Abstract
Background:
A Danish study showed that 90% of the pediatric patients who participated had some time experienced dermatological complications due to treatment with continuous subcutaneous insulin infusion (CSII). This follow-up study describes dermatological complications due to CSII and/or continuous glucose monitoring (CGM) between the two study periods and includes health-related quality of life (HrQoL) measurements.
Methods:
A total of 138 patients (95%) out of 145 patients from the initial study answered an online questionnaire regarding dermatological complications related to CSII and/or CGM, five months later. A second questionnaire (DISABKIDS) regarding HrQoL was sent out to those 138 of which 111 patients completed it. The patients were aged from 2 to 20 years. Descriptive statistics, χ2 tests, and univariate and multivariate analyses were used to analyze the data.
Results:
In total, 81% of the 138 patients continued to have dermatological complications at follow-up. Itching was the most frequently reported complication. Patients using Enlite reported more dermatological complications than those using Libre. In total, 79% of the patients who used barrier cream in the initial study still had dermatological complications five months later. Age, gender, Body Mass Index (BMI), or HbA1c levels showed no significant association with dermatological complications. Patients who perceived dermatological complications as a greater problem had lower HrQoL scores.
Conclusion:
Once dermatological complications start to appear, they become chronic, indicating that the treatments currently available are inadequate. Patients perceiving dermatological complications as a greater problem were associated with lower HrQoL. These findings highlight the need for additional preventive studies.
Introduction
Children diagnosed with type 1 diabetes (T1D) depend on insulin, which can be delivered by continuous subcutaneous insulin infusion (CSII) attached to the body in combination with or without continuous glucose monitoring (CGM). 1 The previous studies have shown that 43% to 90% of the participating children experienced dermatological complications and that these types of complications tend to present in younger children.2,3 Various dermatological complications have been observed including scars, nodules, and eczema (erythema, scaling, papules, and sometimes blisters) due to irritative or allergic contact dermatitis under the adhesives of sensors and pump sets and lipohypertrophy/-atrophy due to insulin infusion. 4
A study from Poland showed that poor well-being during CSII treatment and embarrassment were among the major reasons for discontinuing CSII in children. 5 The effects of dermatological complications due to CSII and/or CGM on health-related quality of life (HrQoL) have received little or no attention, although the previous studies have shown lower HrQoL in children and caregivers with other dermatological diseases.6,7
There have been no previous follow-up studies on children with T1D and dermatological complications as CSII and/or CGM exists.8,9 The purpose of this follow-up study was to describe fluctuations and variations in dermatological complications over time and assess what influence treatment or preventive care has had on dermatological complications in between the two periods, and furthermore, to include measures of HrQoL in these patients.
Methods
Study Design
This was a cross-sectional study following up on an initial study. 2 For both studies, informed consent was obtained from participants’ parents and from adolescents aged 15 years or older. The study was approved by the Danish Data Protection Agency and the Research Ethics Committee (journal number: H-19017821).
Questionnaires
The follow-up questionnaire was sent to all patients four months after the initial study. Both of the questionnaires were sent to either patients or their parents with an online link and were completed at home. Patients answered the questionnaires approximately five months after the initial study. (mean: 5.09 months; 95% confidence interval [CI]: 4.89-5.29).
The questionnaire concerning dermatological complications due to CSII and/or CGM was the same as that used in the initial study. 2 New questions were included regarding differences in treatment since patients had answered the first questionnaire, why changes were made and whether the patients’ dermatological complications had changed.
The DISABKIDS chronic-generic (DCGM-37) and diabetes-specific (DSM-10) modules were used to measure HrQoL.10,11 Both the DCGM-37 and the DSM-10 have been forward and backward translated into Danish and tested valid and reliable. 12 An additional question was added to establish whether the patient or the parent answered the questionnaire. Scores within each dimension were linearly transformed into a 0 to 100 score. The orientation of all subscales was in the same way, and a higher score indicated better HrQoL for all dimensions and overall scores.
Statistics
Descriptive statistics were used to present age, sex, BMI, HbA1c, duration of T1D, and CSII and CGM treatments. Paired t-tests were used to compare dermatological complications reported in the initial and follow-up questionnaires. Independent sample t-tests were used to analyze differences in discomfort scores. Normality was assessed by visual inspection of histograms.
