Abstract
Background:
MiaoMiao (MM) is a Bluetooth transmitter, which when paired with a smart phone/device, converts the Abbott FreeStyle Libre flash glucose monitoring system into a Do-It-Yourself (DIY) continuous glucose monitor (CGM). Families are increasingly adopting DIY CGM solutions, but little is known about parent and child experiences with these add-on technologies. We aimed to explore experiences of families using MM-CGM including challenges faced and their advice to others who may choose to use the technology.
Methods:
Between May and July 2019, we conducted 12 semistructured interviews (in person or via video conference) with parents of children (aged ≤16 years) with type 1 diabetes using MM-CGM. Interviews were audio recorded; professionally transcribed and key themes were identified through thematic analysis.
Results:
Overall, parents used MM-CGM to proactively manage their child’s blood glucose. In all participants, this led to a perceived decrease in frequency of hypoglycemia. Participants reported that the visibility and easy access to blood glucose readings, glucose trends, and customized alarms on parent’s phones decreased their disease burden and improved their sleep quality. Common barriers to using MM-CGM included difficulty of the setting up process, connectivity issues, and lack of support from medical teams.
Conclusion:
This study highlights the potential feasibility of using a DIY CGM system like MM-CGM, which could be an empowering and cost-effective tool for enabling remote monitoring of blood glucose in real time.
Keywords
Introduction
Management of childhood type 1 diabetes mellitus (T1DM) incurs significant treatment burden for both patients and their caregivers. 1 While intensive management reduces long-term complication risk,2,3 in the diabetes control and complications trial (DCCT), it resulted in a threefold increase in severe hypoglycemia, with much of this occurring during sleep. 4 However, the incidence of severe hypoglycemia has decreased over the last two decades with implementation of the results of the DCCT by diabetes teams, insulin analog therapy, and the use of advanced technology. 5 Complications of severe hypoglycemia include seizures, coma, or rarely death. 6 Fear of severe hypoglycemia is common among parents of children with T1DM, with resultant suboptimal glycemic control.5-9
Glucose monitoring is a vital part of both hypoglycemia prevention and intensive diabetes management, and is often the responsibility of the caregiver for children and youth with T1DM, particularly overnight. 10 Each additional capillary glucose measurement up to six daily is associated with a clinically significant reduction of 5 mmol/mol in HbA1c.11,12 Continuous glucose monitoring (CGM) systems that measure interstitial glucose levels every five minutes may offer a range of advantages over traditional capillary glucose monitoring. These systems provide additional information on glucose trends and reduce the need for frequent finger pricks. In addition, with the ability to set glucose threshold alarms (both for hypo- and hyperglycemia), the requirement for overnight testing may also be reduced. 13 Frequent use of CGM translates to improved glycemic control including reduced hypoglycemia. 14 Flash glucose monitoring (FGM), also known as intermittently scanned CGM, measures interstitial glucose and is an alternative CGM device. FGM has the advantage of not requiring capillary glucose testing for calibration; but although it continuously measures interstitial glucose, these readings are only available when the user swipes their reader across the sensor. Therefore, the currently available models are unable to utilize glucose threshold alarms or remote monitoring. Despite these limitations, FGM is now used by 1.5 million people worldwide 15 with its relative popularity arguably attributable to its lower price (with FGM costing approximately $US1600/year vs CGM at $US3200-6400/year). Cost is an important barrier to access/uptake, and while FGM and CGM are funded in some countries, both remain unfunded in New Zealand.
