Abstract
Background:
An anti-inflammatory drug-loaded composite coating (dexamethasone-loaded poly (lactic-co-glycolic acid) [PLGA] microspheres/polyvinyl alcohol [PVA] hydrogel) was previously developed to counter the foreign body reaction to a fully implantable continuous glucose monitoring biosensor. The long-term sensor functionality was ensured in the presence of the drug-loaded composite coating thus facilitating better diabetes control and management. In order to advance such a drug-device combination product toward clinical testing, addressing sterilization remains a key step due to the heterogeneity of the product components. The main objective of this research was to investigate the effect of two terminal sterilization techniques: gamma radiation and ethylene oxide (EO) on the stability of the anti-inflammatory coatings as well as retention of the glucose sensing ability of the implantable sensor.
Method:
The composite coatings, their individual components, and the glucose-sensing elements of the biosensor were subjected to low-temperature gamma radiation and EO cycles. Detailed characterization was conducted on all components before and after sterilization.
Results:
Exposure to gamma radiation affected dexamethasone crystallinity and glucose response linearity of the sensing element, whereas physical aging of microspheres in composite coatings was observed poststerilization with EO. Despite these effects, dexamethasone drug release from coatings was not significantly affected by either technique.
Conclusion:
The research findings indicate that both sterilization techniques are feasible for the sterilization of the dexamethasone-loaded PLGA microspheres/PVA hydrogel composite coatings, while EO was preferred for the sterilization of the glucose-sensing element of the biosensor.
Keywords
Introduction
Sterilization of implantable medical devices is a critical step that has to be considered in the early product development stage to ensure safety and effectiveness of the device. 1 The commonly used sterilization techniques for implantable medical devices are exposure to dry heat, steam, radiation, and ethylene oxide (EO).2,3 However, the growing use of polymers in implantable medical devices alone or in conjunction with typically used device materials such as metals and alloys has introduced constraints on the sterilization techniques that can be used. 4 Biodegradable polymers such as polyesters are heat and moisture sensitive, and thus, cannot be sterilized using dry heat and steam sterilization.3,5 Exposure to ionizing radiation can be damaging to many polymers resulting in degradation due to chain scission as well as cross-linking. 6 Ethylene oxide sterilization is performed typically at high temperatures, which limits the use of this technique to heat sensitive polymers. 7 Several studies have investigated the impact of sterilization techniques on polymer-based formulations and medical devices.8-14 It has been observed that the reported results vary for each particular formulation or medical device since the sterilization process parameters typically need to be modified to accommodate the formulation/device characteristics.
Our group has previously developed a fully implantable glucose biosensor capable of continuous glucose monitoring.15-17 A biocompatible, biodegradable anti-inflammatory drug-eluting coating (composite coating) which encompasses the biosensor has been demonstrated as an effective strategy to counteract the in vivo foreign body reaction triggered by sensor implantation.18-20 In addition, coating allows the maintenance of long-term sensor functionality. The composite coating is composed of dexamethasone-loaded poly (lactic-co-glycolic acid) (PLGA) microspheres dispersed in a polyvinyl alcohol (PVA) hydrogel matrix. This coated implantable glucose biosensor can be classified as a drug-device combination product, which is an extension of implantable medical devices. Typically, electronic devices are sterilized using EO, whereas PLGA-based formulations are sterilized using gamma radiation as reported in the literature. 1 The high processing temperatures used during EO sterilization can be damaging to the polymers present in the composite coatings, and the ionizing gamma radiation is known to damage electronic components. Therefore, the selection of processing parameters used for sterilization for such coated implantable devices is challenging.
In this work, both gamma radiation and EO have been investigated for sterilization of the composite coatings, the individual components of the coatings as well as the glucose-sensing element of the biosensor. A low temperature cycle was used for both techniques and the sterilized samples were compared with nonsterilized controls. The stability of coatings, components, and sensing element was evaluated poststerilization. The composite coatings along with the individual components of the coatings were characterized for drug loading, morphology, glass transition, mechanical properties, and in vitro drug release. The glucose-sensing element was characterized for its glucose-sensing ability before and after sterilization.
Materials and Methods
Materials
Dexamethasone was purchased from Cayman Chemical (Ann Arbor, MI, United States). Poly (lactic-co-glycolic acid) Resomer RG503H 50:50 (MW 25 kD) was a gift from Boehringer-Ingelheim. Low-molecular weight polyvinyl alcohol (LMW-PVA, MW 30-70 kD) was purchased from Sigma-Aldrich (St. Louis, MO, United States). High-molecular weight PVA (HMW-PVA, 99% hydrolyzed, MW 133 kD) was purchased from Polysciences, Inc. (Warrington, PA, United States). Dichloromethane and dimethyl sulfoxide (DMSO, ACS grade) were purchased from Sigma-Aldrich (St. Louis, MO, United States). Acetonitrile (ACN, HPLC grade) and phosphoric acid (PA) were purchased from Fisher Scientific (Pittsburgh, PA, United States). Milli-Q water (EMD Millipore, Burlington, MA, United States) was used for all studies.