Different dermatological complications were seperated into groups for further analyses. The following classification is based on the initial study and what the patients were given as options for dermatological complications in the questionnaire. The main groups were itching, specific current dermatological complications (eczema, wounds, and scars), and all currently visible dermatological complications including lipoatrophy, lipohypertrophy, changes in pigmentation, red/blue dot (small hematomas), and other types. The patients were separated into commonly used pump-duration categories: zero to one year, one to four years, four to six years, and over six years. Univariate and multivariate logistic regression were used to analyze correlation between CSII/CGM type, duration of treatment, specific current dermatological complications, itching, and all current dermatological complications, when adjusted for potential confounders: HbA1c, age, sex, and BMI. Shapiro-Wilk and Kolmogorov-Smirnov tests were used to test for normal distribution with a P-value <.05 confirming that data were not normally distributed. In those cases, values were reported as medians with interquartile ranges instead of means with standard deviation.
For HrQoL, both Wilcoxon tests and multivariate linear regression were used to assess data normality and significance in all eight dimensions as well as the overall scores for generic and diabetes-specific DISABKIDS both with and without HbA1c, age, and sex being adjusted for. Associations between discomfort scores and HrQoL scores were determined using Spearman’s correlation. Bland-Altman plots and unpaired t-tests were used to compare the patients’ responses with those of their parents.
