Abstract

Studies have shown that collaborative parent-child relationships can positively influence both regimen adherence and glycemic control, 1 and that regimen adherence is improved by parental involvement 2 and monitoring of their child’s diabetes-related tasks. 3 We hypothesized that immediate access to diabetes-related data and the ability to review these data retrospectively may increase feelings of confidence/safety in diabetes management and reduce hypoglycemia fear among parents of children/adolescents with type 1 diabetes (T1D).
The Accu-Chek® Connect System (Roche Diabetes Care, Indianapolis, USA) consists of a blood glucose meter, smartphone application (app) and an online data management web portal. The meter connects wirelessly, via low-energy Bluetooth, to the user’s smartphone app, which provides multiple functions to facilitate diabetes management. The system allows users to share real-time glucose data and related information with others through a personal web portal and/or text message.
In this 1-week, observational, mono-centric pilot study, we assessed perceptions of parents of children/adolescents (age 0-17 years) with type 1 diabetes and older children/adolescents (age 8-17 years) who used the system while attending the Friends for Life Conference, held July 6-12, 2015, at Disney’s Coronado Springs Resort, Florida. Participants received the system blood glucose meter, testing supplies, and a smartphone (if needed) and underwent a 15-minute training session on system use. Participants were asked to use the system for 3 or more days.
Pre and post questionnaires were used to assess system use and impact on perceptions of confidence, safety, hypoglycemia fear, system utility and preference to current diabetes management method. A total of 91 parents of T1D children/adolescents and 79 T1D children/adolescents are included in this analysis.
A large majority of parents and children/adolescents reported frequent use of the meter (83.5% and 96.2%, respectively) and smartphone app (65.9% and 87.3%, respectively). The majority of respondents “mostly agreed” that they felt more confident, safer and less worry about hypoglycemia when using the system (Figure 1). Comparison of continuous glucose monitoring (CGM) versus no CGM use showed notable differences in the percentage of parents who reported feelings of increased safety (68.4% vs 80.0%, P = .4437, respectively) and less fear of hypoglycemia (61.4% vs 83.3%, respectively, P = .0521).
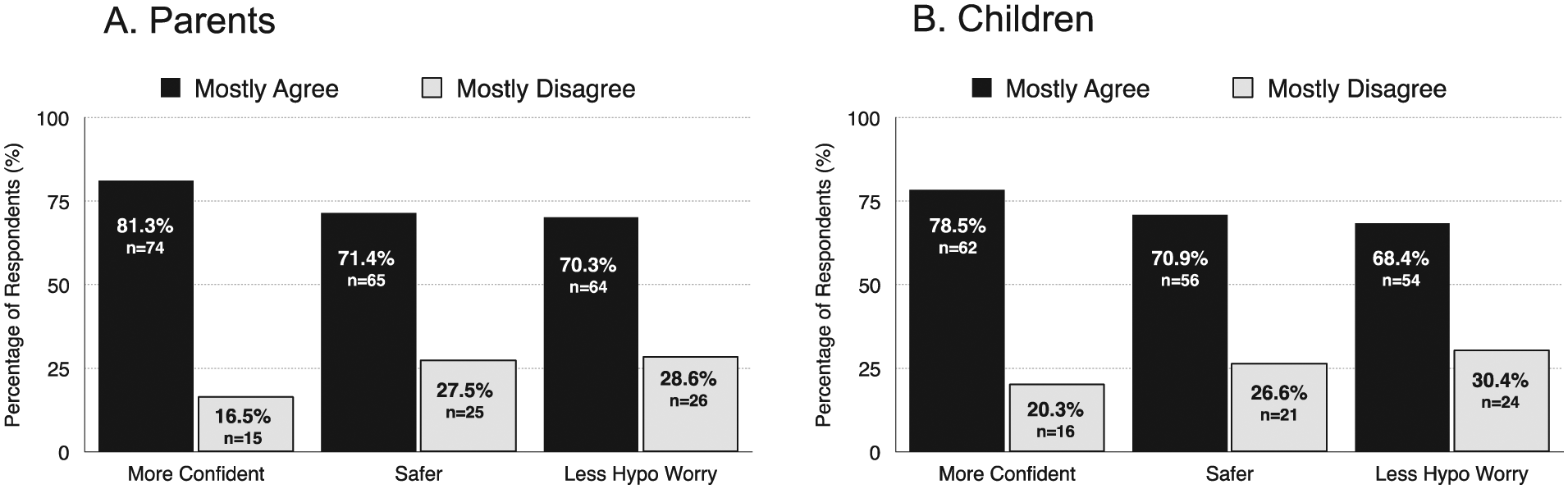
Reported increased confidence, increased safety, and less hypoglycemia fear.
Most parent (73.6%) and children/adolescent (64.6%) respondents reported a preference for using the system over their prior diabetes management method. A notable difference was seen in preference for using the system over previous method between parents of multiple daily insulin injections (MDI)- versus continuous subcutaneous insulin infusion (CSII)-treated children (84.0% vs 69.7%, respectively, P = .1941). Similar perceptions were reported by children/adolescent CGM versus no CGM users: increased safety (62.5% vs 82.1%, P = .0632, respectively) and less fear of hypoglycemia (60.4% vs 78.6%, respectively, P = .0745). Most parents (90.1%) and children/adolescents (93.7%) reported satisfaction with how the system helped them manage diabetes. Most respondents (86.8% parents, 86.1% children/adolescents) reported overall satisfaction with system.
Our study demonstrates that ease of use, combined with accurate, real-time access to glucose data in a device that is easy to learn and adopt can facilitate daily diabetes care, reduce patient and parent anxiety and fear, and support adherence to good self-management practices.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin; MDI, multiple daily insulin injections; SD, standard deviation; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GP has received research funding and consulting fees from Roche Diabetes Care. ML and CGP have received consulting fees from Roche Diagnostics. CR is an employee of Roche Diabetes Care.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