Methods
Preparation of Microspheres
Dexamethasone-loaded PLGA microspheres were prepared using an o/w emulsion evaporation/extraction technique. A total of 200 mg of PLGA was dissolved in 0.4 mL of dichloromethane in 50 mL Teflon tubes. A total of 50 mg of dexamethasone was dissolved in 0.1 mL of DMSO, transferred into the polymer solution, and vortexed at 3000 rpm for ten seconds. An aqueous phase of 1% (w/v) PVA in water mixed with 30% (v/v) DMSO was added to the dexamethasone polymer solution while vortexing at 3000 rpm. The resulting emulsion was diluted (up to 400 mL with water) and held at ambient temperature and pressure for three hours under stirring at 600 rpm and then vacuum dried to remove residual solvent. The hardened microspheres were collected, washed three times by centrifugation, freeze-dried, and stored at 4°C until further use.
Preparation of Composite Coatings
A total of 150 mg of dexamethasone-loaded PLGA microspheres were added to 1 mL of 5% (w/v) HMW-PVA solution in water. The mixture was sonicated for ten seconds to achieve a homogeneous suspension. Composite coatings were prepared by spreading the PLGA microsphere/PVA suspension in a two-piece grooved mold. The molds were further subjected to three freeze-thaw cycles, each cycle consisting of freezing for two hours at −20°C and thawing for one hour at ambient temperature. Following the freeze-thaw cycles, the molds were stored overnight at 4°C. The composite coatings were then removed from the molds and air-dried for several hours.
Gamma Sterilization
Dexamethasone-loaded PLGA microspheres, composite coatings, and their individual components (dexamethasone, PLGA, and HMW-PVA) as well as the glucose-sensing elements were subjected to gamma sterilization by Sterigenics (Corona, CA, United States). The samples were subjected to 25-kGy radiation dose using a 60Co source. The samples were exposed to gamma radiation for approximately eight hours and the temperature during the run was maintained at 4°C.
Ethylene Oxide Sterilization
Dexamethasone-loaded PLGA microspheres, composite coatings, and their individual components (dexamethasone, PLGA, and HMW-PVA) as well as the glucose-sensing elements were subjected to EO sterilization by Sterigenics (Salt Lake City, UT, United States). Prior to sterilization, the samples were preconditioned at 43.3°C and 65% relative humidity for 24 hours. The EO concentration was 699.6 mg/L and the gas dwell time was approximately six hours. The set point temperature during the run was 34°C (with min to max limits of 29°C-39°C).
Characterization of Microspheres and Composite Coatings
Drug Loading
Approximately 5 mg of microspheres or composite coatings (n = 3) were dissolved in 1 mL of DMSO and diluted ten times in acetonitrile. The dexamethasone concentration was measured using reversed phase high-performance liquid chromatography (RP-HPLC, Agilent). Mobile phase: acetonitrile/water/phosphoric acid (40/60/0.5 (v/v/v%)); column: Zorbax C18 (4.6 mm × 15 cm); detection wavelength: 240 nm; and flow rate: 1 mL/min. The drug loading was calculated as % drug by dry weight of either microsphere powder or composite coating.
Thermal Analysis
The glass transition temperature (Tg) of the polymer, microspheres, and composite coatings was measured using a modulated temperature differential scanning calorimeter (TA instrument Q2000). Approximately 4 mg of sample was placed in nonhermetic pans. Samples were subjected to a 2°C/min heating rate and a modulation amplitude of ±0.5°C with a 60-second modulation period. The Tg-midpoint was determined from the reversing heat flow signal and the enthalpy of relaxation peak was observed from the total heat flow signal.
Morphology
The morphology of the microspheres and coatings was determined using a scanning electron microscope (NanoSEM 450, Nova). Dry microspheres or coatings were mounted on carbon taped aluminum stubs and sputter coated with gold for three minutes at 6 mA before imaging.
X-ray Diffraction
The X-ray diffraction (XRD) patterns of crystalline dexamethasone and semicrystalline HMW-PVA samples were studied using an X-ray diffractometer (D2 Phaser, Bruker). Parameters: tube—Cu, voltage—30 kV, and current—10 mA. The scans were performed at a scanning rate of 0.02°/min from 5° to 45° (for dexamethasone) and from 5° to 50° (for HMW-PVA) (2θ values).
Mechanical Analysis of Coatings
Mechanical analysis was performed to obtain Young’s modulus (E) of the composite coatings using the Q800 (TA instruments) dynamic mechanical analysis instrument. The samples were subjected to 0.5 N/min up to 18 N. Young’s modulus was determined as the slope of the linear portion of the stress strain curves.
In Vitro Release Testing
Approximately 5 mg of composite coatings (n = 3) were placed in 10 mL of release medium (10 mM phosphate buffered saline [PBS] containing 0.01% (w/v) sodium azide, pH 7.4) in amber vials. The vials were kept under stirring (100 rpm) in a water bath incubated at 37°C. The entire medium volume was replaced periodically to ensure the maintenance of sink conditions (total dexamethasone concentration in the media never exceeded 10% of the equilibrium solubility, which is 100 μg/mL) and to prevent dexamethasone degradation. The concentration of dexamethasone in the medium was measured via RP-HPLC as described above.
In Vitro Testing of the Glucose-Sensing Element
In vitro testing of the glucose-sensing element (n = 1) of the implantable glucose sensor was performed in a stirred PBS solution (pH 7.4) maintained at 37°C and under an applied potential of 0.6 V vs a Ag/AgCl reference electrode using a CH instrument (Model: CHI840B) electrochemical analyzer. 17 The sensor response vs various glucose concentrations were determined by raising the glucose levels in the test cell by 2 mM every 200 seconds up to 20 mM, following an initial background stabilization period of ~15 minutes.
