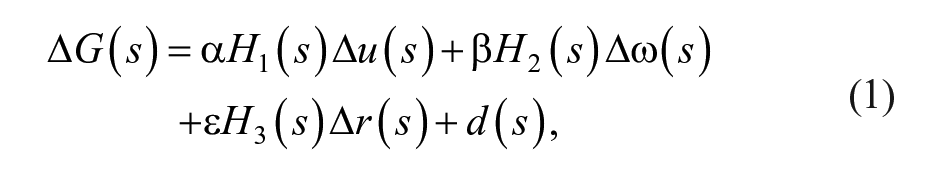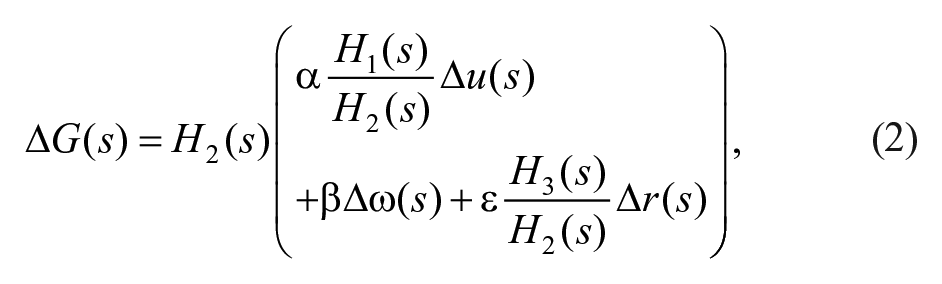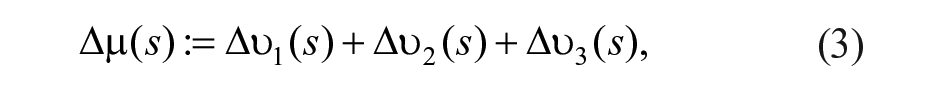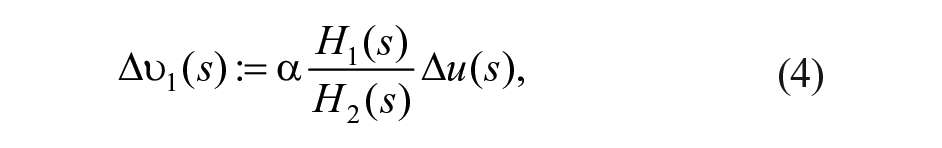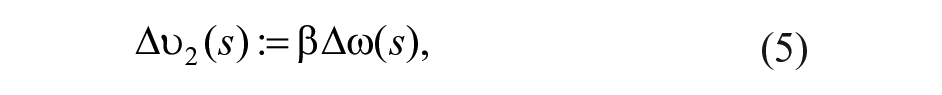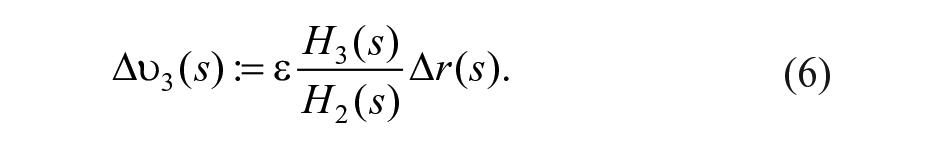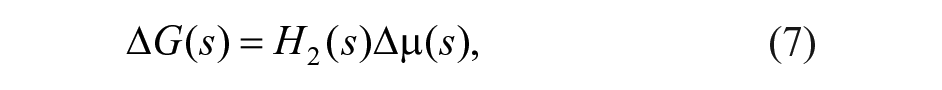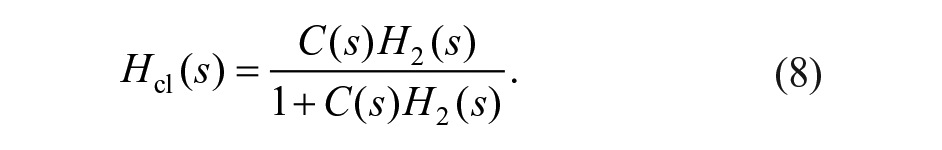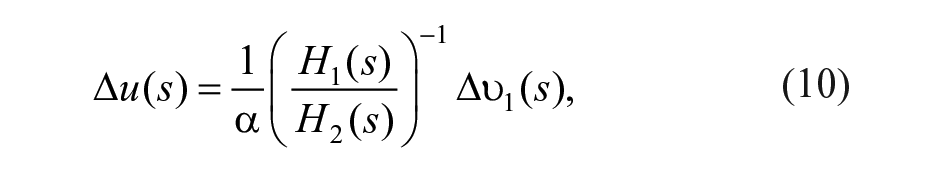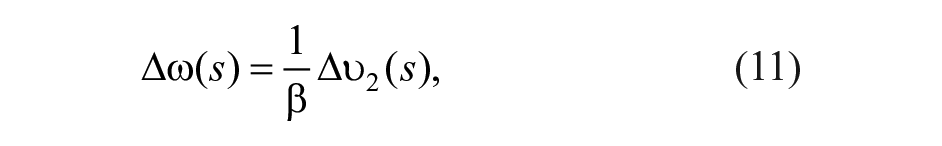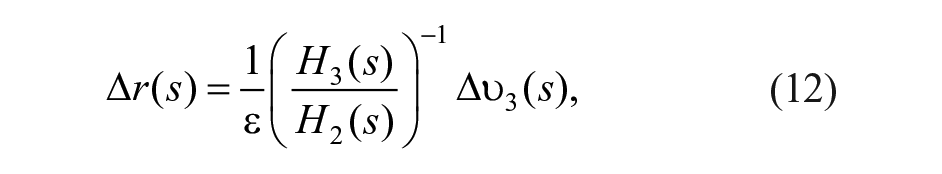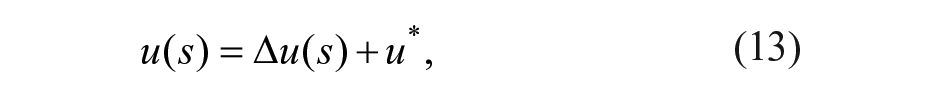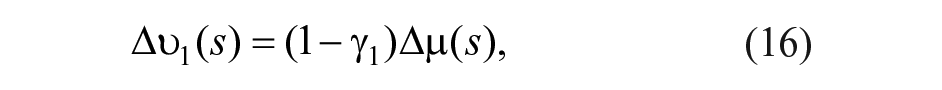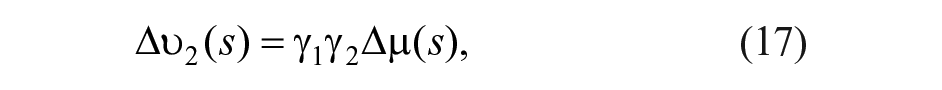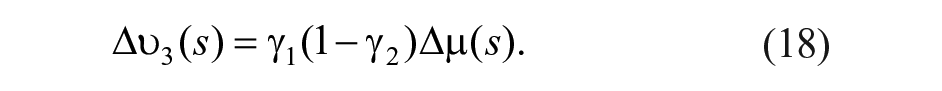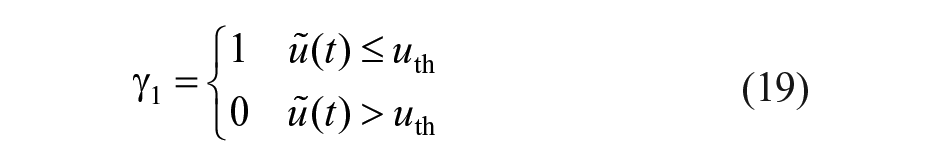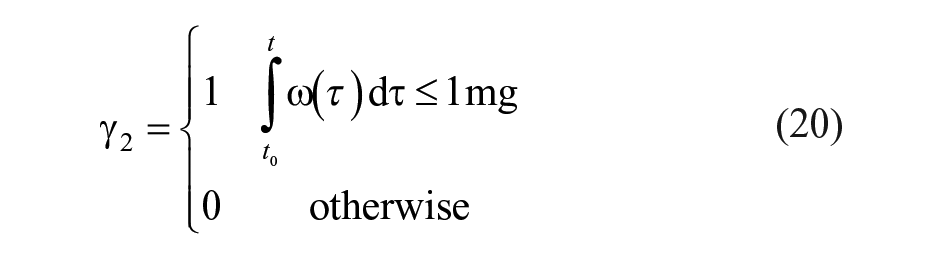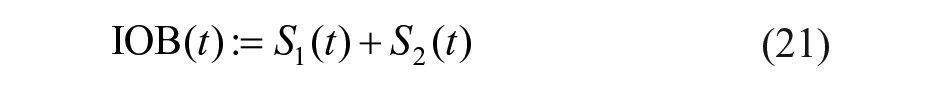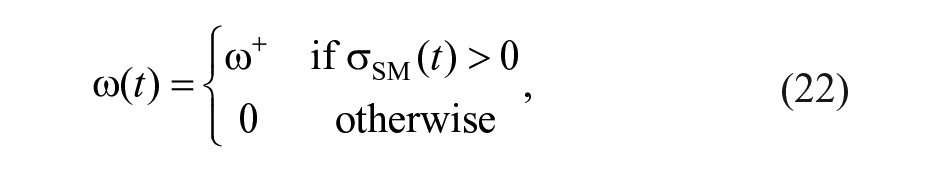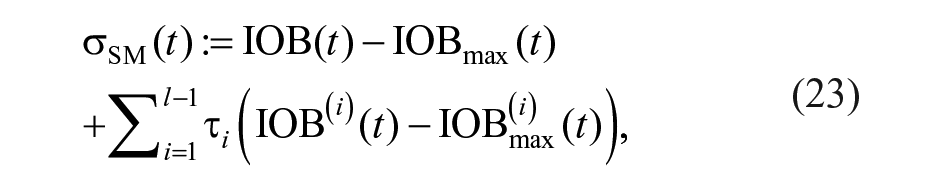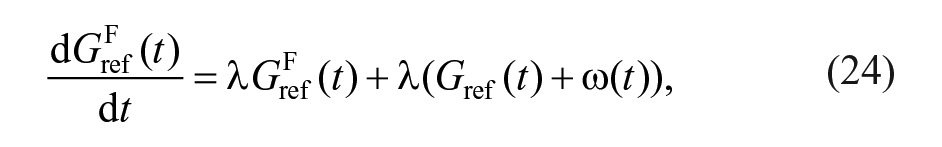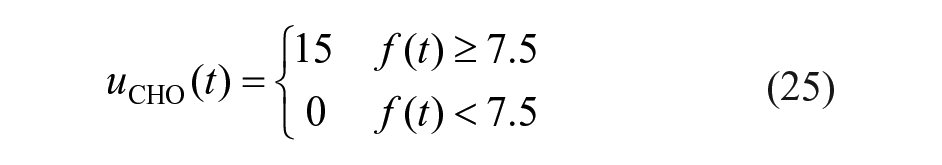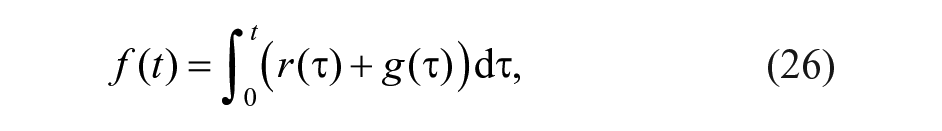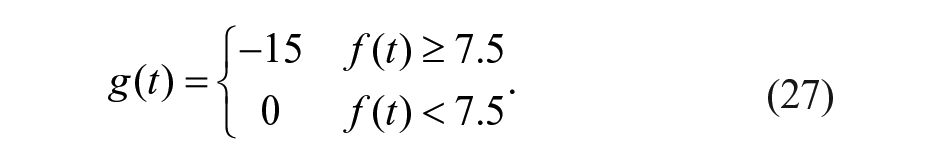Abstract
Background:
An artificial pancreas with insulin and glucagon delivery has the potential to reduce the risk of hypo- and hyperglycemia in people with type 1 diabetes. However, a maximum dose of glucagon of 1 mg/d is recommended, potentially