Abstract
Background:
Intravenous vitamin C therapy has been associated with reduced mortality in patients with sepsis. Of potential concern with this therapy are falsely elevated point-of-care (POC) blood glucose values vs laboratory analyzed (LA) readings. The purpose of this study was to compare POC and LA blood glucose measurements in patients receiving intravenous vitamin C therapy.
Methods:
All adults (≥18 years old) admitted from January 2017 to December 2018 who received at least two doses of intravenous vitamin C and had at least one paired blood glucose collection were eligible for inclusion. The primary endpoint was the accuracy in paired blood glucose values determined using the International Organization for Standardization (ISO) 15197:2013 criteria. Paired values were assessed for clinical impact using the Parkes consensus error grid analysis. A subgroup analysis was conducted to determine the impact of impaired renal function on outcomes.
Results:
Fourteen patients were included for analysis with 46 paired blood glucose levels. Compliance with ISO15197:2013 criteria was met in 34 (73.9%) paired values, which did not meet the minimum criteria for accuracy. Subgroup analysis showed that the paired values from patients with impaired renal function did not meet the minimum requirements for compliance, while those from patients without impaired renal function did. The Parkes error grid showed that the variation in POC measurements likely had minimal clinical impact.
Conclusions:
Our study suggests that most patients receiving vitamin C for sepsis may still be monitored at POC with the glucose meter used in our study with minimal clinical impact.
Keywords
Introduction
Intravenous vitamin C (ascorbic acid) is an adjunct therapy that has shown varying degrees of benefit to a variety of critically ill patient population, most notably in burn resuscitation.1-3 Vitamin C is an antioxidant with anti-inflammatory and immune-modulating effects that mitigate the cellular damage. As described by Marik et al in 2017, intravenous vitamin C gained additional utilization as adjunct treatment for patients with sepsis. 4 While not approved by the US Food and Drug Administration (FDA) for this indication, use of adjunct intravenous vitamin C was shown in a retrospective observational study to decrease mortality by nearly five-fold in 47 patients with severe sepsis and septic shock. 4 However, the small study population coupled with minimal published data on the role of vitamin C in patients with sepsis leaves concern for potential risks associated with use.
Point-of-care (POC) blood glucose testing in the critically ill has demonstrated lower accuracy when compared to noncritically ill patients.5,6 The variability in accuracy of bedside glucose testing has been attributed to physiologic issues endemic in critically ill patients, such as decreased hematocrit7-10 and hypotension requiring vasopressor support,11-13 as well as use of certain medications such as acetaminophen or dopamine.14-17 Intravenous vitamin C has also shown POC interference, particularly associated with falsely elevated POC blood glucose measurements.14,18,19 POC glucose inaccuracies are documented specifically in burn patients receiving high dose intravenous vitamin C infusions. 19 This phenomenon is detrimental, possibly even fatal, if subsequently over-treated with insulin, creating the potential for dangerous hypoglycemic events.
Although falsely elevated POC measures are documented in burn patients receiving 100 to 150 g of vitamin C daily, it has not been well established in patients with sepsis receiving 6 g daily. 19 A study by Smith et al recently described the accuracy of POC glucose measurements in patients with sepsis receiving vitamin C, finding that while the paired values fell outside of the pre-specified margin for accuracy, the difference was not clinically significant. 20 Smith et al included 16 paired values from five patients in their analysis. The purpose of this study was to compare measurements collected from POC capillary blood glucose meters (BGMs) and laboratory analyzed (LA) venous glucose values in a larger group of patients with sepsis during intravenous vitamin C therapy.
Materials and Methods
This was a retrospective, single-center, observational case series of all patients admitted to a critical care unit at Baptist Health Louisville (a 519-bed community hospital in Kentucky in the Midwestern US) who received intravenous vitamin C for adjunct treatment of sepsis from January 1, 2017 to December 31, 2018. To be included for analysis, patients must have been 18 years or older, received ≥ two doses of intravenous vitamin C, and had ≥ one paired blood glucose collection, meaning that the values were drawn within one hour of each other. As this was not an interventional study, all paired glucose readings available were by chance alone. Patients were excluded if they were pregnant at the time of admission or a ward of the state. This study was evaluated and deemed exempt by the hospital’s Institutional Review Board.
