Abstract

Pregnancies complicated by type 1 diabetes (T1D) are associated with reduced quality of life and increased emotional stress. 1 As women aim to maintain the recommended tight glycemic targets of pregnancy, 2 they often report changes in physical responses to hypoglycemia 3 and an increased fear of hypoglycemia. 4 Continuous glucose monitoring (CGM) has been shown to reduce fear of hypoglycemia in nonpregnant adults. 5 CGM can now also be used with remote monitoring by others. We examined whether remote monitoring of CGM trends and alerts by family and friends (CGM Share) could reduce fear of hypoglycemia compared to CGM use alone (CGM Alone) in pregnant women with T1D.
In this single-center, open-label, investigator-initiated pilot study, we recruited women with T1D planning to conceive and in the first trimester of pregnancy. Women in the first trimester were assigned to CGM Alone or CGM Share (Dexcom G4/G5 Platinum® CGM system with or without Share™ based on group assignment) based on their personal possession of an iPhone, iPad, or iPod. Women were provided the CGM system and glucose sensors. Women in the CGM Share group were assigned a main follower who was instructed to download the Share app and set up high and low glucose alert thresholds. Followers were administered questionnaires on a monthly basis inquiring about frequency of hypo- and hyperglycemia alerts, as well as glycemic interventions they performed (frequency and type). Women in the study and their followers both signed informed consent. This protocol was approved by the Western Institutional Review Board (clinicaltrials.gov registration number: NCT02556554). Herein, we report results from the Hypoglycemia Fear Survey (HFS) 6 administered once per trimester and four to six weeks after delivery. Longitudinal mixed effects models were used for analyses of the change in HFS scores over time.
The CGM Alone group had 13 women and CGM Share had 15. In pregnancy, there was no significant difference in mode of insulin delivery (continuous subcutaneous insulin infusion therapy: 69% CGM Alone vs 100% CGM Share). In mixed effects models with an interaction between group and trimester, there were no significant differences in the change in HFS scores. In mixed effects models without interaction, the mean hypoglycemia fear worry and total scores during gestation and postpartum were significantly lower in the CGM Share users ( p = .03 and p = .04, respectively, Table 1).
Hypoglycemia Fear Survey Results in Pregnancy and Postpartum. a
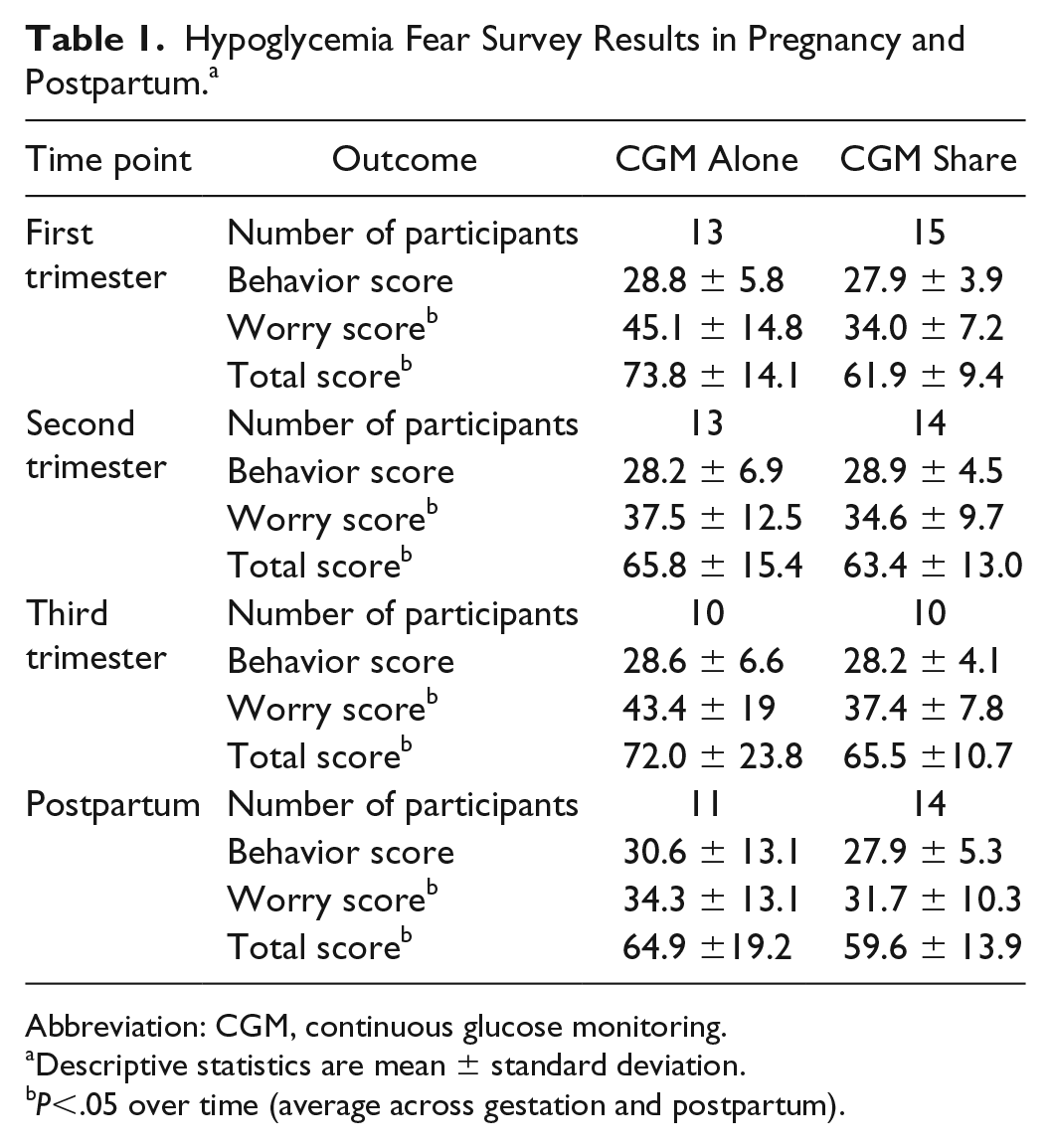
Abbreviation: CGM, continuous glucose monitoring.
Descriptive statistics are mean ± standard deviation.
P<.05 over time (average across gestation and postpartum).
We found that while using CGM with remote monitoring, pregnant women reported a lower fear of hypoglycemia compared to women using CGM Alone. These results should be interpreted with caution, as this clinical trial was not randomized. As women with pregnancies complicated by preexisting diabetes report increased distress, worry, and burden of self-care, 1 interventions that could potentially alleviate some of these burdens should be studied. Expanding a pregnant woman’s support system to include followers of their CGM data could be one way to reduce anxiety related to hypoglycemia as women strive to maintain optimal glycemic control.
Footnotes
Acknowledgements
The authors thank the participants of this study. We thank Mary Voelmle and Satish Garg, MD, for assistance with study procedures and study design, respectively. We thank Tim Vigers for his contribution to the computer programming for CGM data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SP reports research funding from DexCom, Inc., Eli Lilly, JDRF, Leona & Helsmley Charitable Trust, NIDDK, and Sanofi US and consulting for the JAEB center. RG, LP, PJ, JD, and JSB reported no potential conflicts of interest relevant to this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was an investigator-initiated study in part supported by Dexcom, Inc. through the Board of Regents at the University of Colorado Denver. This study was supported by NIH/NCRR Colorado CTSI Grant no. UL1 RR025780. Its contents are the authors’ sole responsibility and do not necessarily represent Dexcom, Inc. or official NIH views.