still requiring rescue carbohydrates in some situations. This work presents a parallel control structure with intrinsic insulin, glucagon, and rescue carbohydrates coordination to overcome glucagon limitations when needed.
Methods:
The coordinated controller that combines insulin, glucagon, and rescue carbohydrate suggestions (DH-CC-CHO) was compared with the insulin and glucagon delivery coordinated controller (DH-CC). The impact of carbohydrate quantization for practical delivery was also assessed. An in silico study using the UVA-Padova simulator, extended to include exercise and various sources of variability, was performed.
Results:
DH-CC and DH-CC-CHO performed similarly with regard to mean glucose (126.25 [123.43; 130.73] vs 127.92 [123.99; 132.97] mg/dL, P = .088), time in range (93.04 [90.00; 95.92] vs 92.91 [90.05; 95.75]%, P = .508), time above 180 mg/dL (4.94 [2.72; 7.53] vs 4.99 [2.93; 7.24]%, P = .966), time below 70 mg/dL (0.61 [0.09; 1.75] vs 0.96 [0.23; 2.17]%, P = .1364), insulin delivery (43.50 [38.68; 51.75] vs 42.86 [38.58; 51.36] U/d, P = .383), and glucagon delivery (0.75 [0.40; 1.83] vs 0.76 [0.43; 0.99] mg/d, P = .407). Time below 54 mg/dL was different (0.00 [0.00; 0.05] vs 0.00 [0.00; 0.16]%, P = .036), although non-clinically significant. This was due to the carbs quantization effect in a specific patient, as no statistical difference was found when carbs were not quantized (0.00 [0.00; 0.05] vs 0.00 [0.00; 0.00]%, P = .265).
Conclusions:
The new strategy of automatic rescue carbohydrates suggestion in coordination with insulin and glucagon delivery to overcome constraints on daily glucagon delivery was successfully evaluated in an in silico proof of concept.
Keywords
Introduction
An artificial pancreas (AP) is an automated insulin delivery system aiming at the improvement of glucose control in people with type 1 diabetes (T1D). 1 A single-hormone AP (SHAP) consists of a continuous glucose monitor, an insulin pump, and a control algorithm that modulates insulin infusion quasi-continuously. Dual-hormone AP (DHAP) systems also introduce glucagon infusion as control action to compensate the unidirectional effect of insulin on glucose. Recent developments in stable soluble glucagon formulations 2 are paving the way to such systems although no commercial formulation is yet available. However, long-term safety of glucagon delivery is unknown.