Collected patient demographics consisted of age, sex, height, weight, and history of diabetes at admission. Timing of intravenous vitamin C, insulin, dextrose, and vasopressors were documented throughout vitamin C therapy duration. Use of other medications including acetaminophen, hydrocortisone, methylprednisolone, and insulin was also collected. The timing and measurements of blood glucose, both POC and LA, were collected while receiving vitamin C therapy. Daily serum creatinine was collected throughout treatment and used for assessing patient renal function. While serum creatinine is not an ideal marker for renal function in critically ill patients, the retrospective nature of this study limited our ability to consistently obtain a more useful marker such as a timed 24-hour urine creatinine or urine output. 21 Use and timing of intermittent hemodialysis (IHD) and continuous renal replacement therapy (CRRT) were also noted along with corresponding hematocrit values.
POC capillary glucose levels were evaluated using Accu-Chek® Inform II (Roche) BGMs at patient bedside using the glucose-dehydrogenase-pyrroloquinoline (PQQ) amphoteric method, while LA measures were completed by Cobas c702 analyzer (Roche) using the hexokinase spectrophotometric method. 22 As noted previously, paired POC and LA measures consisted of levels drawn within one hour of each other. Paired blood glucose levels must have been collected within six hours of administration of IV vitamin C. Blood glucose measurements occurring while patients were on continuous intravenous infusions of insulin, vasopressors, or dextrose were included if the infusion rate remained unchanged between collections. Paired glucose values were excluded if patients were administered insulin or initiated on vasopressor therapy between collections.
The primary endpoint of this study was to evaluate the accuracy of POC glucose measures among paired POC and LA glucose measures. BGM accuracy among paired POC and LA measures was assessed according to the ISO15197:2013 criteria. 23 For BGM measures to be deemed within compliance of the criteria, 95% of sample POC blood glucose values would need to be ± 15 mg/dL of reference LA ≤100 mg/dL, or within a 15% difference of reference LA >100 mg/dL. 23 BGM accuracy was further evaluated using the Bland-Altman Method to assess limits of agreement among paired blood glucose measurements. 24
Secondary endpoints included the clinical significance of difference in paired glucose values, and the accuracy of POC glucose measures among paired POC and LA blood glucose measures in patients based on renal function: nonimpaired, impaired, and patients requiring CRRT. Renal function was a fluctuating measure and determined with each day patients were receiving IV vitamin C therapy. Paired blood glucose values categorized as nonimpaired renal function were defined by collection within a 24-hour period of a patient’s serum creatinine <150% of the patient’s baseline creatinine level as available through the institution’s electronic medical record. 25 In instances for which an outpatient baseline creatinine level was not available, the patient’s creatinine level on admission was considered baseline. Paired blood glucose values determined to be impaired renal function were defined by blood serum creatinine ≥150% of the patient’s baseline creatinine level or receipt of IHD. 25 CRRT paired blood glucose values were defined by collection of levels within a 24-hour period as CRRT was performed.
Clinical significance of differences in paired blood glucose was assessed using the Parkes consensus error grid. 26 The Parkes consensus error grid is divided into five zones assessing the magnitude of BGM variation to the reference level. Each zone represents a degree of risk on patient outcomes if clinical action is taken based on POC measures. Zone A represents a difference between paired blood glucose values that would not affect clinical actions. Zone B is defined by altered clinical actions that would likely have little to no effect on patient outcomes. Zone C represents deviation in clinical outcomes that are likely to affect clinical outcome. Zone D represents altered clinical action that would pose a significant medical risk, and Zone E defines clinical actions that would have dangerous consequences on patient outcome. For this study, Parkes consensus error grid analysis was generated using the technology provided by the Blood Glucose Monitoring System (BGMS) Surveillance Program. 27
Results
Twenty-two patients were identified as receiving adjunct intravenous vitamin C for the treatment of sepsis between January 2017 and December 2018 at Baptist Health Louisville. No patients identified were pregnant nor wards of the state. All patients were ≥18 years old. Of the 22 patients, three were excluded for receiving less than two doses of intravenous vitamin C, and five excluded for lack of paired measures. Fourteen patients were included in statistical analysis, with a total of 62 paired measures identified for potential inclusion in the analysis. Sixteen paired glucose values were excluded based on the pre-determined parameters for exclusion stated in the methods, leaving a total of 46 paired glucose values for evaluation.
Table 1 displays the characteristics of the patients included in this study. The median age of the 14 patients included in analysis was 60.5 (IQR 52-66.25) years. Only three patients were noted as having a history of diabetes mellitus prior to admission. The median duration of vitamin C infusion among the study population was 54.6 hours. All but one patient required vasopressor therapy while receiving vitamin C therapy. Similarly, all but one of the study patients received intravenous hydrocortisone in conjunction with vitamin C. One patient received methylprednisolone instead of hydrocortisone due to a history of chronic obstructive pulmonary disease. All patients were treated with vitamin C 1500 mg IV every six hours, and thiamine 200 mg IV every 12 hours throughout the duration of vitamin C treatment. Ten patients were administered at least one dose of correctional insulin aspart, while two patients received intravenous insulin for the management of diabetic ketoacidosis during the course of vitamin C therapy. Three patients received CRRT during vitamin C therapy, while impaired renal function and nonimpaired renal function accounted for five and six patients, respectively.
