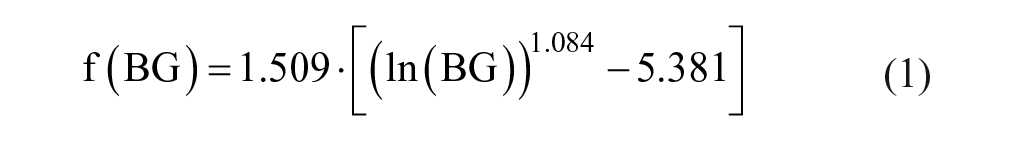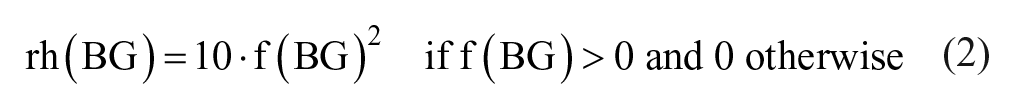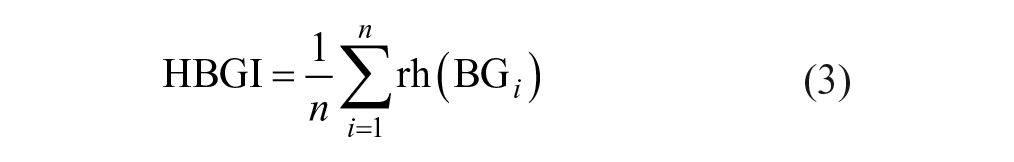Abstract
Background:
iGlarLixi is an injectable combination of long acting insulin glargine (iGlar) and glucagon-like peptide 1 receptor agonist lixisenatide in a fixed ratio, which was proven safe and effective for the treatment of type 2 diabetes. Lixisenatide and iGlar act differently on fasting and postprandial plasma glucose (fasting plasma glucose [FPG] and postprandial glucose [PPG]). Here, we deconstruct quantitatively their respective FPG and PPG effects.
Method:
This post hoc study analyzes data from the Lixilan-O trial, where 1170 subjects with type 2 diabetes were randomly assigned to 30 weeks of once daily injections of lixisenatide, iGlar, and iGlarLixi (1:2:2). The FPG and PPG components of glucose control were assessed in terms of mean glucose (fasting mean plasma glucose [FMPG] and prandial mean plasma glucose [PMPG], respectively). The MPGP was computed across all meals as a delta between post- and premeal glucose; glucose variability was measured by the high blood glucose index (HBGI) (fasting HBGI and prandial HBGI [PHBGI], respectively), and glycemic exposure measured by area under the curve (AUC) computed overall. All metrics were derived from seven-point self-monitoring glucose profiles.
Results:
Insulin glargine lowered significantly FMPG by 15.3 mg/dL (P < .01) without any significant change in PMPG. Lixisenatide, when added to iGlar, reduced PMPG by 9.7 mg/dL (P < .01), AUC by 96.3 mg∙h/dL (P < .01), and PHBGI by 2.4 (P < .01), primarily due to attenuation of PPG and without significant change in mean FPG.
Conclusion:
Insulin glargine and lixisenatide act selectively on FPG and PPG. Their combination iGlarLixi offers more effective glucose control than its components due to the cumulative effect on FPG and PPG, which is evidenced by reduced average glycemia, glycemic exposure, and glucose variability.
Keywords
Introduction
Glycemic control can be deconstructed into a postprandial glucose (PPG) component and a fasting plasma glucose (FPG) component. Both contribute to the overall glycemic control and were shown to be predictive of glycated hemoglobin (HbA1c). 1 Glycemic lowering agents can act on the fasting component, on the postprandial component, or on both. Each of these effects has their own benefits and drawbacks, and an optimal balance between both strategies would be best to maximize benefits and limit drawbacks, eg, reduce HbA1c without increasing the risk for hypoglycemia.
Insulin glargine (iGlar), being a long acting insulin with a relatively flat profile, acts at a “basal level,” reducing glucose levels consistently throughout the day. As such, both fasting blood glucose levels and postprandial levels are reduced when iGlar is added. 2 This type of strategy has been referred to as a fasting reduction strategy.