Since the demonstration of feasibility of DHAP systems in humans, 3 several studies have targeted head-to-head comparisons between SHAP and DHAP systems.4-10 A recent review of results 11 concluded that during nocturnal period SHAP was enough for a good glucose control while DHAP proved superior performance in reduction of hypoglycemia overall and during exercise; benefits in postprandial control, reduction of severe hypoglycemia and mean glucose are unclear. In the four-arm four-day outpatient study with three moderate-intensity aerobic exercise sessions by Castle et al 5 DHAP achieved lower time in hypoglycemia during exercise compared to SHAP and predictive-low-glucose-suspend, and similar to standard care where pre-exercise insulin adjustments were allowed. However, despite the use of glucagon and wearables to detect exercise, hypoglycemia was still present (1.3% [1.0] overall and 3.4% [4.5] during exercise; mean [SD]).
Current DHAP systems are based on an insulin controller and a glucagon controller which is activated in certain circumstances in order to initiate the counterregulatory action. These independent control loops may create unwanted interactions among hormones delivery reducing effectiveness. Besides, an excess of plasma insulin has been found to reduce effectiveness of glucagon microboluses by El Youssef et al 12 which does not support the design of DHAP systems with aggressive insulin infusion considering the availability of glucagon to compensate the increased risk of hypoglycemia. Indeed, physiologically there is a coordination between insulin and glucagon secretion. 13 On the one hand, an increment in plasma insulin levels produces a suppression of glucagon secretion in patients with T1D; and a decrement in insulin levels together with low plasma glucose concentration stimulates glucagon secretion. 14 On the other hand, alpha cells anticipate the possible hyperglycemic rebounds due to glucagon secretion by means of beta cell sensitization. 15
Motivated by this paracrine communication, control algorithms incorporating coordinated insulin and glucagon delivery have been investigated. In Herrero et al 16 potentiation of insulin by glucagon was incorporated into the Imperial College AP system, reporting in silico a reduction in hyperglycemia without increased hypoglycemia. In Bondia et al17,18 a control algorithm with intrinsic coordination based on a collaborative parallel control formulation was first introduced. Thorough in silico evaluation showed the benefit of coordination with lower glucagon delivery, although room for improvement under exercise was identified. 19 Further refinements incorporating sliding mode reference conditioning (SMRC) techniques for insulin-on-board (IOB) limitation were carried out in Moscardó 20 (see section 7.6). When compared to the original algorithm, 19 the refined controller showed improvements in percentage of time in target (92.98% [3.24] vs 91.56% [3.42]) and time in hypoglycemia (1.45% [2.01] vs 3.40% [2.92]) in a two-week scenario with daily 60-minute exercise sessions. Nevertheless, some few patients required glucagon delivery higher than 1 mg/d (0.75 mg/d [0.40; 1.83]; median [25-75 percentiles]).
In this work, parallel control structure (dual-hormone coordinated control; DH-CC) is further exploited to compensate such excess of glucagon need in some patients with the integration of rescue carbohydrate as an alternative control route (dual-hormone coordinated control with carbohydrates; DH-CC-CHO). Parallel control computes a virtual control action (control effort) that later is distributed into the different control actions to get a combined effect equal to the needed control effort. This gives rise to a flexible control structure where different configurations combining insulin, automatic rescue carbs suggestions, and glucagon can be designed by reconfiguring the distribution logics. DH-CC and DH-CC-CHO are compared to analyze the benefits of this new proposal during the challenging exercise scenario in Moscardó et al.19,20
Methods
Coordinated Parallel Control DHAP Including Rescue Carbohydrates
Coordinated control techniques for multiple-input-single-output systems have been developed in different ways in literature. 21 Of interest is the concept of habituating control,22,23 where control actions are classified as “slow and cheap” (primary) and “fast and expensive” (secondary). In a DHAP, the “fast and expensive” control action can be associated to glucagon, with a faster subcutaneous PK/PD, although its delivery must be restricted to 1 mg/d due to the possible side effects such as nausea, vomiting, and headache 24 ; instead, the “slow and cheap” action is associated to insulin. Additional inputs can also overcome saturation problems of the primary action contributing to the additional control effort not able to be provided by the latter.
These concepts can be cast into a parallel control structure where a main controller computes the needed control effort (virtual control action), which is later distributed by a divisor among the available control actions, giving rise to intrinsic coordination. 25 This technique was applied to derive a DHAP coordinating insulin and glucagon delivery19,20 (see diagram in black in Figure 1). In this controller, the divisor was designed so that glucagon acts as a secondary control action when insulin delivery is below a given threshold (set in this case to 75% of basal insulin infusion) for hypoglycemia mitigation.