Results
Drug Loading
Figure 1(a) and (b) shows the drug loading in the nonsterilized, gamma, and EO sterilized microspheres and coatings. There were no significant differences observed in the dexamethasone loading of the microspheres and composite coatings postgamma sterilization and in the dexamethasone loading of the composite coatings post-EO sterilization compared to the respective nonsterilized controls. Owing to the deformation of the microsphere samples as a result of EO sterilization, drug loading was not determined in these samples.
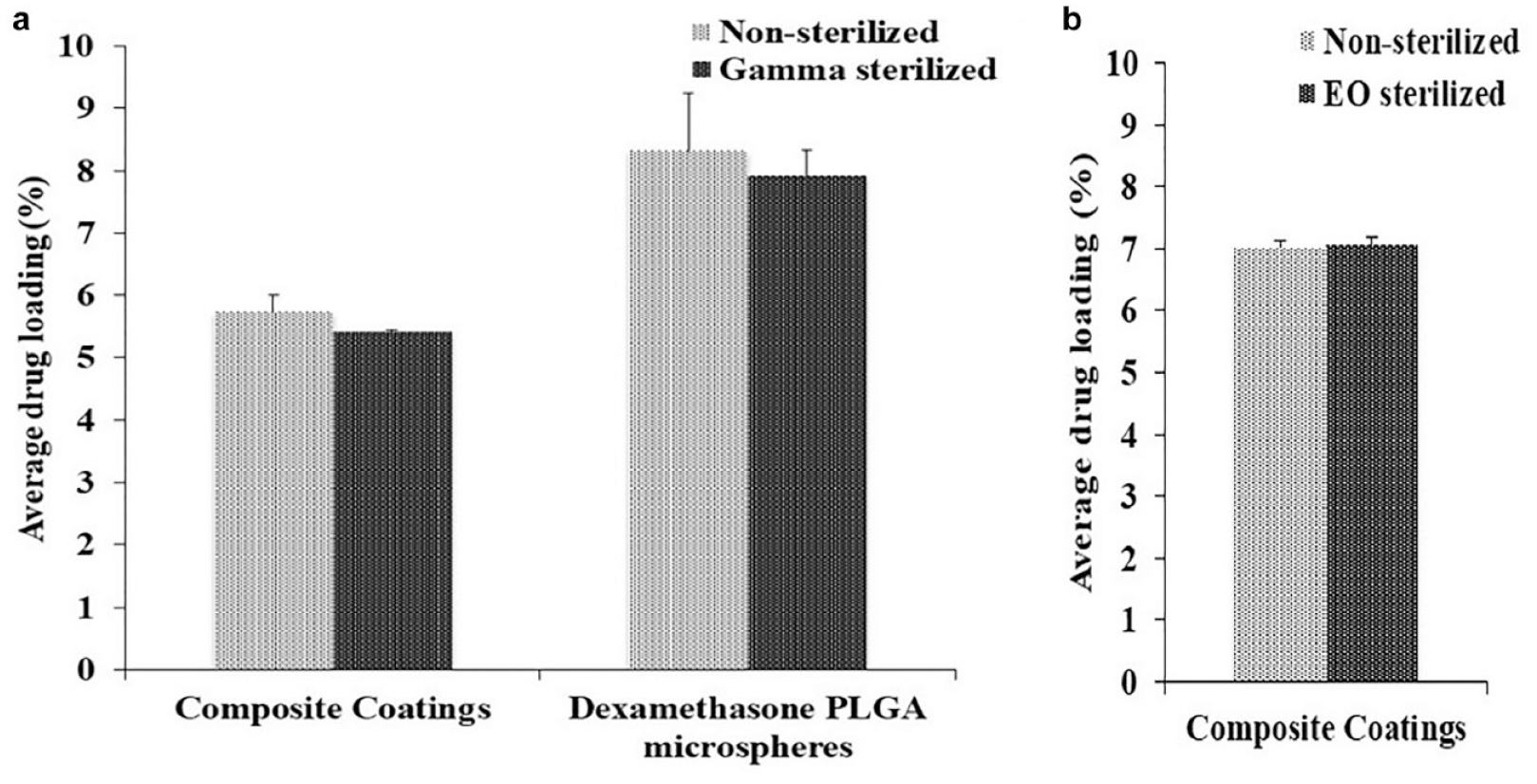
Drug loading: (a) nonsterilized and gamma sterilized composite coatings and microspheres and (b) nonsterilized and ethylene oxide sterilized composite coatings.
Morphology
Figure 2 shows scanning electron microscopy (SEM) images of nonsterilized, gamma sterilized, and EO sterilized microspheres and composite coatings. There were no morphological differences observed between the nonsterilized and gamma sterilized dexamethasone PLGA microspheres (Figure 2(a-1) and (b-1)) and composite coatings (Figure 2(a-3) and (b-3)). However, a deformation was observed in the dexamethasone PLGA microspheres post-EO sterilization (Figure 2(c-1)). The microspheres appeared to have aggregated to from a large mass as seen in the SEM image (indicated by the blue arrow). It was speculated that the observed deformation might be a result of PLGA plasticization. It is to be noted that due to the sample deformation observed in microspheres sterilized with EO, the same magnification could not be used for all microsphere samples. However, in the composite coatings post-EO sterilization (Figure 2(c-3)), the hydrogel matrix maintained the individual distribution of microspheres similar to that observed in nonsterilized coatings. Interestingly, there was a major morphology change observed in the microspheres embedded in the composite coatings sterilized by EO. As observed earlier in the cross-sectioned images of the nonsterilized composite coatings, multiple small pores were present throughout the microspheres. However, in the EO sterilized composite coatings, these pores were not observed. Instead, a cavity was observed in the center of some microspheres (indicated by the yellow arrows). In addition, some microspheres appeared to have shrunk and separated from the hydrogel matrix and some were subsequently displaced from the composite coatings while sectioning leaving behind pits. The observed morphology changes revealed polymer (PLGA) densification within the microspheres. Polymer densification might have occurred due to physical aging of dexamethasone PLGA microspheres as a result of the closeness of the processing temperature used during the EO cycle (34°C) to Tg of the PLGA polymer (45°C).