A third-party device (MiaoMiao, MM) has recently entered the market. 16 In combination with FGM, it has the potential to offer many of the benefits of commercial CGM at a fraction of the price (MM can be purchased online for $US139 as a one-off cost). MiaoMiao, along with other similar devices, converts FGM into a Do-It-Yourself continuous glucose monitor (MM-CGM). MiaoMiao is placed over the standard FGM sensor, uses near-field communication to read raw data from the FGM sensor, and then transmits this via Bluetooth to a paired smart device, bypassing the official algorithm. The raw data are then processed by an algorithm in a nonofficial CGM app: either a proprietary third-party CGM app, Glimp 17 or Tomato, 18 or an open source CGM app developed by the #WeAreNotWaiting movement, Spike, or xDrip+.16,19 #WeAreNotWaiting is a patient-led diabetes technology innovation and advocacy movement that developed in response to the slow rate of development of patient-centered digital diabetes technologies. 20 As DIY projects, users are responsible for setting up their own system with limited online support provided by the wider #WeAreNotWaiting community. Whereas FGM is factory calibrated and unable to receive finger prick calibrations, finger prick calibration is a requirement of these systems, which is anecdotally a positive feature.
Patients and their families are adopting MM-CGM as an affordable CGM solution. However, this product/system has no regulatory approval or safety data and the literature on the patient/family experience in using a MM-CGM system is lacking. To date, there has been no study investigating the experience of those families who were early adopters of using this technology, usually without any recommendation from their medical team. Therefore, we aimed to explore experiences of families using MM-CGM, including reasons for choosing MM-CGM over other glucose monitoring approaches, the broad impacts this technology has on children and adolescents with T1DM and their families, challenges while using this technology, and families’ recommendations to others who may choose to use DIY CGM solutions.
Methods
Recruitment
The study was conducted from May to July 2019. This study was approved by the University of Otago Human Ethics Committee (reference number: H19/002). The inclusion criteria included having a child ≤16 years diagnosed with T1DM, with current or recent use of MiaoMaio (MiaoMiao Smart Reader, BL512, Shanghai High Brilliant Health Technology Co. Ltd., China), English fluency, and willingness to participate with no other restriction. Participants were recruited using two approaches, initially from Southern District Health Board diabetes clinics (n = 4) and then advertisements posted to New Zealand T1DM Facebook groups (n = 8). This enabled recruitment of those who were broadly representative of parents who have chosen to use MM-CGM in New Zealand.
Study Design
Twelve semistructured interviews were conducted with the parents. The final sample size was determined when data saturation was reached, in that, for interviews 10 to 12, no new information or perspectives regarding the main themes became apparent. Before conducting the study, an interview guide was developed based on a literature review and consensus between investigators with expertise in child health, pediatric endocrinology, and qualitative research.
Data Collection and Analysis
All participants completed a demographic questionnaire to collect basic demographic criteria including age, gender, ethnicity, parent’s and child’s age, and duration of MM-CGM use. Other clinical information such as recent HbA1c, duration of diabetes, and method of insulin administration was collected from the primary diabetes care physician after getting parental consent. The Diabetes Treatment Satisfaction Questionnaires (DTSQs) 21 were also used for better understanding and quantitative evaluation of the level of satisfaction these participants experience. The DTSQs were analyzed according to the scoring analysis instructions from the health psychology research unit. 21 One of the research team investigators (SB or ME) carried out the interviews, with (BW, SB, and ME) all involved in interview 1.
The interviews were conducted face to face (n = 3) or via Zoom (Zoom Video communications, San Jose, California, United States; n = 9) in accordance with participants’ preference. Interviews lasted 50 to 75 minutes and were digitally recorded. Recordings were transcribed verbatim, then checked for their accuracy and de-identified.
The transcripts were coded thematically 22 using a framework organized by the study objectives (ie, exploring parents’ perceptions of wanting a CGM system, learning about MM-CGM, setting up MM-CGM, and impacts and experiences after using the system) in NVivo software (NVivo 12, QRS International Pty Ltd., VIC, Australia). All transcripts were independently coded according to the framework by two investigators (ME and SB). Following this, members of the research team (ME, SB, HC, and BW) discussed codes and themes, achieving consensus that all key experiences had been captured. The frequency for each theme and subtheme was quantified to aid determination of thematic saturation, as well as to demonstrate the common experiences among families.