Glucagon-like peptide 1 (GLP-1) is a hormone secreted by the L-cells of the gut. Its role in the glucose-insulin network is thoroughly explained in the following reference. 3 Lixisenatide is a glucagon-like peptide 1 receptor agonist (GLP1-RA). These agents have been proven to be safe and effective in lowering blood glucose levels for the treatment of type 2 diabetes. Acting via stimulation of insulin secretion in a glucose dependent fashion, the inhibition of glucagon secretion and gastric emptying, and induction of satiety, short acting GLP-1 RAs, such as lixisenatide were shown particularly effective in dampening PPG excursions 4 without inducing hypoglycemia. 5 In fact, GLP-1 RAs enhance the postprandial insulin secretion, further raising plasma insulin levels which in turn intensify glucose uptake from the liver and other insulin-dependent tissues. 6 In addition, the reduced glucagon secretion further limits the endogenous glucose production and contributes to reducing PPG peaks. Finally, the gastric emptying is delayed allowing more time for the body to reduce PPG excursions, while increased satiety also reduces food intakes, hence promoting a reduction of postmeal glucose excursions. This type of strategy will be referred to as a postprandial reduction strategy.
Using a combination of iGlar and lixisenatide was shown to reduce HbA1c and glucose variability.7,8 The injectable iGlarLixi is a fixed ratio combination of the two components shown to have similar benefits in clinical trials. 9 The Lixilan-O 9 clinical trial compared iGlarLixi, lixisenatide, and iGlar and observed a significant lowering of HbA1c in the iGlarLixi group compared to lixisenatide alone and iGlar alone. More specifically, the Lixilan-O study demonstrated a larger HbA1c decrease from baseline achieved with iGlarLixi compared to iGlar and lixisenatide (−1.6%, −1.3%, and −0.9%, respectively). In addition, FPG levels decreased by −63, −59, and −27 mg/dL and two-hour postprandial values by −103, −59, and −83 mg/dL from baseline for, respectively, iGlarLixi, iGlar, and lixisenatide. We can speculate that this effect is due to the differential action of iGlar and lixisenatide on the FPG and PPG components of glycemic control. Thus, using data from the previously published Lixilan-O trial, 9 this analysis assessed the differential contribution of lixisenatide and iGlar to the effect of their combination iGlarLixi on the fasting and postprandial components of glucose control.
Methods
Study Design
This post hoc analysis compares lixisenatide, iGlar, and iGlarLixi groups through measurements of glycemic control using data from the LixiLan-O 9 trial (NCTO2058147). This randomized, parallel-group, multinational, multicenter trial enrolled 1170 subjects with type 2 diabetes uncontrolled with metformin. The participants were randomized 1:2:2 to once daily lixisenatide (234 subjects), iGlar (467 subjects), or iGlarLixi (469 subjects). Lixisenatide was titrated from 10 µg to a fixed dose of 20 µg, while iGlar and iGlarLixi were titrated to achieve a FPG target of 80 to 100 mg/dL, up to a dose cap of 60 U iGlar. Two pens administered iGlarLixi in ratios of 2:1 or 3:1 (U of iGlar/µg of lixisenatide) never to exceed 20 µg of lixisenatide. The mixture was titrated once a week starting with 10 U of insulin in a 2:1 ratio (2 U of iGlar and 1 µg of lixisenatide) to achieve fasting levels of 80 to 100 mg/dL. When participants required more than 40 U, they were switched to a 3:1 ratio mixture. 9 The injection of iGlarLixi was performed before breakfast. Lixisenatide was administered before breakfast or dinner at the discretion of the patient and remained the same throughout the study. The injection of iGlar could be done anytime but remained at about the same time throughout the study.