Patient Demographics (n = 14).
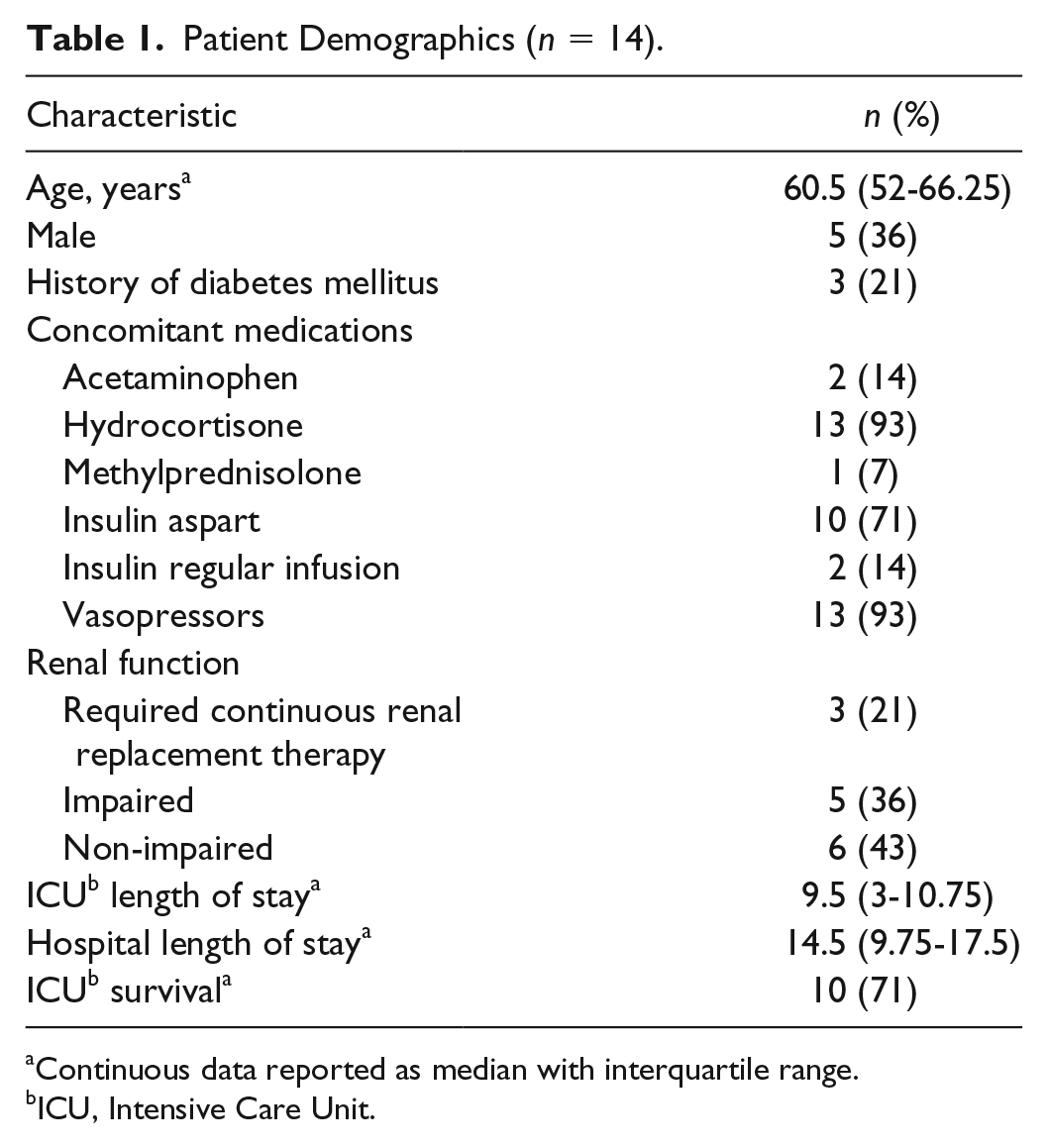
Continuous data reported as median with interquartile range.
ICU, Intensive Care Unit.
