the VR intervention experience causes any true effects. Participants rated the experience as feeling fairly realistic, and feedback was given on how to improve the experience to be even more realistic, the majority noted that the avatar’s inability to speak took away from the realism, which was a feature that we chose not to build given the small pilot nature and budget of the project. Even with these limitations, a significant decrease in negative affect was observed. This indicates less state of anger, contempt, disgust, nervousness, and distress after participating in the VR intervention experience. 34 Furthermore, there was a decreased trend in overweight preoccupation, which indicates that participants were less occupied with being overweight after the VR experience. 35
Conclusion
A VR intervention aimed at exposing participants to common barriers, concerns, and daily tasks around CL systems is acceptable among patients with T1D, does not produce VR-related motion sickness, and is quick to deliver. We have shown that the VR intervention experience is feasible and has the opportunity to expose many individuals to common barriers and hassles of using FDA approved and DIY CL systems. More is needed to determine the specific effects of this intervention experience as well as where this VR experience may be most appropriately utilized.
Footnotes
Acknowledgements
We are grateful for the ancillary support to complete this project from our research team including Sarah Hanes and Regan Barley.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a pilot award from the Stanford Diabetes Research Center from a grant sponsored by NIH (P30DK116074).