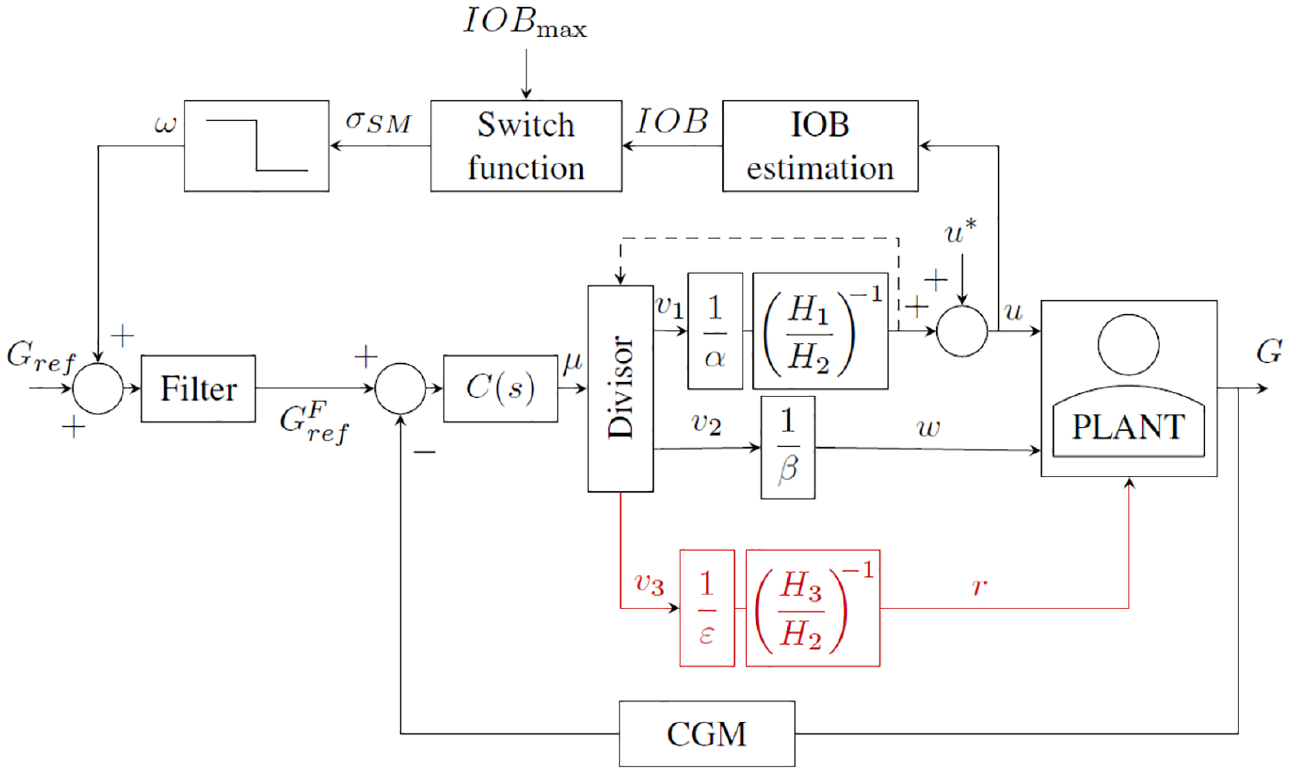
Block diagram of the proposed coordinated parallel control DHAP including rescue carbohydrates. An external SMRC loop is also considered for insulin-on-board limitation which modulates glucose target. DHAP, dual-hormone artificial pancreas; SMRC, sliding mode reference conditioning.
However, in some occasions the delivery of glucagon is not enough to prevent hypoglycemia, as revealed by the need of rescue carbohydrates in DHAP clinical studies, or an overdelivery of glucagon beyond the limit of 1 mg/d may result. In these cases, integration of automatic suggestion of rescue carbohydrates into a DHAP can overcome these limitations. To this end, a third control action can be incorporated into the above-described parallel control strategy (branch highlighted in red in Figure 1), considering ideal administration of rescue carbohydrate, with a posteriori quantization for practical dosing by the patient. In this work, constraint on the maximum delivery of glucagon per day is addressed through the replacement of glucagon as a secondary action by rescue carbohydrate when the accumulated glucagon in a given 24-hour time window is greater than the total daily dose restriction imposed.
In the following the proposed controller is described. Consider the system linearization
where ∆G(s) is the deviation of plasma glucose concentration from the equilibrium value G*; ∆u(s) is the deviation of insulin infusion from its equilibrium value u*; ∆ω(s) is the deviation of glucagon infusion from the equilibrium value ω*, which is null; ∆r(s) is the fast-acting rescue carbohydrate intake; and d(s) is a disturbance (eg, glycemic effect of meal and exercise). Transfer functions
In the absence of disturbance, the plant with faster dynamics, ie, H2(s), is factorized as follows:
leading to the following definition of the control effort
Note that now equation (2) can be expressed as a single-input-single-output system in terms of the new virtual control action:
where no saturation constraints apply to
The controller
where
A key element is the design of the divisor that distributes the virtual control action
where
Counterregulatory actions are designed to be delivered only when insulin infusion is below a certain threshold,
where
The switching due to equations (19) and (20) does not affect the closed-loop transfer function. Therefore, the closed-loop transfer function remains unaltered and it will be stable by design conditions on
An SMRC loop26,27 is also considered for the limitation of IOB. Note that this does not modify the stability of the closed-loop system since it acts on the glucose reference. Here, IOB is represented by subcutaneous insulin compartments in Hovorka model,
28
Given the system in Figure 1 and an upper limit of IOB,
where
where
Rescue Carbohydrate Quantization
Rescue carbohydrate control action is quantized in rescue events of 15 g for practical administration by the patient. The event will be triggered only when an accumulated rescue carbohydrate action over half this dose is required and will not be triggered again unless the dose administered in excess was required, repeating the strategy.