A full breakdown of how paired glucose values compared with ISO15197:2013 criteria ranges is available in Table 2. The mean time between POC and LA blood glucose collection among all paired values was 14.5 minutes (SD = 16.2). Compliance with ISO15197:2013 criteria (as defined previously) was met in 34 (73.9%) of the 46 paired blood glucose values evaluated, which fell below the 95% threshold required to be deemed in compliance with the criteria. Figure 1 displays the difference in POC measures as compared to the reference LA measures as evaluated via the Bland-Altman method. The mean difference found among the 46 paired measures was 0.33 mg/dL. Three (6.5%) paired blood glucose values fell outside of the 95% limits of agreement. Although POC testing did not meet ISO15197:2013 criteria, the POC variation likely had little clinical impact on patient outcomes. Thirty-nine (84.8%) paired blood glucose values fell within Zone A in the Parkes consensus error grid analysis (shown in Figure 2), with six (13%) and one (2.2%) in Zone B and C, respectively. No interventions to address glucose levels occurred in any of the patients evaluated.
Difference of Point-of-Care (POC) Compared to Laboratory Analyzed (LA) for All Paired Glucose Values (n = 46).
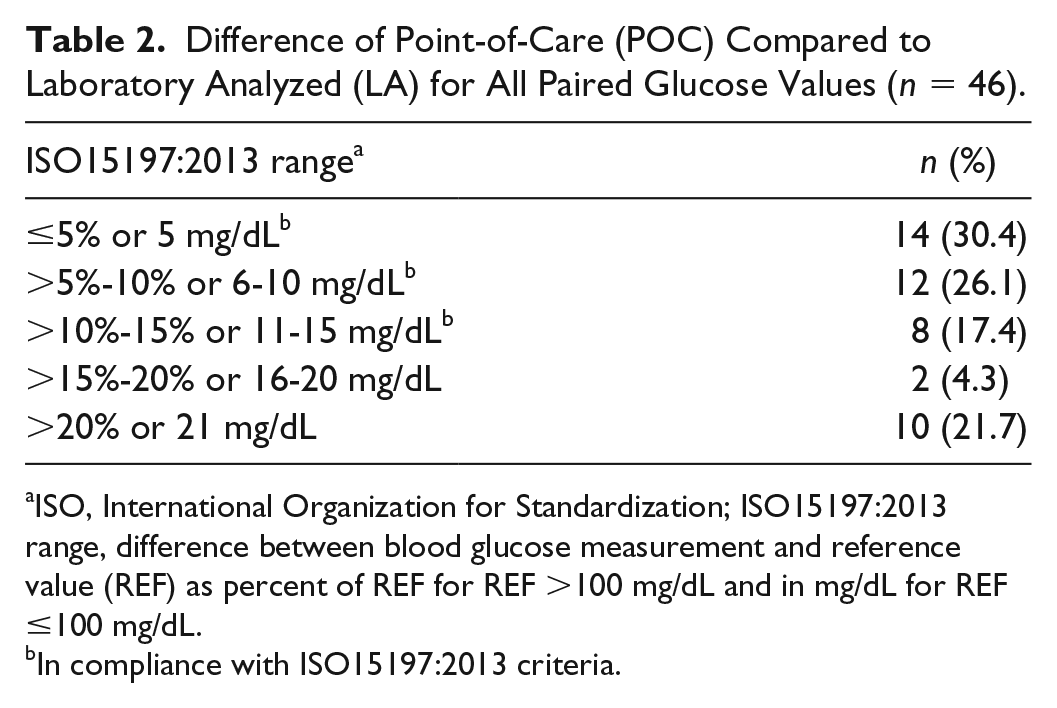
ISO, International Organization for Standardization; ISO15197:2013 range, difference between blood glucose measurement and reference value (REF) as percent of REF for REF >100 mg/dL and in mg/dL for REF ≤100 mg/dL.
In compliance with ISO15197:2013 criteria.