Results
The demographic details of 12 participating families are shown in Table 1. Details of the MM-CGM system, the applications used by participants, and the number of people using follower functions are shown in Table 1 and Figure 1.
Characteristics of the Participants.

Abbreviations: DTSQs, Diabetes Treatment Satisfaction Questionnaires; MDI, multiple daily injections; MM, MiaoMiao; MM-CGM, MiaoMiao continuous glucose monitor.
NZDep13 index is a deprivation index based on household address with one being the least deprived and ten being the most deprived.
No artificial pancreas/closed loop users.
Applications used for the “follower” function; this included Nightscout, Nightguard, xDrip+, and Tomato applications.
HbA1c is the most recent haemoglobin A1c obtained from the participants’ medical team after getting informed consent.
DTSQs treatment satisfaction maximum and minimum scores are 60 and 0, respectively; status version for parents was used.
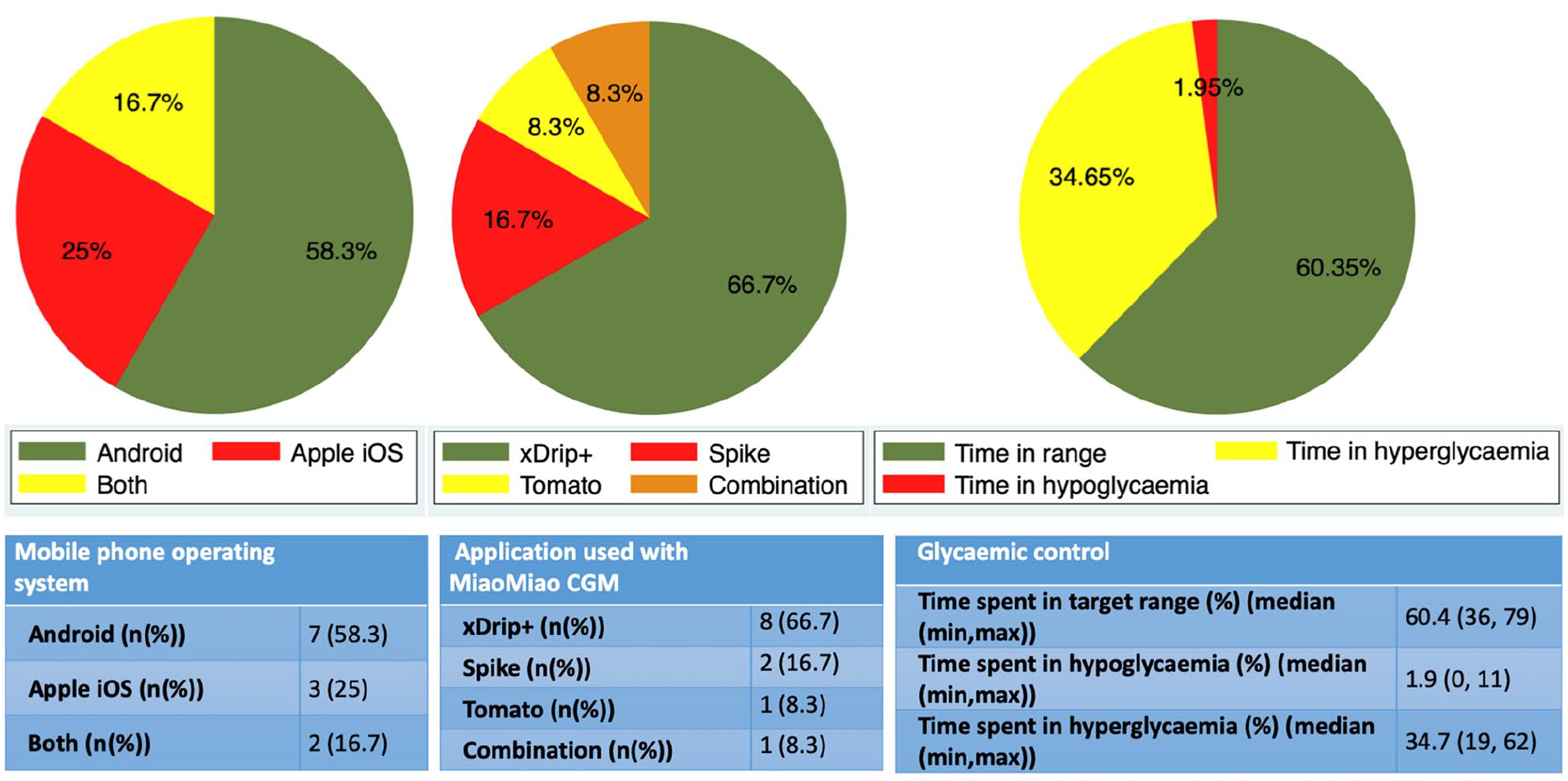
Glycemic control, used applications, and type of mobile phone used by participants.