The quantized rescue carbohydrate control action,
where
A value of 7.5 g in equation (25) was used similarly to Beneyto et al 29 although other values could be considered.
Controllers Tuning
Table 1 shows the values of the parameters for the DHAP without and with rescue carbohydrates. As said before, DH-CC-CHO is an improvement of DH-CC structure by means of the addition of a third loop. Thus, both structures share the tuning of the master controller parameters, which was manually tuned to achieve the best possible glycemic outcomes (ie, percentage of time in range [70, 180] mg/dL and percentage of time below target).
Controller Parameters.
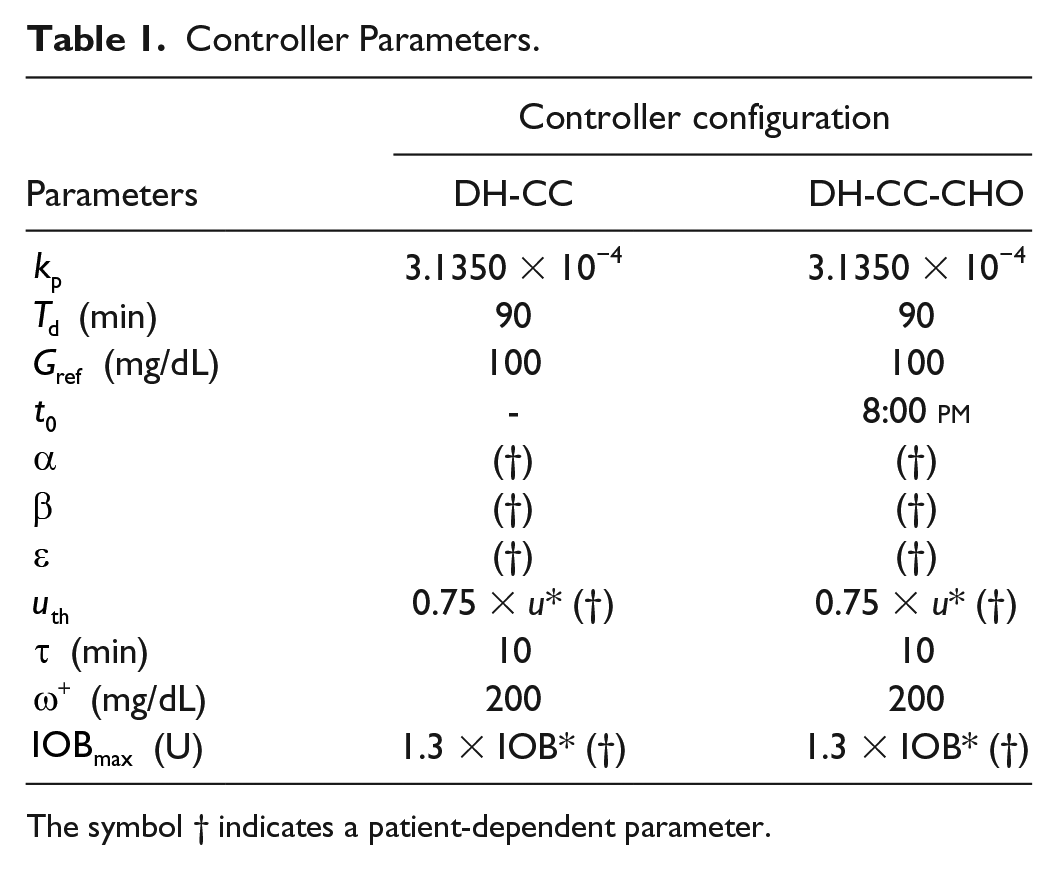
The symbol † indicates a patient-dependent parameter.