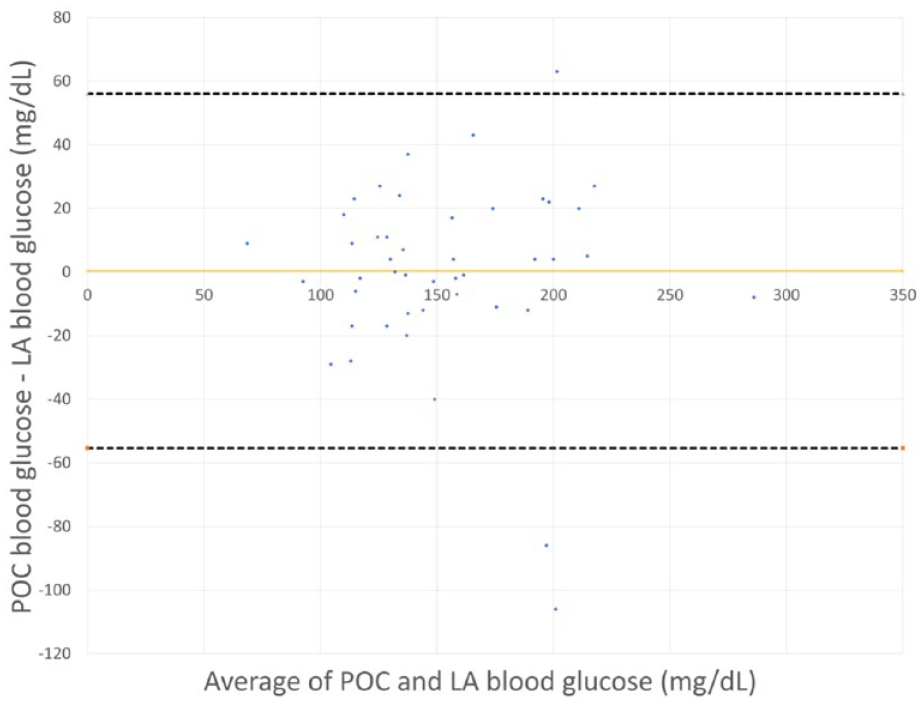
Comparative difference between paired point-of-care (POC) and laboratory-analyzed (LA) blood glucose levels. The solid line represents the mean difference among the compared data (also known as bias), while the dashed lines represent the 95% limits of agreement among the data points.
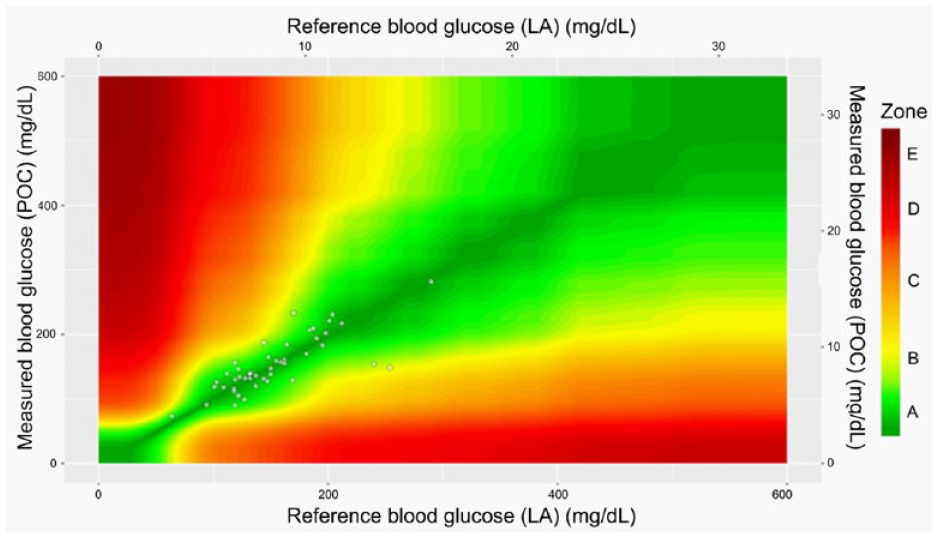
Parkes consensus error grid analysis of paired blood glucose readings between measured blood glucose (point-of-care; POC) and reference blood glucose (laboratory analyzed; LA). The Parkes error grid is divided into five zones assessing the magnitude of variation to the reference level. Each zone represents a degree of risk on patient outcomes if clinical action is taken based on POC measures. Zone A represents a difference between paired blood glucose values that would not affect clinical actions. Zone B is defined by altered clinical actions that would likely have little to no effect on patient outcomes. Zone C represents deviation in clinical outcomes that are likely to affect clinical outcome. Zone D represents altered clinical action that would pose a significant medical risk, and Zone E defines clinical actions that would have dangerous consequences on patient outcome.
Full results for compliance with ISO15197:2013 criteria for paired glucose values when stratified for patient renal function are available in Table 3. Seventeen paired blood glucose values were identified as nonimpaired renal function. Compliance with ISO15197:2013 criteria was met in fourteen (82.4%) paired blood glucose values of those identified in nonimpaired renal function patients. There were 11 paired blood glucose values in the subgroup analysis for impaired renal function. Compliance with ISO15197:2013 criteria was met in five (45.5%) paired blood glucose values of the 11 evaluated. Eighteen paired blood glucose values were collected within 24 hours of patients receiving CRRT. Compliance with ISO15197:2013 criteria was met in 15 (83.3%) paired blood glucose values of those identified in patients receiving CRRT.
Difference of Point-of-Care (POC) Compared to Laboratory Analyzed (LA) for All Paired Glucose Values Based on Renal Function (n = 46).