Level of significance was assessed at 5%, and 95% CIs were calculated. For different analyses, both Microsoft Excel and SAS software were used.
Results
Figure 1 shows a patient inclusion flowchart and data on the development of dermatological complications in this population. Table 1 shows basic demographics for the patients from Herlev and Hillerød Hospitals.
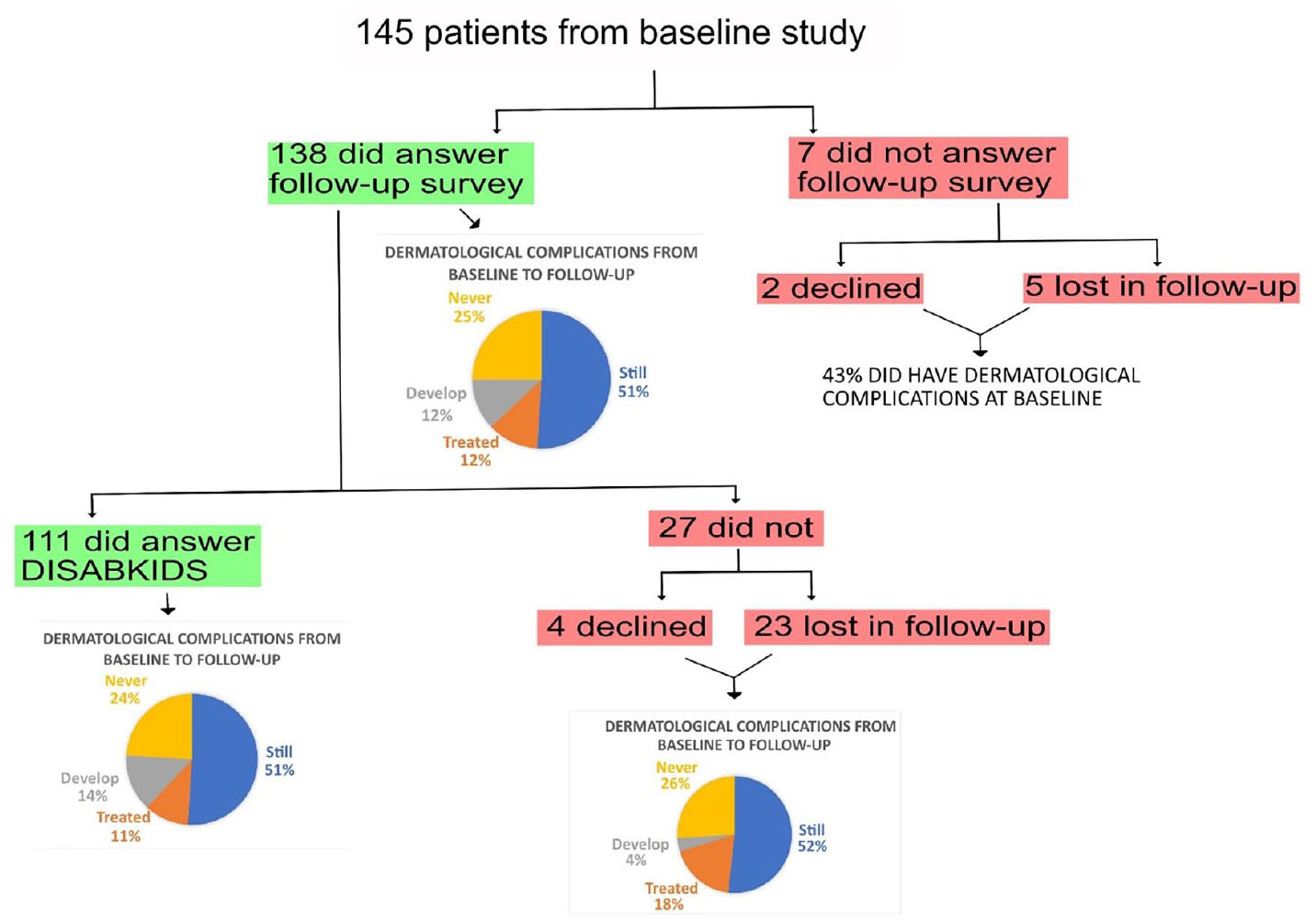
Patient inclusion flowchart. Patients from both Herlev and Hillerød Hospitals were included. The mean follow-up time was 5.09 months (CI: 4.89-5.29).
Basic Demographics for Participating Patients.
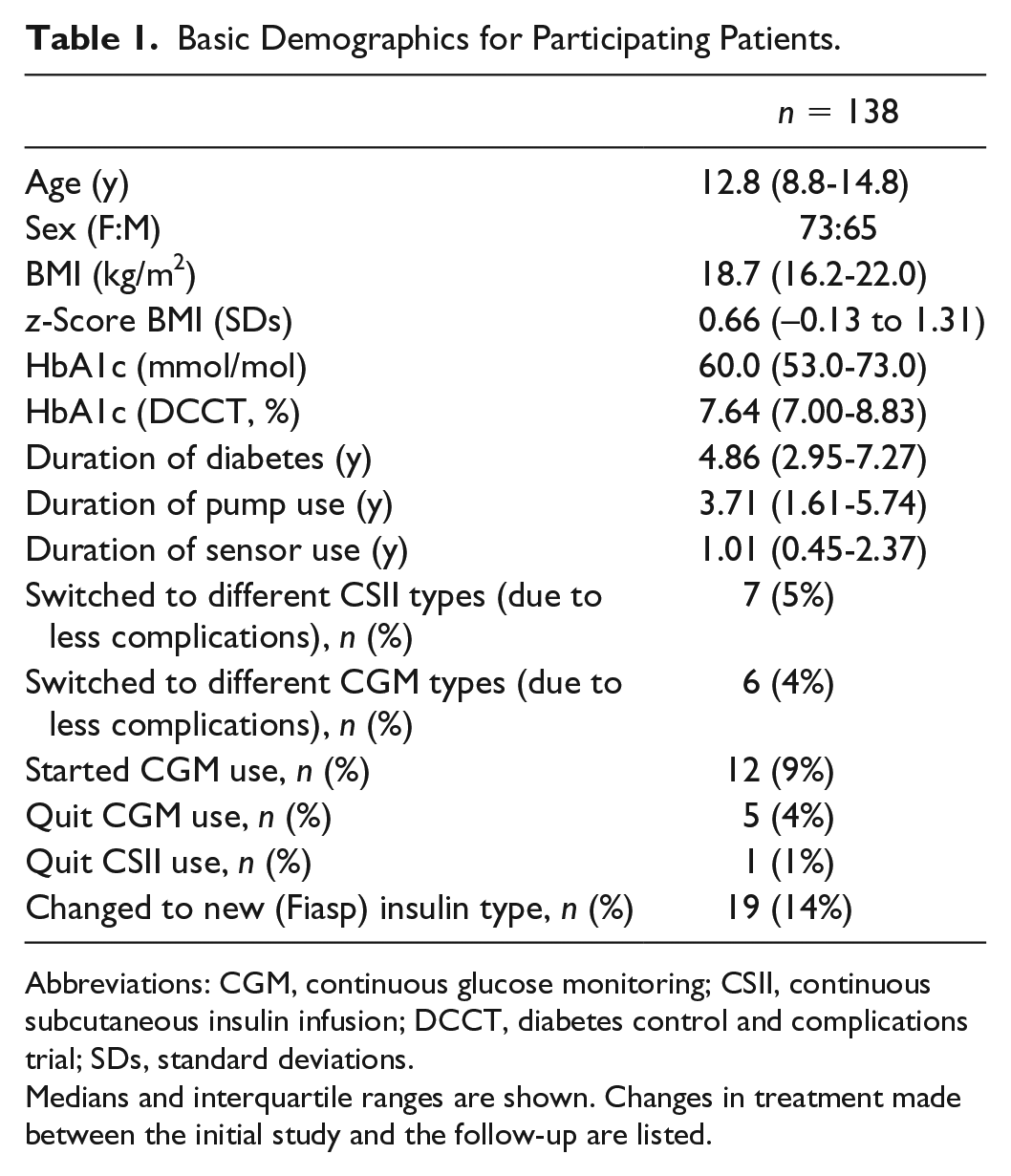
Abbreviations: CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DCCT, diabetes control and complications trial; SDs, standard deviations.
Medians and interquartile ranges are shown. Changes in treatment made between the initial study and the follow-up are listed.
Comparison of CSII and CGM From the Initial to Follow-up Study
In total, 136 patients were currently using CSII and 93 patients were using CGM (two patients used CGM only). For five months since the initial trial, itching was the most frequent complication and was reported by 78.7% of CSII users and 60.2% of CGM users. Other frequent complaints during this period were dry wounds (CSII: 37.5% and CGM: 26.9%) and eczema (CSII: 30.9% and CGM: 40.9%).