Assessments
As reported in the study of Rosenstock et al, 9 the Lixilan-O study continued for 30 weeks and participants performed seven-point (prebreakfast, two hours postbreakfast, prelunch, two hours postlunch, predinner, two hours postdinner, and bedtime) self-monitoring plasma glucose (SMPG) profiles, once at baseline and once at termination. The seven-point SMPG profiles (shown in Figure 1) were used to compute mean plasma glucose (MPG), area under the curve (AUC), and the high blood glucose index (HBGI). Mean plasma glucose and HBGI were computed both in the fasting and postprandial state, while AUC was computed overall. The fasting state metrics (fasting mean plasma glucose [FMPG] and fasting high blood glucose index [FHBGI]) were computed from the prebreakfast SMBG point. The postprandial state metrics (prandial mean plasma glucose [PMPG] and prandial high blood glucose index [PHBGI]) were averaged across all postmeal SMPG points (breakfast, lunch, and dinner). While PMPG was computed from delta pre-postmeal SMPG points, PHBGI was computed directly from the postmeal points. Because HBGI is a marker of glycemic variability in the high normoglycemic and hyperglycemic ranges, its interpretation as a differential metric would be challenging.10,11 Mean plasma glucose was used as a marker of average glycemia, AUC was used to quantify glycemic exposure, and the HBGI was used as a marker of glucose variability in the hyperglycemic range. 11 The HBGI was computed using its established formula based on a logarithmic transformation of the glucose measurement scale, which emphasized the hyperglycemic component of glucose variability above an optimal target 11 :
With blood glucose (BG) in mg/dL,
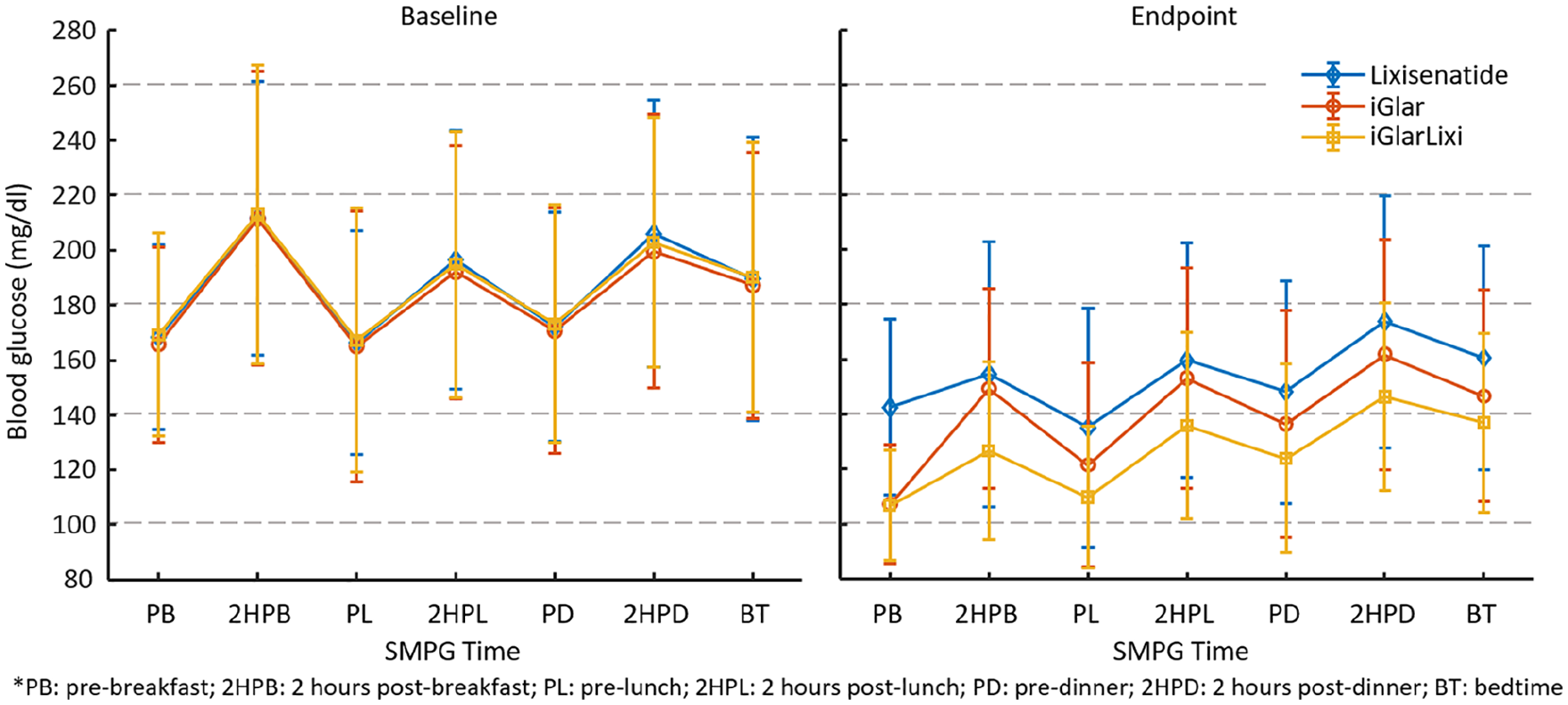
Evolution of the seven-point SMPG profiles [mean and standard deviation] under different interventions. This is a reproduction of a figure already published. 9
Mean plasma glucose, AUC, and HBGI were computed from postprandial and fasting SMPG measurements in the seven-point profiles. The differences of these metrics between the study groups were used to compare and visualize the relative effects of iGlar, lixisenatide, and iGlarLixi on FPG and PPG.