Overall, participants described their experience of using MM-CGM as positive, expressing general satisfaction with the device. This was confirmed by the results of DTSQs (Table 1). All participants noted positive aspects of remote monitoring and safety alarms with predictive trend arrows and graphs and associated increased awareness of hypo/hyperglycemia. Results are presented in relation to the five key study objectives (detailed in Tables 2 and 3).
Reasons for Choosing MiaoMiao Continuous Glucose Monitor and the Setting Up Process: Representative Quotes.

Abbreviations: DIY CGM, Do-It-Yourself continuous glucose monitor; MM, MiaoMiao; MM-CGM, MiaoMiao continuous glucose monitor.
Number of participants describing each theme/subtheme.
Glycemic Control, Positive Impacts, and Recommendations: Representative Quotes.

Abbreviations: MM, MiaoMiao; MM-CGM, MiaoMiao continuous glucose monitor.
Number of participants describing each theme/subtheme.
Reasons for Choosing MM-CGM
Families relied on Facebook groups (12/12) and wider internet searches to look for full CGM alternatives to FGM. Two families had tried commercial CGM, but for most families, the price of commercial CGM was reported as prohibitive (9/12). During these internet searches, families reported that MM was the most frequently recommended device for building DIY CGM. Beyond price, key reasons reported for choosing MM-CGM included its perceived reasonable size (7/12) as compared to other DIY CGM and Dexcom G4 CGM, with some families (4/12) reporting the previous negative experiences with other CGM systems, relating to bulky size, poor connectivity (quote 5), or painful insertion (quote 6).
MiaoMiao Continuous Glucose Monitor Initiation, Setting Up Process, and Other Negative Issues
The majority of participants (10/12) found the initial setup process challenging. The most difficult challenges were related to setting up Nightscout and sharing CGM data between iOS and android devices. iOS users (n = 5) reported difficulties with the open source CGM app, Spike. While the majority of parents (10/12) were able to set up the system using online resources and support via Facebook groups, two parents sought technical assistance from someone other than their own partner (quotes 13 and 14). Participants expressed a desire for a “plug and play” alternative to the multistep process that was required (quote 15).
Some families (n = 3) reported that the transmission of the data from the primary collecting device to follower (caregivers) devices was more stable when connected on Wi-Fi as compared to connection on mobile data. The majority (n = 9) noticed temporary signal loss when the primary collecting device was not in the Bluetooth range of the MM (quotes 23, 24, and 25). Many (7/12) noted a lag period between MM CGM readings and actual capillary blood glucose levels (quotes 20, 21, and 22). In addition, five participants (5/12) reported that the first MM transmitter they used was faulty or cracked and they had to replace it. None of the families currently used the adhesive stickers that were enclosed in the MM package directly applied to the skin as they were noted to cause skin reactions. They reported only using these stickers between MM and FGM sensors. Most of them (11/12) used additional adhesive sport tapes.