Patients who were treated with CSII and/or CGM during both trials were compared and the results of comparing current dermatological complications to CSII and CGM are shown in Figure 2. There were no significant differences between the two time periods of answering the questionnaires (P > .05) for neither CSII nor CGM. We investigated whether the same patients were experiencing any dermatologial complications to either CSII or CGM in both the initial study and the follow-up (Figure 1). Followup showed that 88 patients (64%) out of 138 were currently experiencing dermatological complications and 71 (81%) of those also had complications during the initial study. For symptoms of itching, results showed that 114 patients (82%) were currently experiencing itching and 89 (78%) of these 114 patients also had itching during the initial study. These results showed that if patients experienced itching or dermatological complications initially, the complications were likely to persist (P < .0001 for both).
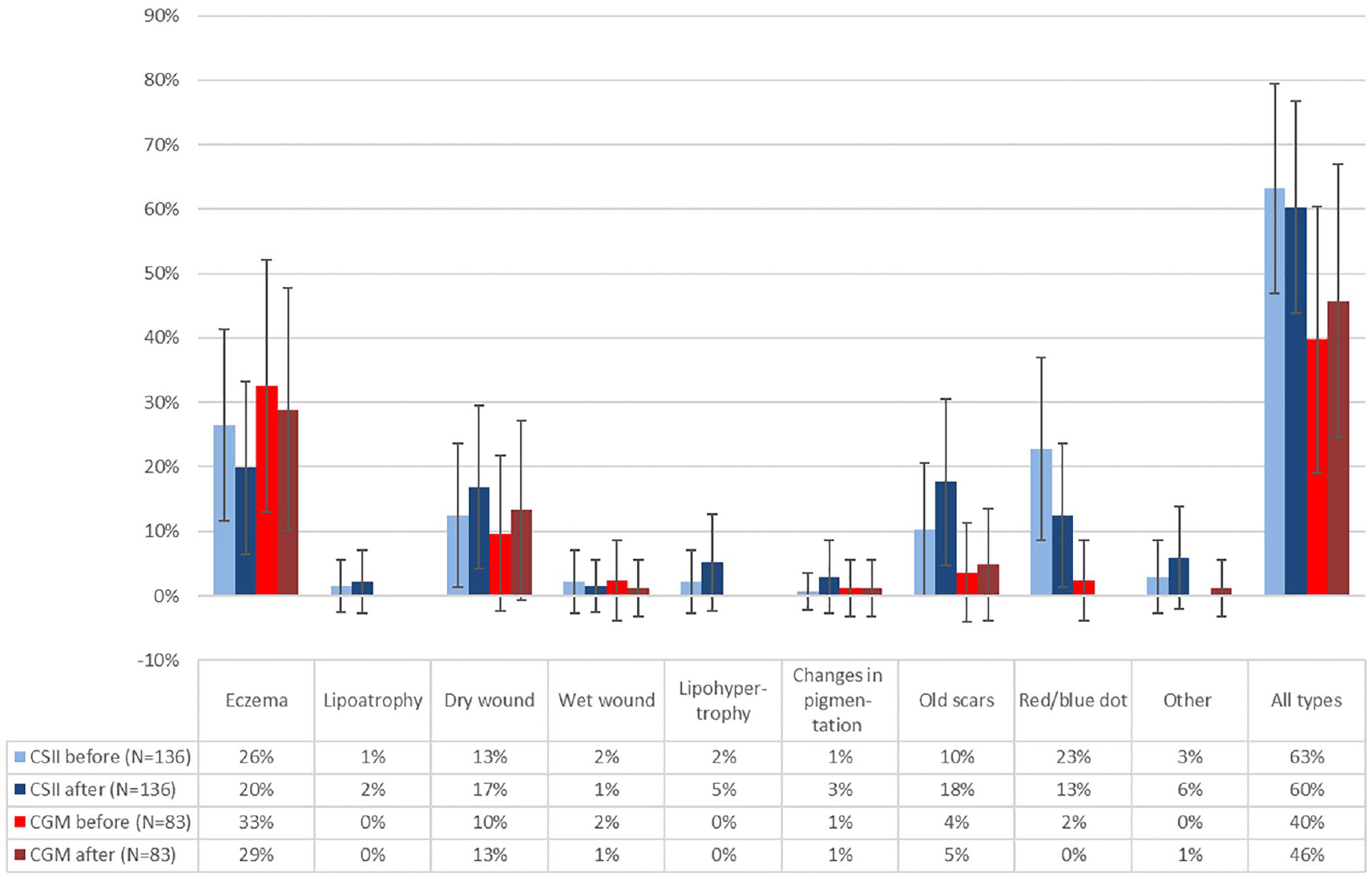
Comparison of the current dermatological complications in the same continuous subcutaneous insulin infusion and continuous glucose monitoring patients from the initial to follow-up study with 95% confidence intervals.
In the follow-up questionnaires, patients also rated their discomfort caused by any current dermatological complications. The patients rated their discomfort to dermatological complications related to CGM as being between a “big problem” to “very big problem” (mean: 3.26), whereas CSII complications were rated as a “moderate problem” to “big problem” (mean: 2.63).
Between the initial and follow-up periods, 17 patients (12%) had been using a type of barrier cream (eg, Secura No-Sting Barrier Film and Brava Skin Barrier Wipe) and 79% of these patients still had dermatological complications five months later. In total, 17 patients (10 for CSII and 15 for CGM) were using patches (an extra adhesive under the CGM or CSII to avoid dermatological reactions to the device, eg, Duoderm and Tegaderm) and an association between having all current skin problems during the initial study and developing eczema five months later was found (odds ratio [OR]: 5.6).
No significant associations were found with any seasons: summer, spring, fall, and winter (P > .05 for all).
CSII/CGM Type and Duration Compared With Skin Problems and Itching
Analyzing for CSII and all current skin problems, specific current skin problems, and itching, neither sex, BMI, z-score BMI, HbA1c levels, nor CSII type showed any significant differences (P > .05). Age had a preventive effect on specific current skin problems (OR: 0.898; P < .05), whereas duration of diabetes and treatment with CSII had a worsening effect on itching (OR: 1.2; P < .05 for both). The pump-duration category one to four years was associated with a higher incidence of dermatological complications including both all current skin problems (P < .001), specific current skin problems (P < .05), and itching (P < .05).