Statistical Analysis
Patients were included if they had complete seven-point SMPG profiles at baseline and at week 30. A t-test was performed on MPG, AUC, and HBGI reductions, and between groups utilizing both fasting and postprandial measurements.
Results
Mean Plasma Glucose
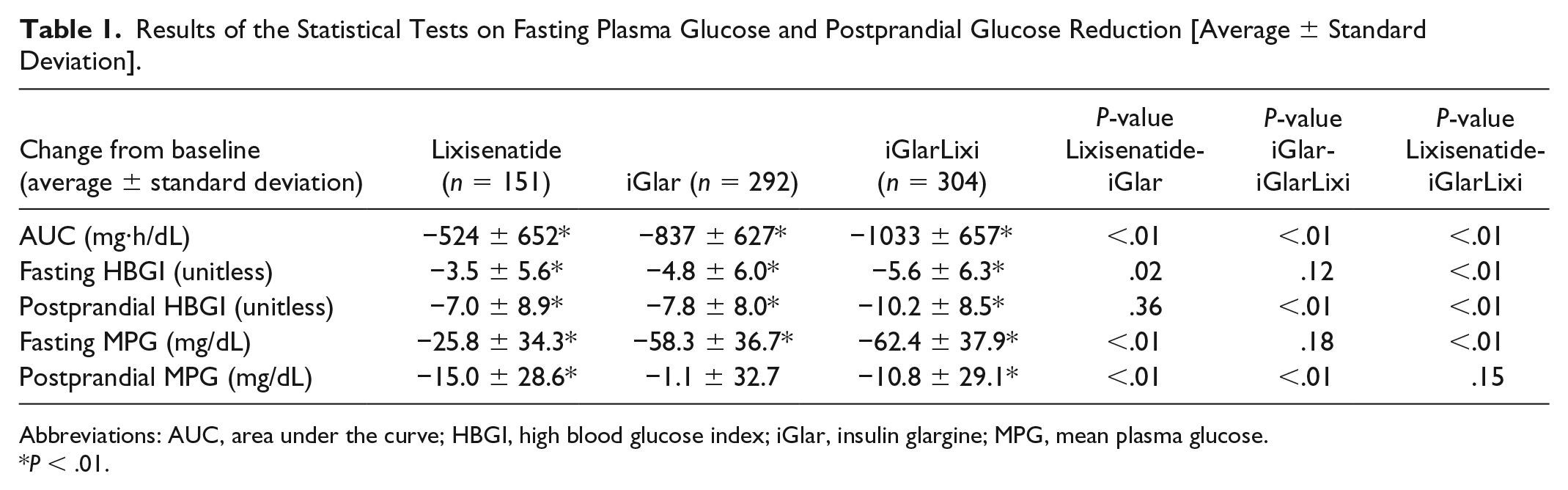
Reduction of FMPG and PMPG achieved by iGlar, lixisenatide, and iGlarLixi are reported in Table 1. Insulin glargine achieved a significant reduction in FMPG but did not reduce significantly PMPG. When added to iGlar, lixisenatide enhanced significantly the reduction of PMPG and resulted in a nonsignificant reduction of FMPG. These relative effects are presented in Figure 2.
Results of the Statistical Tests on Fasting Plasma Glucose and Postprandial Glucose Reduction [Average ± Standard Deviation].