Scanning electron microscopy images of sterilized and nonsterilized microspheres and composite coatings.
As the hydrogel matrix prevented the merging of microspheres, the HMW-PVA, which is the major component of the hydrogel matrix, was itself evaluated for stability post-EO exposure. No differences were identified in nonsterilized and EO sterilized HMW-PVA samples (Figure 3(a-4) and (c-4)). The plasticization of the PLGA polymer itself post-EO sterilization was evident (Figure 3(c-5)) and was similar to that observed in the EO sterilized dexamethasone PLGA microspheres. It is to be noted that due to the sample deformation observed in PLGA sterilized with EO, the same magnification could not be used for all samples.

Scanning electron microscopy images of sterilized and nonsterilized high-molecular weight polyvinyl alcohol and poly (lactic-co-glycolic acid) polymer alone.
Thermal Analysis
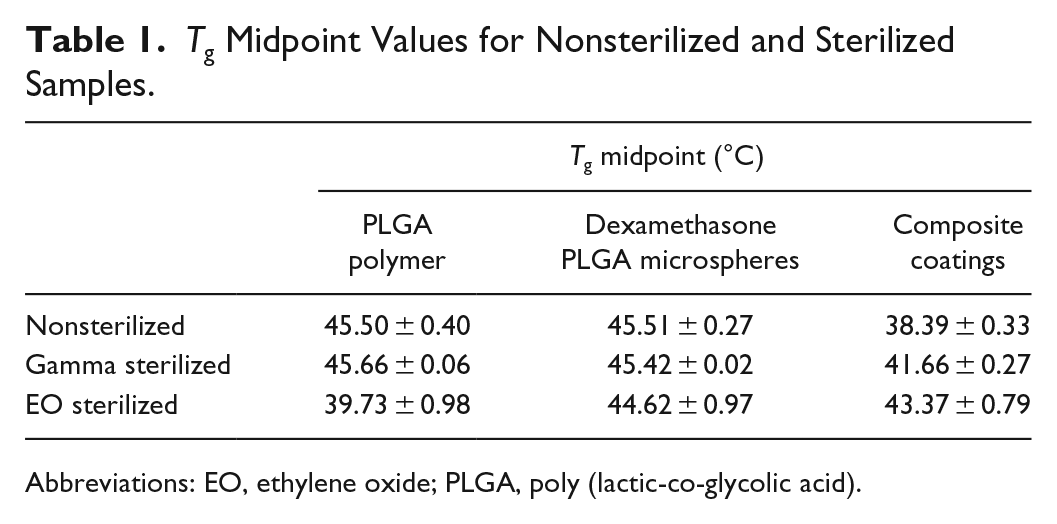
The midpoint Tg values are summarized in Table 1 for different sample types and the differential scanning calorimetry (DSC) curves with the Tg shifts are shown in Figure 4. It was observed that Tg of EO sterilized PLGA polymer (Figure 4(a)) decreased compared to nonsterilized and gamma sterilized samples confirming polymer plasticization (observed in Figures 2(c-1) and 3(c-5)). The DSC curve for dexamethasone PLGA microsphere samples (post-EO sterilization) had a wavy baseline (Figure 4(b)), which is probably a result of the inadequate contact between the sample and the aluminum pan due to deformation of the sample post-EO exposure. Both the nonsterilized and gamma sterilized microspheres had similar Tg midpoints. A large enthalpy of relaxation peak was observed in the EO sterilized composite coatings compared to the gamma and the nonsterilized composite coatings (Figure 4(c)). In addition, it was observed that the composite coatings sterilized using both techniques exhibited an increase in the Tg value (Figure 4(d)). In the case of the EO sterilized coatings, the increase in Tg confirms that more heat had to be supplied in order to induce movement in the polymer chains (enthalpy recovery) owing to the polymer densification that occurred during physical aging (at the EO processing temperature).
Tg Midpoint Values for Nonsterilized and Sterilized Samples.
Abbreviations: EO, ethylene oxide; PLGA, poly (lactic-co-glycolic acid).