For all the evaluated subjects, parameters were fixed to the same value except parameters
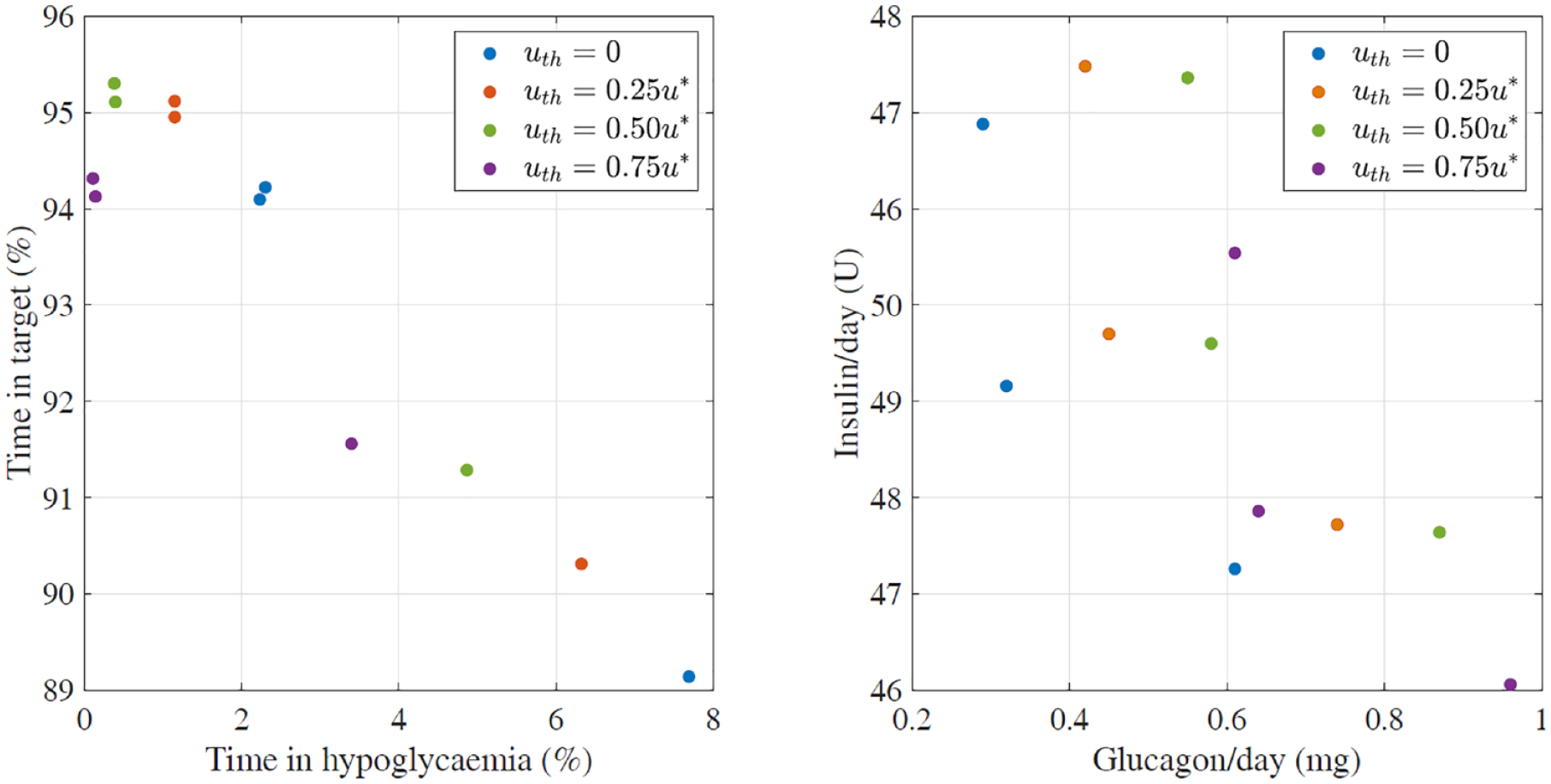
Effect of the threshold value
In Silico Evaluations
The educational version of the UVA/Padova simulator 30 with the addition of the exercise model in Schiavon et al 31 and intra-day and intra-subject variabilities were used to assess and compare the above control structures.
Intra-day variability was incorporated into the simulator by modifying some of the parameters: meal variability was emulated by introducing meal-size variability (CV = 10%), meal-time variability (STD = 20 minutes), and uncertainty in the carbohydrate estimation (uniform distribution between −30% and +40%). Variability of meal absorption rate
To emulate intra-subject variability, insulin absorption model parameters
The scenario considered meal and exercise. The selected daily pattern of carbohydrate doses was 7:00
A two-week scenario duration was used to compare DH-CC with DH-CC-CHO configurations in 100 adults. The subjects are based on the 10 adults that are available in the educational version of the UVA/Padova simulator, with 10 repetitions each getting different instances of variability. Moreover, the chosen basal insulin infusion rates,
Data Analysis
In order to carry out the comparison between both proposed control structures, the standard glycemic control metrics 32 were used: mean blood glucose (MG); percentage time in target range [70,180] mg/dL (TIR); percentage time below target (<70 mg/dL and <54 mg/dL); percentage time above target (>180 mg/dL); daily average of insulin delivered in units of insulin (INS); and daily average of glucagon delivered in mg (GGON). Statistical differences were assessed by the non-parametric Wilcoxon signed-rank test due to non-normality of the data. Significant P-value was .05.
Results and Discussion
Table 2 shows the results corresponding to the cohort analyzed for both control structures, DH-CC (column 1) and DH-CC-CHO (column 2). For a better analysis, configurations of DH-CC-CHO with no quantization of rescue carbohydrate (column 3) and DH-CC with limitation of glucagon to 1 mg/d without additional carbs (column 4) are also presented.
Evaluation Results.