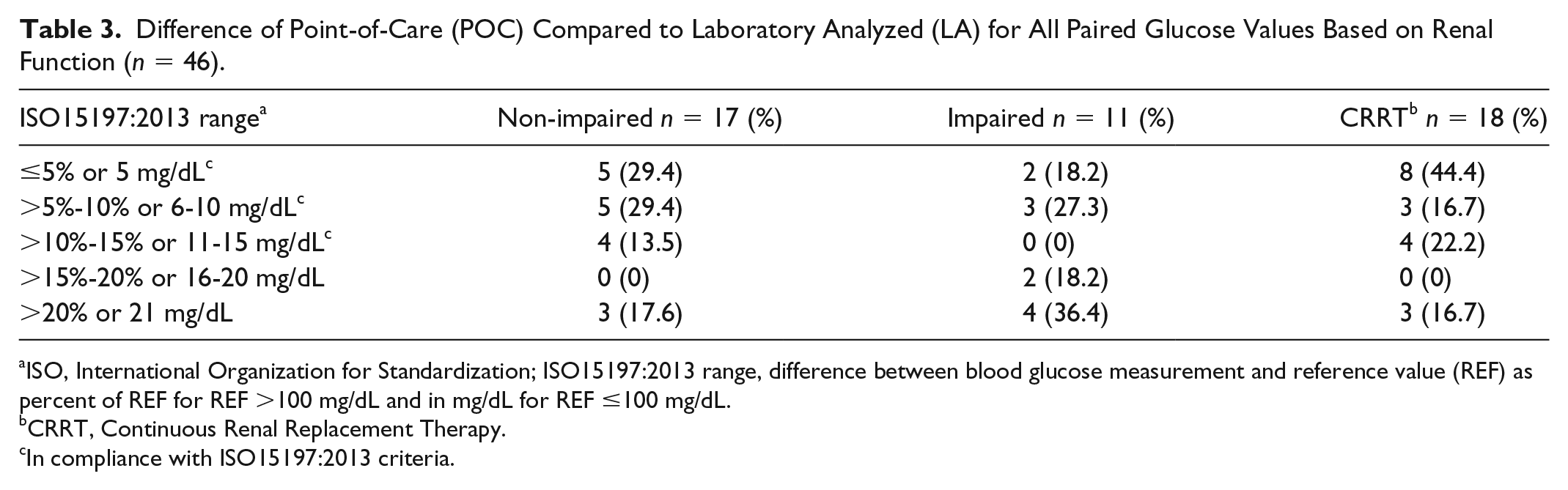
ISO, International Organization for Standardization; ISO15197:2013 range, difference between blood glucose measurement and reference value (REF) as percent of REF for REF >100 mg/dL and in mg/dL for REF ≤100 mg/dL.
CRRT, Continuous Renal Replacement Therapy.
In compliance with ISO15197:2013 criteria.
A subgroup analysis was conducted assessing the differences in variables around the collection of POC and LA blood glucose values (ie, time between collections, timing of vitamin C infusion). Supplemental Table 1 displays factors that could have potentially impacted the variability in POC and LA blood glucose measures across three varying degrees of patient renal function at the time of blood glucose collection. The median time between POC and LA collections was similar in both CRRT and impaired groups, though the nonimpaired renal function group showed a longer time interval between paired blood glucose values. There was a notable difference in the proportion of patients in the impaired renal function group who received concomitant vasopressor therapy (91%), as compared with the CRRT group (56%) or the nonimpaired renal function group (41%).
Discussion
Falsely elevated POC blood glucose measures have been reported safety a -risk in burn patients receiving large doses of vitamin C between 100 and 150 g daily and has even reportedly caused death when over-treated with insulin.18,19 It is recommended that glucose management should be managed with laboratory analyzed methods for burn patients receiving vitamin C. 19 The results of this study provide insight to the accuracy of- POC accuracy in patients with sepsis, and furthermore evaluates patient risk when treatment decisions are made based on the varying degree of POC accuracy compared to reference measures.
There is only one other similar study to-date evaluating the accuracy of bedside POC blood glucose management in patients with sepsis receiving 6 g of intravenous vitamin C therapy daily. Smith et al evaluated 16 paired glucose values from nine patients using the ISO15197:2013 criteria as well as the Bland-Altman method and Parkes consensus error grid analysis. 20 In their study, the accuracy of the POC BGM (Abbott Freestyle Precision Pro) was 81.3%, which failed to meet the minimum accuracy requirement of ISO15197:2013 criteria of 95%. This was similar to our study, which used the Accu-Chek® Inform II BGM and found an accuracy of 73.9% of paired glucose values, also below the minimum ISO15191:2013 accuracy requirement of 95%. Additionally, Smith et al found that while the accuracy failed to meet the minimum ISO15191:2013 requirement, the Parkes consensus error grid analysis showed that the variation was expected to have no impact on clinical outcome. Our study found the overwhelming majority (84.8%) of the paired glucose values fell within Zone A, meaning no expected impact on clinical action. Given the known variability that can occur between POC BGMs, 28 our results add additional value with both a higher number of paired values and patients evaluated and the use of a different POC BGM.