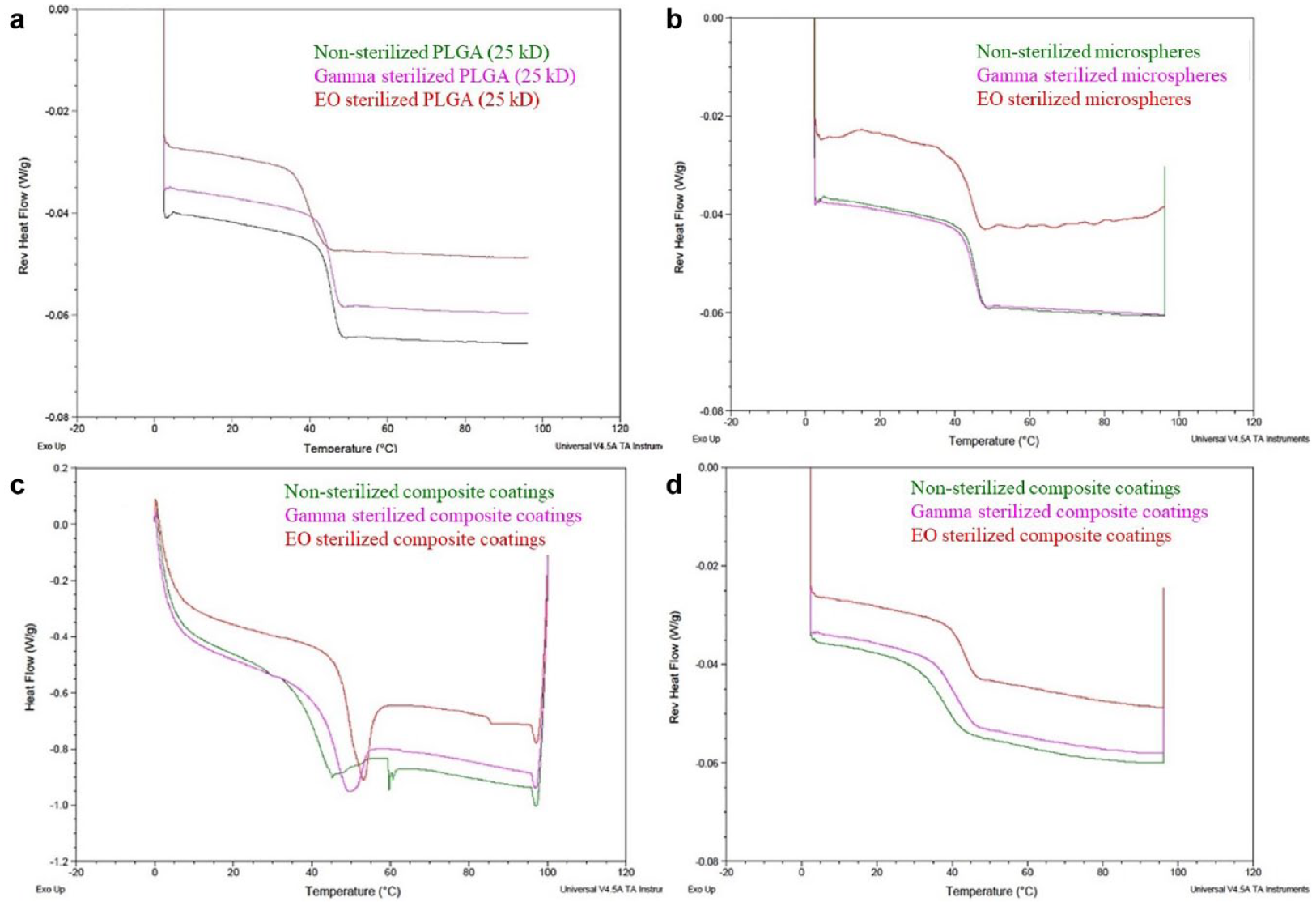
Differential scanning calorimetry curves of nonsterilized (green), gamma sterilized (pink), and ethylene oxide sterilized (red) samples: (a) poly (lactic-co-glycolic acid) polymer alone, (b) dexamethasone poly (lactic-co-glycolic acid) microspheres, (c) composite coatings (Tg along with enthalpy relaxation), and (d) composite coatings.
X-ray Diffraction
The XRD patterns (Figure 5(a)) observed for nonsterilized, gamma, and EO sterilized HMW-PVA overlapped well and showed the characteristic peak of PVA at 2θ = 19.8° in all three samples. The overlap also demonstrated the stability of HMW-PVA toward both sterilization techniques. In addition, the XRD patterns of dexamethasone, which is a crystalline small molecule, were also evaluated before and after sterilization (Figure 5(b)). It was observed that gamma sterilized dexamethasone showed significant peak shifts from the characteristic peaks (labeled in the graph) as well as the appearance of new peaks. However, no significant peak shifts were observed in EO sterilized dexamethasone.