Abbreviations: AUC, area under the curve; HBGI, high blood glucose index; iGlar, insulin glargine; MPG, mean plasma glucose.
P < .01.
Deconstruction of mean plasma glucose into fasting and postprandial components. Postprandial glucose was computed from all three meals and as a difference pre-postmeal glucose.
Area Under the Curve
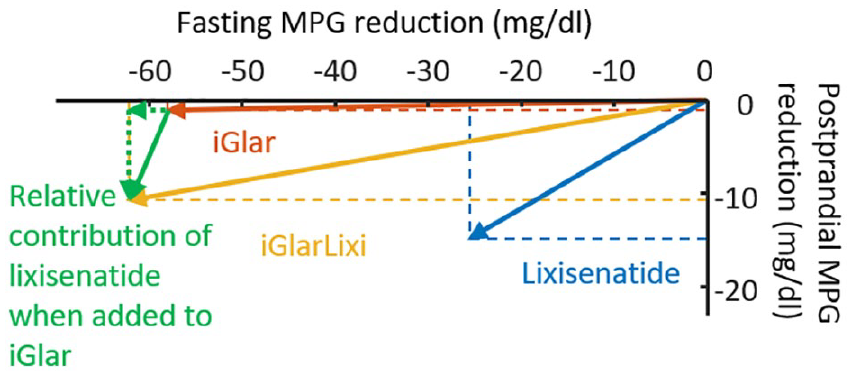
Changes in AUC induced by iGlar, lixisenatide, and iGlarLixi are given in Table 1 and in Figure 3. Significant reduction of AUC was found both with iGlar and when lixisenatide was added to iGlar.
Reduction from baseline in area under the curve [mean and standard deviation].
Glucose Variability (HBGI)
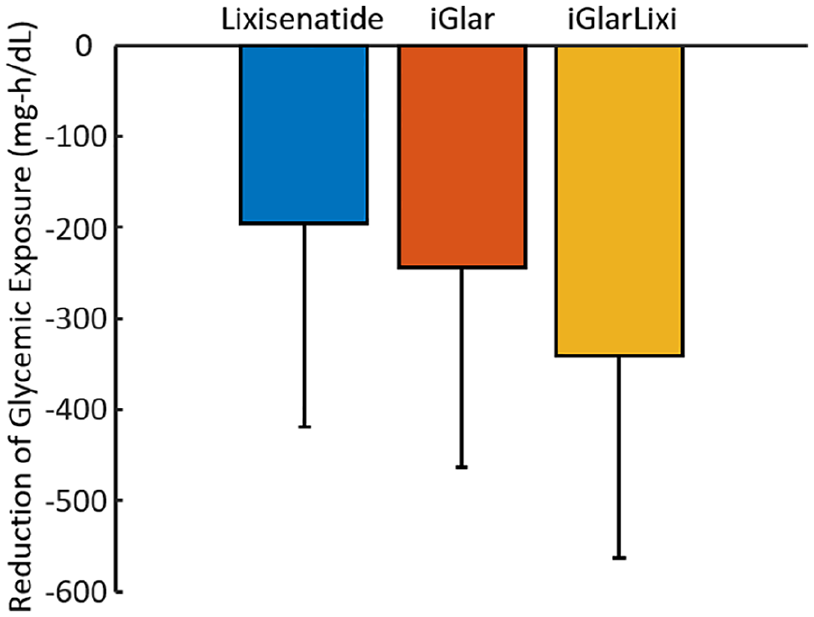
Reductions of FHBGI and PHBGI achieved by iGlar, lixisenatide, and iGlarLixi are reported in Table 1. Insulin glargine resulted in a significant reduction in both FHBGI and PHBGI. When added to iGlar, lixisenatide achieved a significant reduction of the postprandial component and a nonsignificant reduction of the fasting component. On the other hand, when added to lixisenatide, iGlar achieves a significant reduction of both the postprandial and the fasting components. The decomposed representation for HBGI is shown in Figure 4.
Deconstruction of glucose variability into a fasting and postprandial component. Postprandial high blood glucose index was computed from all three meals.