When analyzing CGM types, patients were either using Enlite, Libre, or Dexcom. Dexcom was excluded from the analysis due to the lack of power with only six patients using Dexcom. Among 94 patients who were currently using CGM, 75% of the patients using Enlite were experiencing itching in response to CGM treatment vs 41% of the patients on Libre. In total, 58% of the patients on Enlite had the current dermatological complications against 21% using Libre. No significance in age, sex, BMI, z-score BMI, HbA1c levels, or diabetes duration was found (P > .05). The duration of CGM treatment was found to be associated with the risk of itching (OR: 1.75; P < .05), although the duration of CGM treatment depends on CGM type (P < .05). Simple logistic regression analysis showed that patients using Enlite compared to Libre experienced more dermatological complications in connection with both all current skin problems and specific current skin problems (OR: 5.4; P < .001 and OR: 6.1; P < .001, respectively). When using multivariate logistic regression to disregard the impact of CGM duration, association was still present (OR: 5.1; P < .05 and OR: 5.4; P < .05). No association between CGM type and itching was found after multivariate analysis (P > .05).
Health-Related Quality of Life
A total of 111 children/adolescents and their parents were analyzed separately for each dimension (Table 2). The HrQoL scores ranged from 0 to 100. The separate scores for every subscale were for all subscales in the range 53.4 to 77.7. The subscale “treatment” from the DSM-10 had the lowest mean score of 55.0 (95% CI: 54.3-55.9). These results were observed for both children and parents, regardless of whether the current skin complications were specific or not. For both children and parents, when each dimension was compared separately with all current dermatological complications, specific current dermatological complications, and itching, it showed no significant differences for either CSII or CGM (P > .05 for all), even when adjusted for age, sex, and HbA1c.
Mean Scores for All DISABKIDS Dimensions With Confidence Intervals.
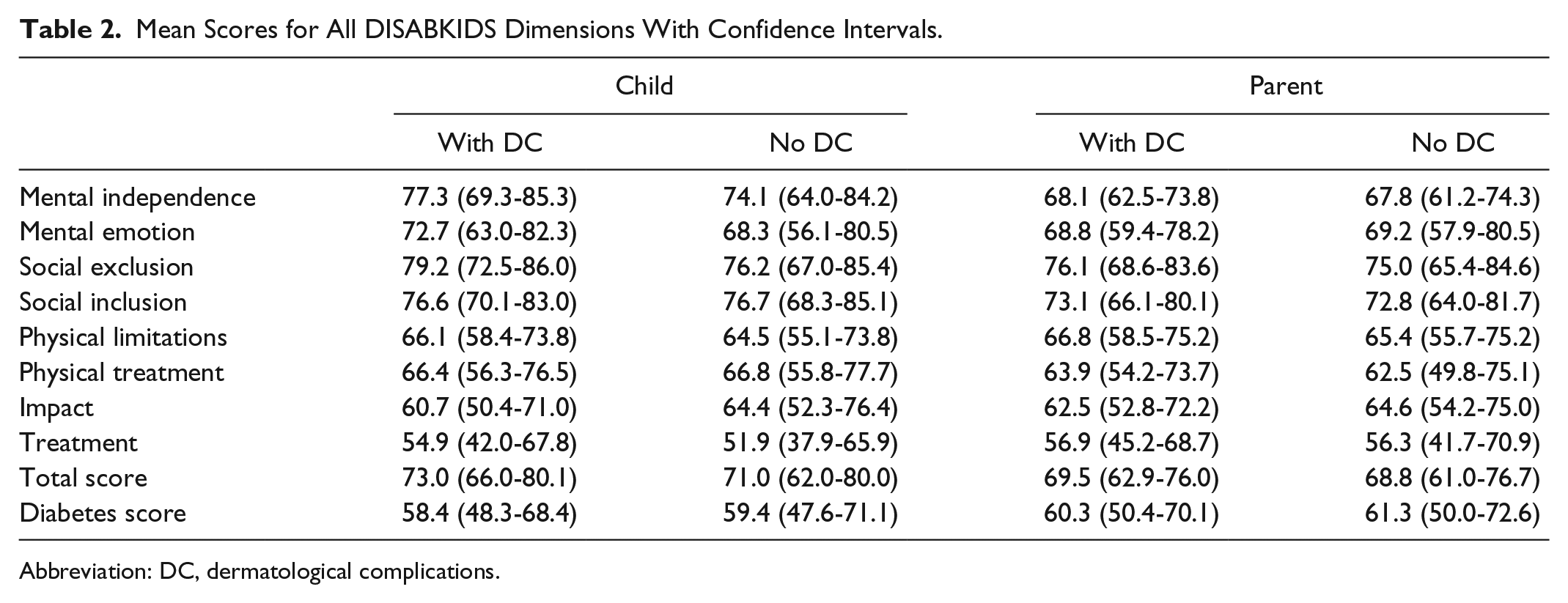
Abbreviation: DC, dermatological complications.