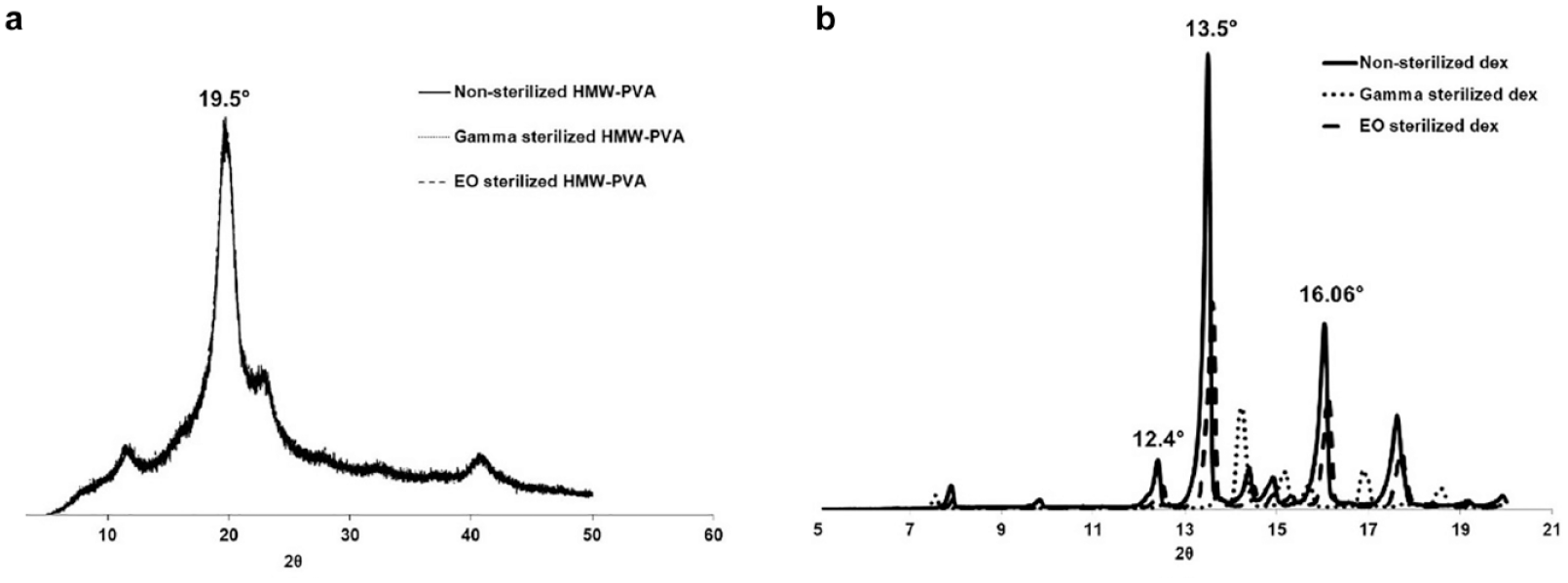
X-ray diffraction patterns of nonsterilized, gamma, and ethylene oxide sterilized: (a) high-molecular weight polyvinyl alcohol and (b) dexamethasone.
Mechanical Analysis
Young’s modulus was calculated as the slope of the linear part of the stress vs strain curve (Figure 6). It was observed from the stress vs strain curves that both nonsterilized and gamma sterilized samples overlapped in the initial linear range (0%-0.2% strain), whereas the EO sterilized samples had a completely different profile. Young’s modulus for nonsterilized and gamma sterilized composite coatings was 259.7 and 208.4 MPa, respectively. A high Young’s modulus of 577 MPa was observed for EO sterilized composite coatings, which demonstrated the brittleness of these coatings resulting from PLGA densification in the microspheres.
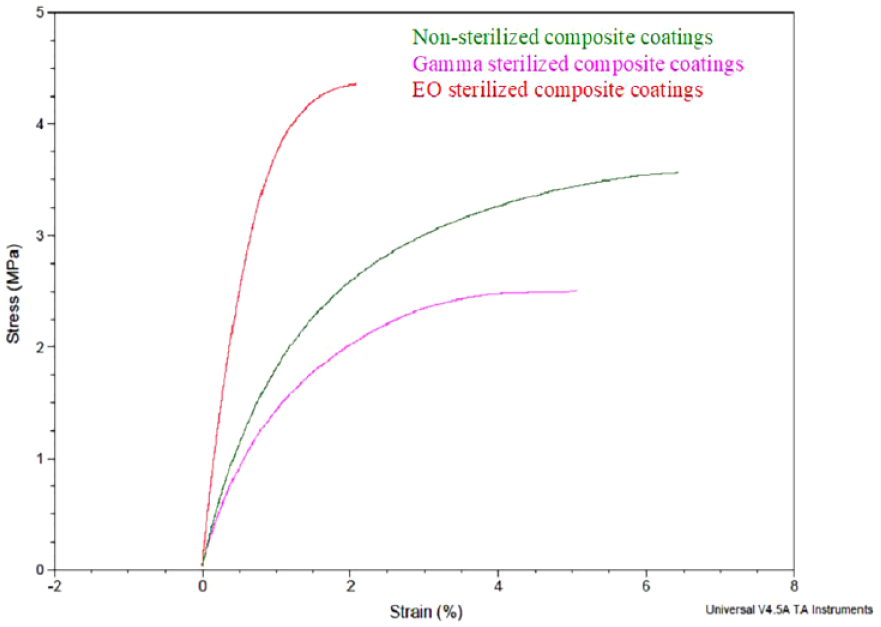
Stress vs strain curves of nonsterilized, gamma, and ethylene oxide sterilized composite coatings.
In Vitro Release Testing
The in vitro release profiles of nonsterilized and gamma sterilized composite coatings were similar to each other (Figure 7(a)). The differences observed in the dexamethasone cumulative release in the zero order release phase of gamma-sterilized coatings at later timepoints were within 10% of the nonsterilized controls. There was no effect on the burst release of samples postgamma sterilization.