Discussion
This post hoc study used data from the published Lixilan-O Phase 3 clinical trial to assess the effects of lixisenatide and iGlar administered separately or in a fixed-ratio combination. These effects were deconstructed into fasting and postprandial components, which corresponds to the mechanisms of action of these agents. Because glargine has long action and a fairly flat profile, 13 it is able to decrease fasting glucose levels but is less effective in reducing rapid postprandial excursions. This kinetics explains the finding that iGlar achieved larger FPG than PPG reduction. This effect was confirmed by metrics of glycemic exposure (eg, AUC) and hyperglycemic glucose variability (HBGI). The larger impact of iGlar on FPG vs PPG (see the rectangular shape of iGlar action in Figure 2) is consistent with the inability of most patients with type 2 diabetes using long acting insulin alone to reach HbA1c targets while avoiding hypoglycemia; when optimal fasting values are reached, postprandial excursions remain prominent.
On the other hand, lixisenatide has a mechanism of action particularly efficient after meals, and as such, decreases postprandial excursions significantly and is less effective in reducing fasting glucose levels. Its actions explain the larger reduction of PPG compared to FPG. The overall effect of lixisenatide was confirmed by metrics of glycemic exposure (eg, AUC) and hyperglycemic glucose variability (eg, HBGI). Since under most conditions, postprandial glycemic levels are higher than those in fasting state, a combination of lixisenatide and glargine would seem more appropriate than glargine alone to simultaneously reach optimal fasting and postprandial glycemic levels (see the rectangular shape of lixisenatide action in Figure 2). Indeed, the combination of lixisenatide and insulin glargine (iGlarLixi) achieved reduction in both average PPG and FPG (see differential effect in Figure 2), without further significant reduction of the FPG component.
These findings are consistent with the previously published results. In terms of HbA1c reduction, glargine, iGlar, and iGlarLixi achieved changes of −0.9% ± 0.005%, −1.3% ± 0.004% and −1.6% ± 0.004%, respectively. 9 Glycemic variability, as measured by the mean absolute glucose excursion which emphasizes postprandial glucose values, was reduced by 7 ± 29, 2 ± 36, and 7 ± 34 mg/dL for lixisenatide, iGlar, and iGlarLixi, respectively. 7
As mentioned in the original study manuscript, 9 the injected amount of insulin was similar between groups using insulin in the final study week (40 ± 15 U for iGlar and 40 ± 15 U for iGlarLixi). From Figures 2 and 4, it can be noted that the effects of lixisenatide and iGlar are not perfectly additive (the green vector has a different length and may have a different direction than the blue vector). This is likely due to interactions between the components of iGlarLixi or/and nonlinearities in the metabolic dynamics.
Limitations of this study include the following: The performed analyses were not prespecified. Moreover, data were obtained from SMPG rather than continuous glucose monitoring. Thus, glucose variability and glycemic exposure were assessed with reduced resolution of the measurement methods.
Conclusion
Insulin glargine and lixisenatide act selectively on FPG and PPG. With this combination, iGlarLixi offers more effective glucose control than its components due to the cumulative effect of both iGlar and lixisenatide, demonstrated by the reduction in both glucose variability and glycemic exposure.
Footnotes
Acknowledgements
The present study data were made available by Sanofi.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BPK reports grants and personal fees from Sanofi, during the conduct of the study; he further reports grants from National Institutes of Health, personal fees and nonfinancial support from Dexcom, personal fees and nonfinancial support from Tandem Diabetes Care, outside the submitted work; and in addition, he has patents with royalties paid to Dexcom and Sanofi, managed by the University of Virginia. ER has received honoraria as consultant/advisor from Abbott, Air Liquide SI, Becton-Dickinson, Cellnovo, Dexcom Inc., Eli-Lilly, Insulet Inc., Johnson & Johnson (Animas, LifeScan), Medtronic, Novo-Nordisk, and Roche and Sanofi-Aventis. GEU is partly supported by research grants from the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR002378 from the Clinical and Translational Science Award program and a National Institutes of Health (NIH) grant U30, P30DK11102, and has received research grant support to Emory University for investigator-initiated studies from Sanofi, Novo Nordisk, and Dexcom. ER has received research support from Abbott, Dexcom Inc., Insulet Inc., Roche and Tandem.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.