The mechanism hypothesized to explain falsely elevated POC measurements is related to the glucose-dehydrogenase-PQQ amphoteric method commonly employed by BGMs to determine serum blood glucose. 22 In this method, glucose dehydrogenase uses glucose to convert nicotinamide adenine dinucleotide to 1,4-dihydronicotinamide adenine dinucleotide (NADH). NADH then donates an electron to the electrode inside the BGM, ultimately measuring blood glucose levels. Electron donation to the electrode within the BGM is not specific to glucose, as other molecules are notes to participate in electron donation.6,18 Molecules similar in structure to glucose (such as maltose, galactose, and xylose) can be detected by glucose-dehydrogenase-PQQ amphoteric methods, generating elevated blood glucose measurements.6,29,30 Similarly, several medications, intravenous vitamin C included, have been determined to inflate POC glucose measurements.7,18,19,29-32
Conversely, laboratory analyzed blood glucose measurements are void of inaccuracies in the presence of vitamin C. The hexokinase spectrophotometric method, commonly used for laboratory analyzed blood glucose, does not generate measurements based on electron donation as the glucose-dehydrogenase-PQQ amphoteric method, but instead by measuring NADH.6,18 This method is highly specific to glucose, therefore is not impacted by vitamin C, or other molecules in the collected specimen. Use of LA blood glucose is recommended to POC bedside monitoring in patients receiving high-dose intravenous vitamin C.18,19,29
Our study suggests that the varying mechanisms of blood glucose testing did have an impact in the accuracy of POC testing in the patients receiving intravenous vitamin C for the treatment of sepsis. Compliance with ISO15197:2013 criteria was identified in 73.9% of the paired blood glucose values, which did not meet the after-market lower bound of acceptance of the BGMS Surveillance Program. Our results were similar to those found by Smith et al, which showed compliance with 81.3% of POC measures. 20 Although the Accu-Chek® Inform II BGM did not meet performance standards in this specific patient population, the differences between POC measures and LA measures were found to have a minimal risk to patients according to the Parkes error grid (shown in Figure 2). The theoretical interaction vitamin C has with POC testing assumes that POC inaccuracies would likely be greater than the LA comparator; however, our study suggests that intravenous vitamin C therapy for patients with sepsis likely did not have a clinically significant impact on POC inaccuracies.
High serum vitamin C levels have been shown to have a direct impact on the accuracy of POC readings. With increase in serum vitamin C, BGMs are more likely to generate elevated blood glucose readings.18,19,30,33 A study by de Grooth et al evaluating the pharmacokinetics of vitamin C in critically ill patients found that bolus doses of 2 g per day resulted in normal serum concentrations of 29-50 mg/L one hour after infusion, whereas bolus daily doses of 10 g resulted in supranormal serum concentrations of 186-244 mg/L one hour after infusion. 34 However, it is important to note that the study by de Grooth et al excluded patients with renal function, which was not the case for our study. There is no available literature evaluating serum vitamin C concentrations in patients with sepsis; however, it has been estimated that receiving 1.5 g of intravenous vitamin C every six hours results serum concentrations around 35.2 mg/L. 35 This estimated serum concentration is much lower than those found in patients receiving high-dose intravenous vitamin C; however, any change in the potential for interference with POC glucose measurements is unknown.
Serum vitamin C levels are highly dependent on the dose administered as well as on its elimination. Vitamin C is a water-soluble vitamin freely filtered in the glomerulus and reabsorbed in the proximal tubules via the sodium-dependent vitamin C transporter 1. 36 Given this dependence on renal elimination, it can be hypothesized that clearance and elimination of vitamin C from the body is impacted by renal function. Critically, ill patients with sepsis often have a varying degree of renal function from baseline, as these patients may have acute kidney injury from hypoperfusion. It is unclear if the estimated serum concentration of 35.3 mg/L in patients receiving 6 g of intravenous vitamin C daily takes patient renal function into consideration.