Patients who rated the dermatological complications as a huge discomfort also scored lower in HrQoL, with negative Spearman correlations for each subscale. The treatment subscale in the DSM-10 had a Spearman correlation of −0.45. Table 1 shows that six patients discontinued (five CGM and one CSII) device treatment. Among these, 50% reported a discomfort score of “moderate problem” to “big problem” during the initial survey. Analyses of each of the subscales revealed that parents rated the children’s HrQoL as lower than did the children themselves (P < .0001). The total mean generic scores were 69.1 for parents and 72.0 for children.
Discussion
After five months, our results showed similar prevalence of dermatological complications when patients with T1D use modern technology for insulin delivering and glucose measuring. This demonstrates and emphasizes that it is still a continuous and concerning problem in the same population.
Our results showed that 81% of the affected patients in the initial study also had dermatological complications at follow-up. The results also showed an association with having all current dermatological complications in the initial study and developing eczema five months later, when using patches. This is probably not due to the patches causing eczema, but instead because patients with more severe dermatological complications use patches. Furthermore, some patches are not suitable to prevent allergic contact dermatitis, since the barrier created is still too thin, and some patches might cause irritation themselves. Regarding the use of barrier cream, 79% of these patients still experienced dermatological complications after follow-up. This could be due to the fact that barrier creams are only able to strengthen skin barrier for up to three days. Thus, this does not help with the CGM which is attached on to the skin for 6 to 14 days. This indicates that once dermatological complications start to appear, they become chronic even though some patients had been using the current preventive and available treatment in between the two periods indicating that the treatments currently available are inadequate.
Depending on the definition of dermatological complications, the previous studies have shown up to 94% of patients experience dermatological complications related to CSII.3,9 In this study, relatively few patients changed treatments during observation period. Therefore, there was insufficient power to determine the effect of these changes on dermatological complications. Among87 patients who had dermatological complications in the initial study, only 16 (18%) reported relief from their complications and 17 (33%) were developing dermatological complications at follow-up. These problems are becoming more widely recognized. However, there exist only few suggestions for treatment and prevention. 13
At five months, our patients still regarded the impact of CGM complications as worse than those due to CSII. This may be because CSII systems are replaced more frequently than CGM systems. The CSII have to be relocated to a new site every two to three days on average, whereas the CGM needs to be changed every 6 to 14 days. 14 The patients in our study followed these guidelines for the most part. Nonetheless, very few patients stopped using CGM or CSII, indicating that the benefits of using the technology outweigh the negative impact of dermatological complications.
We confirmed that there is a relationship between duration of CSII treatment and dermatological complications. This was also confirmed by another recent study. 15 Longer duration had little effect on overall prevalence but was strongly associated with itching. Similar results were observed for CGM where dermatological complications to Libre (21%) have emerged in contrast to none at our initial survey though still with a significantly lower prevalence than for the Enlite sensor (58%). During the initial study, no patient had been using Libre for more than one year. At follow-up, all the dermatological complications and itching appeared in patients who had been using the Libre system for the longest durations. Therefore, we would expect an increase in dermatological complications among Libre users in the future, unless effective preventive treatments can be established.