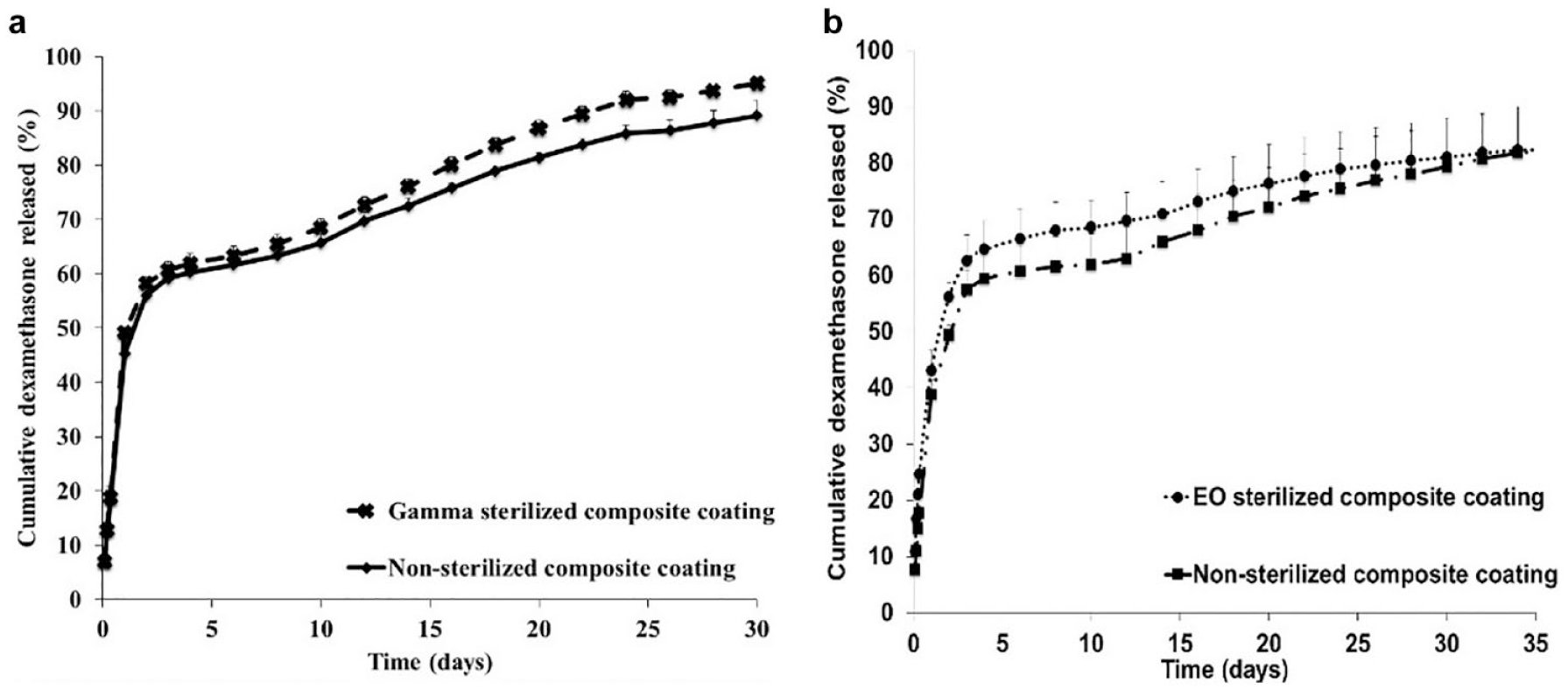
In vitro release profiles of nonsterilized, gamma, and ethylene oxide sterilized composite coatings.
The in vitro release profiles of EO sterilized composite coatings demonstrated a faster initial dexamethasone release and slower zero order release compared to its nonsterilized control. The high initial burst release may be due to the morphology change of the microspheres observed in the composite coatings post-EO sterilization, resulting in redistribution of dexamethasone crystals along with the observed polymer densification. The slow zero order release may be due to the long time required for water uptake and subsequent polymer degradation owing to the tight packing of the PLGA polymer chains postdensification.
In Vitro Testing of the Glucose-Sensing Element
The sensor response curves shown in Figure 8 were plotted using the absolute current value at different glucose concentrations. The amperometric response toward increasing glucose concentrations for nonsterilized and EO sterilized sensing elements were determined to be linear. However, the linearity of the sensing element was lost postgamma sterilization. A potential reason for this may be radiation induced damage to the polyurethane layer on the sensing element, which is responsible to maintain glucose sensing linearity.
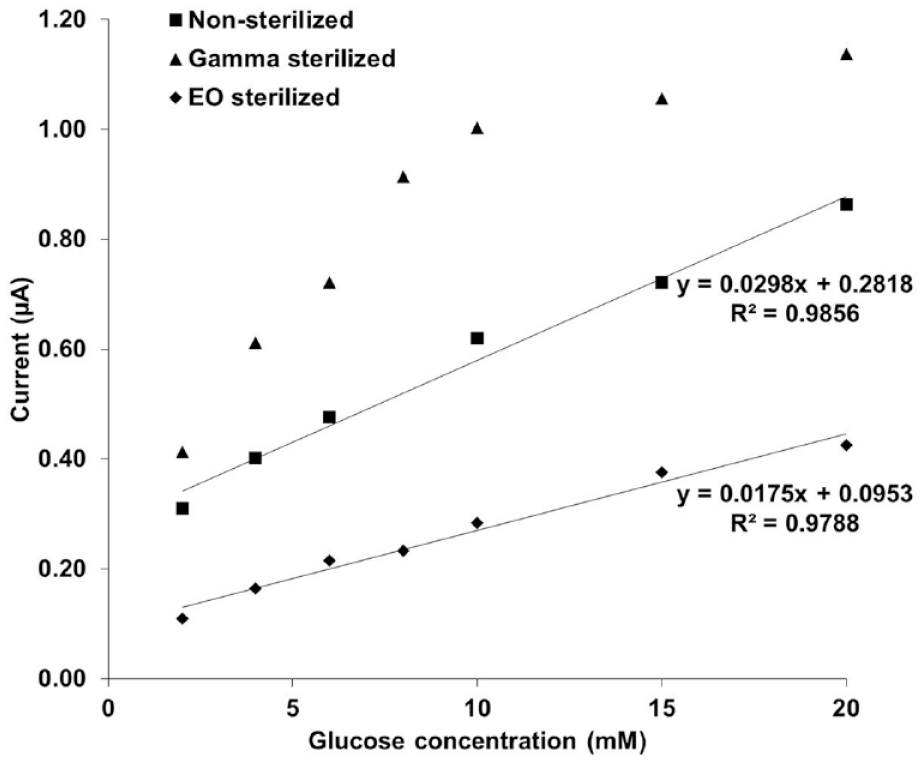
Current (µA) vs glucose concentration (mM) calibration curves for nonsterilized, gamma, and ethylene oxide sterilized glucose sensor.