In our study, patient renal function did appear to have an impact on the risk of POC inaccuracies. However, firm conclusions are difficult to draw given the relatively small number of patients and paired glucose values evaluated as well as the comparatively higher use of vasopressors in the impaired renal function group (a drug class thought to interfere with POC glucose readings). There was a larger difference seen among the paired blood glucose values taken from patients with renal impairment (not requiring CRRT). However, the differences identified did not seem to have a major impact on patient risk using the Parkes error grid analysis. Although patients with sepsis do not receive the high doses of vitamin C as compared to burn patients that have documented reports of this phenomenon, the results of this study do suggest the possible role that impaired renal function may have in the risk of POC glucose inaccuracy. However, as noted previously, the small numbers of patients and paired glucose values in our study do not allow for firm conclusions to be drawn. Additionally, our study did not assess serum vitamin C levels so no definitive conclusions can be drawn. It is also notable that the proportion of patients receiving concomitant vasopressor therapy was significantly higher in the impaired renal function group (compared to the CRRT or nonimpaired renal function groups), as previous data have suggested that POC capillary blood glucose readings may be affected by vasopressor administration. 37 But given the small number of paired values in our subgroup analysis, however, it is difficult to draw conclusions. Further research with a larger number of patients is needed.
Recent literature has suggested that blood glucose measurements can be affected by the duration of time from when a blood sample is drawn from the patients to the time it is analyzed, with delays of 15 minutes or more possibly affecting outcomes.14,38 In an effort to assess the potential impact of this effect, a secondary subgroup analysis was performed of paired values in our data set that were drawn within 15 minutes of each other. Thirty-one of the 46 paired glucose values (67.4%) were drawn within 15 minutes of each another, with the mean time between paired values in this subgroup of 5.4 minutes (SD = 4.8). As shown in supplemental Table 2, 25 of the 31 (80.7%) paired values drawn within 15 minutes of one another met the ISO15197:2013 criteria, which fell below the 95% threshold for compliance. However, as shown in supplemental Table 3, 31 of the 31 (100%) paired values drawn within 15 minutes of one another fell within Zones A or B of the Parkes consensus error grid, meaning that a clinical impact was unlikely. Further studies should be conducted with more importance placed on the potential impact of postphlebotomy delays on processing and analysis.
While the results of this study are significant, there are also important limitations to note as they may have had an impact on reported results. First, our institution uses POC capillary glucose readings in critically ill patients like those enrolled in our study. Recent data have shown that the accuracy of capillary glucose is uncertain compared to that of arterial glucose. 39 It is unknown how this may have affected our results. Only 46 matched readings from 14 patients were eligible for inclusion in our analysis. Given this small number of samples, it is difficult to conclude significant differences exist or do not exist in this area. However, published data in this area are extremely limited. Our results support those of previous studies, as noted previously, and we believe they could help clinicians make decisions about how to use and interpret POC glucose values in septic patients receiving intravenous vitamin C therapy. There has also been recent evidence published that the POC BGM employed by our institution and used in this study (Accu-Chek® Inform II) showed analytical interference with ascorbic acid, lactose, galactose, N-acetylcysteine and glutathione in neonatal patients. 40 Further studies should be conducted with both this BGM and others to assess interference of ascorbic acid as well as other chemicals on blood glucose accuracy. Additionally, this study was a retrospective case series; thus, all data collected were highly dependent on the accuracy of documentation. Results were specific to the time of blood glucose collections, titration, and administration of several medications. Inaccuracies in documentation could have skewed the results. Another limitation to this study was the high variability among all paired blood glucose values in time between collections, co-administration of medications that could potentially interfere with POC glucose measurements, patient renal function, and underlying patient co-morbidities. POC accuracy is certainly multifaceted, and many of these circumstances could have impacted results. Furthermore, our facility exclusively uses Accu-Chek® Inform II BGMs; thus, the accuracy of other devices was unable to be evaluated.
Conclusion
While POC glucose monitor has previously been shown to be unreliable in certain populations receiving high doses of intravenous vitamin C, the results from our study suggest that many patients with sepsis receiving intravenous vitamin C therapy can be managed with POC glucose monitoring using the Accu-Chek® Inform II POC BGM without significant clinical impact. Further research should investigate the clinical outcomes of POC testing in patients with impaired renal function, using a wider variety of POC BGMs, and with a larger cohort of patients across several institutions.
Supplemental Material
POWELL_Supplementary_File_Rev1 – Supplemental material for Impact of High-Dose Intravenous Vitamin C for Treatment of Sepsis on Point-of-Care Blood Glucose Readings
Supplemental material, POWELL_Supplementary_File_Rev1 for Impact of High-Dose Intravenous Vitamin C for Treatment of Sepsis on Point-of-Care Blood Glucose Readings by Anna Peyton Howell, Jenna L. Parrett and Daniel R. Malcom in Journal of Diabetes Science and Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