Although the overall total generic score was 70.6, we demonstrated lower HrQoL in patients reporting high discomfort scores, indicating that HrQoL could be used as a screening tool to identify patients who are most stressed. DISABKIDS has proven capable of assessing quality of life in the other studies.10,11 In general, the patients and their parents had HrQoL scores above 60 for all dimensions except for treatment in the DSM-10. Health-related quality of life and dermatological complications in CSII and CGM users have not been studied previously, but lower quality of life in children with eczema caused by other factors has been observed. 16
Results from a follow-up study on adults with dermatological complications due to CSII and/or CGM also showed an association between high Problem Areas in Diabetes (PAID) scores (ie, worse HrQoL) and more dermatological complications. 17 Perhaps, other patient reported outcome (PRO) measures, such as PAID-T (Problem Areas in Diabetes - Teenagers) or Diabetes Distress, could be used to identify differences in patients with or without dermatological complications. Problem Areas in Diabetes - Teenagers focuses on treatment challenges, whereas Diabetes Distress focuses on stress. In contrast, DISABKIDS measures social, mental, and physical well-being. Dermatological complications in relation to CSII and/or CGM also have a high incidence in adults with T1D. 18 Associations among high PAID scores, increased HbA1c levels, and depression symptoms have been demonstrated.19-21
Many parents rate their children as having lower health and higher burden of diabetes than the children themselves. 22 A recent Danish study on children with T1D also found that parents rated their children’s HrQoL as lower than did the children themselves. Our study showed a similar tendency.
The total HrQoL score for our population (70.6) was lower than that in this recent Danish study (73.6) and studies from Sweden and Norway. 23 The total score may not be directly comparable because DISABKIDS consists of several subscales and probably country-specific adjustments are needed.
The main strength of our study was the high completion rate for both questionnaires, probably because these were easy to complete at home in a short time. Thus, no following examination by a physician was made and the collected data represented was therefore subjective. The classification of dermatological complications was therefore a minor challenge, since it was based on the classification used in the questionnaire. The questionnaire on dermatological complications did not distinguish between child and parent reporting. However, when assessing HrQoL using DISABKIDS, this factor was taken into account. For the patients aged from two to seven years, the parents were used as proxies. Since this is a follow-up study on a cross-sectional study, we cannot infer direct causality, but can only describe associations.
Our findings highlight the importance of more research and better communication between medical practitioners and the medical industry. Studies have shown that the adhesives used to attach CSII and CGM to the body, more specifically the adhesives using acrylates, are a major cause of the allergic reactions observed.24,25 Isobornyl acrylate (IBOA) has been identified as a major allergen in FreeStyle Libre and Enlite sensors and is associated with dermatological complications such as severe eczema and itchiness, causing patients to discontinue treatment with CGM.26,27 Silicone-based adhesives are suggested as a better option. 14 Other case studies have shown that hydrocolloid blister plaster or film-forming agents might have a preventive effect on recurrence of allergic contact dermatitis. These adhesives might be a better option for some patients until CGMs without IBOA are available.28,29 Many medical manufacturers continue to use acrylates in their adhesives today, either because they are unaware of this problem or the costs. By the beginning of 2018, the Danish Medicines Agency had collected only 15 reports of dermatological complications to CSII and/or CGM in total, although our follow-up has shown a remarkable higher incidence. Therefore, medical practitioners and patients need to be encouraged to report these side effects more often. Patients with known allergic contact dermatitis should be informed and encouraged to use an adhesive without IBOA.
Conclusion
In conclusion, we found no improvement in dermatological complications to CSII and CGM over a five-month period. This suggests that better preventive care and new treatment modalities are needed since greater levels of discomfort are associated with lower quality of life and an increased disease burden.
Footnotes
Acknowledgements
The authors thank all the children/adolescents and their parents who participated in this study for taking their time to complete the questionnaires and for making this research possible.. Proofreading was done by Bobby Brown.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.T. Weng, C. Zachariae, and K. B. Christensen have nothing to declare. J. Svensson serves as an adviser to Medtronic, Janssen, and Novo Nordisk. She owns shares in Novo Nordisk, and has received fees for speaking on behalf of Medtronic, Sanofi, Novo Nordisk, and Bayer AG. A.K. Berg has received fees for speaking at symposia from Novo Nordisk.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Aage Bangs fund, Aase and Ejnar Danielsen’s fund, Else and Mogens Wedell-Wedellsborg’s fund, and Conva-tec Unomedical.