Glycemic Control and Growing Confidence With MM-CGM
All families depended on MM-CGM to inform proactive use of corrective insulin dosing or carbohydrate consumption to minimize out-of-target glucose levels. They found having these data available on their phones in real time to be more useful and informative than glucose readings obtained from finger pricking or FGM scanning. They reported more stable blood glucose levels which were reflected by increased self-reported time spent in range (Figure 1) and decreased self-reported frequency of hypoglycemia (12/12). The system was also reported as facilitating learning about diabetes, with families reporting growing capability in making treatment decisions in response to glycemic fluctuations and trend arrow readings. Most families used “low” alerts/alarms (set between 3.8 and 6.0 mmol/L) and “high” alerts (set between 8 and 24.0 mmol/L). In all cases, these low and high alerts/alarms were reported as being set by families without input from their diabetes teams. Furthermore, none of our participants reported receiving any guidance from their medical teams regarding safety alerts nor the frequency of calibration in order to get accurate results from the system. Therefore, while all participants calibrated, calibration frequency ranged from five times a day as mentioned by one family to only two times per week.
Nearly all parents (11/12) primarily used MM-CGM adjunctively with confirmatory capillary finger pricks when managing hypo/hyperglycemia. One teenage girl was reported to only use MM-CGM data (no adjunctive capillary input) to make treatment decisions at school (quote 31). Nine families described MM-CGM to be accurate and reliable, some reported that the device was more accurate than the FGM (n = 5). However, one family reported trusting FGM more.
Positive Impacts, Improved Quality of Life, and Better Sleep Quality
All of our participants reported a better quality of life after using the device with reduced parental stress and anxiety. Some allowed their children to have more social activities, such as being out with their friends and having a sleepover, as remote monitoring alleviated their previous concerns relating to hypoglycemia. Additionally, MM-CGM was noted to contribute to improved parental sleep quality/quantity (n = 10). This was due to decreased frequency and necessity of nocturnal blood glucose monitoring. Nearly all the parents noticed that the burden of multiple waking every night to check glucose levels, which was reported as ranging from two to five times per night, was dramatically reduced using MM-CGM. However, MM-CGM did not eliminate all T1DM-related sleep disruption. One family had decided to use the system only during the night to utilize out-of-range alerts. Another family reported that their adolescent decided to switch off the alarms at school because the child did not want to draw attention and the parents did not follow the blood glucose levels during school time.
Continued Use of MM-CGM and Recommendations for Others
All of our participants (12/12) planned to continue to use MM-CGM and recommended it to other families because of its affordability and functionality as a reliable remote CGM. A perceived benefit regarding the rechargeable long-life battery of the device was reported by the majority of participating families (11/12), which can last two to four weeks. The only ongoing cost was the cost of the FGM sensors. Some (3/12) suggested others would be able to set up the system provided they followed setup instructions in a logical manner. The remainder suggested that the difficulty of the setup process was a barrier to potential users. Parents hoped that commercial CGM devices would get funded by the government. Some (3/12) would prefer to be able to purchase the MM transmitter from a New Zealand-based company, so they would get after-sale customer service. Also, some specifically wished for more involvement and guidance from their medical teams (n = 5), both in setup and reviewing data and safety alerts; none of our participants reported any guidance from their medical teams before purchase. Two families reported that their medical teams were not familiar with MM-CGM.
Discussion
To our knowledge, this is the first study to explore any type of DIY CGM. The main findings highlight the generally positive self-reported experiences of parents of children with T1DM using MM-CGM, including reductions in fear of hypoglycemia, improvements in parental and child sleep quality, glycemic understanding and control, and decreased overall disease burden. Negative aspects were also reported, including the burden of setup, signal loss, lack of after-purchase customer service, and their wish to have more support and involvement from diabetes health professionals. Clearly, while MM-CGM holds promise, there remains a need for improvement and further research.