Discussion
Dexamethasone loading in the microspheres and the composite coatings is one of the most important critical quality attributes as it has an impact on the total amount of drug present as well as on the drug release from the composite coatings. The sterilization techniques and parameters used, especially gamma radiation, can contribute negatively toward dexamethasone loading by inducing drug/polymer degradation and thus needed investigation. The results of the current studies indicate that there were no negative effects on dexamethasone loading in microspheres and composite coatings postgamma sterilization and in composite coatings post-EO sterilization (Figure 1).
The morphology of the microspheres and the composite coatings has not affected postgamma sterilization (Figure 2). However, major morphological changes were revealed in microspheres alone as well as microspheres embedded in the PVA hydrogel matrix (composite coatings) post-EO sterilization (Figure 2). Polymer densification was observed in the microspheres present in hydrogel matrix. This morphology change was due to the physical aging of PLGA microspheres caused as a result of the closeness of the processing temperature used during the EO cycle (34°C) to PLGA polymer Tg (45°C). It is known that amorphous materials are not in thermodynamic equilibrium at temperatures below their glass transition and tend to approach equilibrium, a process known as physical aging.21,22 The rate of physical aging depends on the proximity of the processing temperature to the glass transition temperature of the polymer. As a result of the presence of a solid support matrix provided by the HMW-PVA, the PLGA chains were constrained to move only within the space of the microspheres. The rearranging of PLGA polymer chains resulted in polymer densification, which was evident by the polymer shrinkage and cavity formation observed (free volume decrease). Polymer densification was not evident in the SEM images of the nonsterilized and the gamma sterilized composite coatings.
The glass transition analysis results provided confirmation that the physical aging occurred post-EO sterilization in two ways: (i) large enthalpy relaxation peak observed in EO sterilized composite coatings (Figure 4(c)) and (ii) increase in Tg indicating enthalpy recovery to counteract polymer densification (Figure 4(d)). It is to be noted that a small enthalpy relaxation peak was also observed in composite coatings postgamma sterilization, which did not affect the morphology or drug release.
In order to evaluate the stability of the crystalline (dexamethasone) and semicrystalline (HMW-PVA) components of the composite coatings, XRD patterns were analyzed. The results indicated that the HMW-PVA was stable poststerilization by both techniques. Dexamethasone was stable post-EO sterilization; however, it showed significant peak shifts post-gamma sterilization. The peak shifts may indicate a changing lattice structure (ie, presence of a different polymorph) and this requires further investigation for confirmation. However, the dexamethasone loading was unaffected and no degradation peaks were observed in the in vitro release analysis of composite coatings postgamma sterilization.
The results obtained for the mechanical analysis of the composite coatings in this work were analyzed using a more qualitative approach. The steep slope (high Young’s modulus) of the linear portion of the stress vs strain curve of the composite coatings post-EO sterilization indicated the brittle nature of these samples due to polymer densification. Whereas, the overlapping linear portions of the nonsterilized and gamma sterilized composite coatings indicated more flexibility in both samples.
Despite the radiation-induced changes observed in XRD patterns of dexamethasone, the in vitro drug release was similar before and after exposure to gamma radiation. Due to the morphology change in EO sterilized composite coatings, a high burst release followed by a slow zero order release phase was observed. During the PLGA polymer chain rearrangement, dexamethasone drug crystals can also get redistributed within the microspheres, resulting in high burst release. In addition, the close packing of these PLGA chains might result in slowing the polymer degradation and subsequent drug release.
The in vitro testing of the glucose-sensing element of the implantable biosensor was critical in determining the linearity toward physiological glucose concentrations poststerilization. Glucose sensing linearity is essential to ensure sensor functionality. The gamma radiation appears to have damaged the polyurethane layer (glucose flux limiting membrane 15 ) of the glucose-sensing element, which can be inferred based on the saturation of current attained at high glucose concentrations. The EO sterilized glucose-sensing element maintained linearity, similar to the nonsterilized control, however, the EO sterilized biosensors had a slightly reduced sensitivity to glucose, indicating that the sterilization step needs to be taken into account before design freeze of the sensing element.
Conclusion
Low-temperature gamma sterilization was identified as a suitable technique for sterilization of composite coatings and microspheres, whereas low-temperature EO was identified as a preferred technique for sterilization of dexamethasone, high-molecular weight PVA, and glucose-sensing elements of the implantable biosensor. Low-temperature EO demonstrates potential to be used as a single technique approach for sterilization of the entire drug device combination product. However, future studies investigating the impact on glucose diffusion through the composite coatings post-EO sterilization and long-term sensor functionality need to be performed.
Footnotes
Acknowledgements
The authors thank Sterigenics for assisting with sterilization of samples.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FP and FJ declare a competing financial interest with Biorasis Inc. as they both are cofounders of the company. MK and AL declare a competing financial interest with Biorasis Inc. as they both were partially employed by this company at the time of this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank US Army Department of Defense (Award W81XWH-15C-0069) for funding.