Importantly, a reduction in overall frequency of hypoglycemia and fear of hypoglycemia was reported by all families. These benefits regarding hypoglycemia translated for many families into improved peace of mind and reduced anxiety, especially at night. While this needs confirmation from well-designed trials, it is notable that the i-HART CGM trial reported potential benefits of real-time CGM over FGM in reducing the time spent in hypoglycemia and in improving fear of hypoglycemia. 23 Several parents reported better sleep quality/quantity while using MM-CGM. This is supported by the general CGM literature.24,25 Similarly, our results suggest that improved parental sleep may also improve sleep quality for children.
In addition to benefits around hypoglycemia, as with the previous CGM studies,26-28 these participants reported better glycemic control. The benefits of remote monitoring of glucose levels were frequently mentioned and contributed to a parental feeling of improved safety. The literature on CGM is mixed, with recent data supporting improvements in parental psychological measures including fear of hypoglycemia, 13 although negative aspects have also been reported, including heightened worry and anxiety as a result of increased awareness of their child’s glucose measurements, 25 which was reported by one family in our study. The ability to take timely action on receiving a preset alarm seemed to provide reassurance to our participants.
Being a DIY diabetes technology, there were barriers facing users of MM-CGM, especially during the initial setup which was described as a multistep, complex process. Although the MM hardware came fully built, setup of software on collecting and follower devices was undertaken by users without input from their medical team or the manufacturer. Instead, online resources and peer support from others within the DIY diabetes community facilitated the setting up process and ongoing use. Furthermore, the participants reported neither receiving support from their medical team regarding setting alarms for glycemic excursions nor receiving education on best calibration practices. Both alarm settings and calibration practices varied significantly among families. Advice from clinicians regarding alarms and calibration practices may assist future users to minimize alarm fatigue 29 and maximize the accuracy of their CGM. 30
Although the variation in calibration practices may appear concerning, nearly all of our participants (apart from one teenager) reported using MM-CGM as an adjunctive device for out-of-range readings and regularly performed confirmatory capillary glucose tests. Similarly, the connectivity issues which participants experienced were partially mitigated by the open-source CGM apps being designed to cope with failure, with their ability to set alarms for signal loss. Parents trusted the device and the open-source software that they used alongside the device. This brings into light the importance of examining healthcare professionals’ perceptions of this new open resource “nonapproved” CGM technology and their views for best practice. 31
Generalizing the results of our study to other families considering DIY CGM remains uncertain. These participants were highly motivated and most of them were highly educated; they were also from a less deprived socioeconomic position and all were of New Zealand European ethnicity. In addition, as with other CGM data, overall parental technology satisfaction (as measured by DTSQs) was also seen with the use of MM-CGM,32-35 although DTSQs prior to use were not measured for comparison. This could be another limitation point as well as the self-reported improvement in the glycemic control. The findings of this study represent parental self-reported experience after using MM-CGM and clearly more objective trial data are required. Strengths of this study include the in-depth examination of the parental experience of using MM-CGM. Though the interview guide was predefined, the interviewers could explore new insights as they arose, aiding our understanding of parental experiences. Although the sample size was small, no new significant insights emerged during the final three interviews, suggesting that the key experiences of caregivers using the system were identified.
Conclusion
In conclusion, this study confirms that families impacted by T1DM are using DIY CGM systems, and in this sample reported generally positive experiences, and recommended these systems to other families. The use of DIY CGM is primarily driven by the affordability of the system compared to commercial CGM. Important benefits appear to cluster around perceptions of improved safety, quality of life, reduced parental anxiety, improved night time diabetes care, and importantly decreased frequency of severe hypoglycemia. Negatives with this DIY CGM solution do exist and awareness of these are important for future users and diabetes health professionals so all are fully informed before beginning MM-CGM use. These findings highlight the potential feasibility of using a DIY CGM system; but clearly before healthcare professionals can readily recommend these systems, further research building on the findings of this study is needed to confirm the efficacy, safety, and reliability of DIY CGM systems.
Footnotes
Acknowledgements
The authors wish to thank the participants for their generous involvement in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Department of Women’s and Children’s Health, Dunedin School of Medicine, University of Otago, New Zealand.