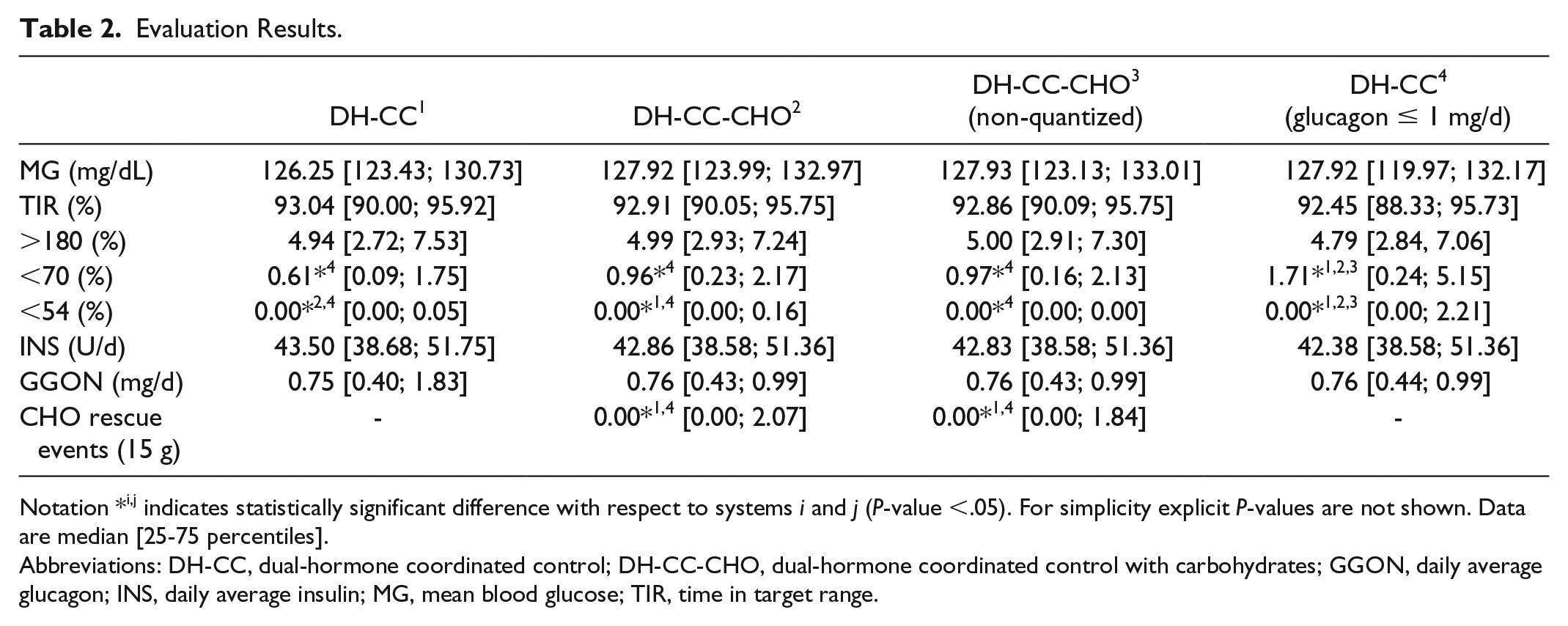
Notation *i,j indicates statistically significant difference with respect to systems i and j (P-value <.05). For simplicity explicit P-values are not shown. Data are median [25-75 percentiles].
Abbreviations: DH-CC, dual-hormone coordinated control; DH-CC-CHO, dual-hormone coordinated control with carbohydrates; GGON, daily average glucagon; INS, daily average insulin; MG, mean blood glucose; TIR, time in target range.
Performance of DH-CC and DH-CC-CHO was similar in terms of mean glucose (P = .088), time in range (P = .508), time above 180 mg/dL (P = .966), time below 70 mg/dL (P = .1364), insulin delivery (P = .383), and glucagon delivery (P = .407). For DH-CC, glucagon doses higher than 1 mg/d were only required by 30 adults (derived from the same three patients in the original ten-adult cohort) who seem to be more glucagon resistant. Statistically significant difference in time below 54 mg/dL was found (P = .036), although median time was 0% and 75-percentile 0.16% with low clinical significance. All events were mainly related to the same patient. This difference was not found in the case of DH-CC-CHO (non-quantized) (P = .265), where ideal continuous administration of carbs is considered, which indicates that rescue carbohydrate quantization might be responsible for the above difference, especially for that patient. However, no statistical difference was found in any metrics between DH-CC-CHO and DH-CC-CHO (non-quantized) concluding no major impact of the quantization process in the system performance. Limitation of glucagon delivery to 1 mg/d without additional carb intake to complete the required control effort (column 4 in Table 2) yielded higher time below 70 and 54 mg/dL, with statistical significance with all the other three configurations analyzed. If studies where delivered glucagon was greater than 1 mg/d for DH-CC are analyzed (n = 30), the addition of rescue CHO (DH-CC-CHO) implied a glucagon delivery lower than 1 mg/d (0.997 [0.997; 0.998] mg/d) and the need of 2.04 [1.71; 2.5] CHO rescues (1 rescue = 15 g).
Figure 3 illustrates the performance of the controllers of two patients where glucagon limitation was needed, as compared to glucagon limitation without rescue carb intake. Two days instead of all 14 days are represented in order to depict better the behavior described by the considered approaches. It can be observed that in these patients limitation of glucagon delivery to 1 mg/d would have provoked hypoglycemia during the afternoon (green dashed line), which was successfully avoided by the DH-CC-CHO controller (solid blue line) with the automatic suggestion of rescue carbs contributing with the same control effort than the needed glucagon, achieving a similar performance than DH-CC (dashed purple line).
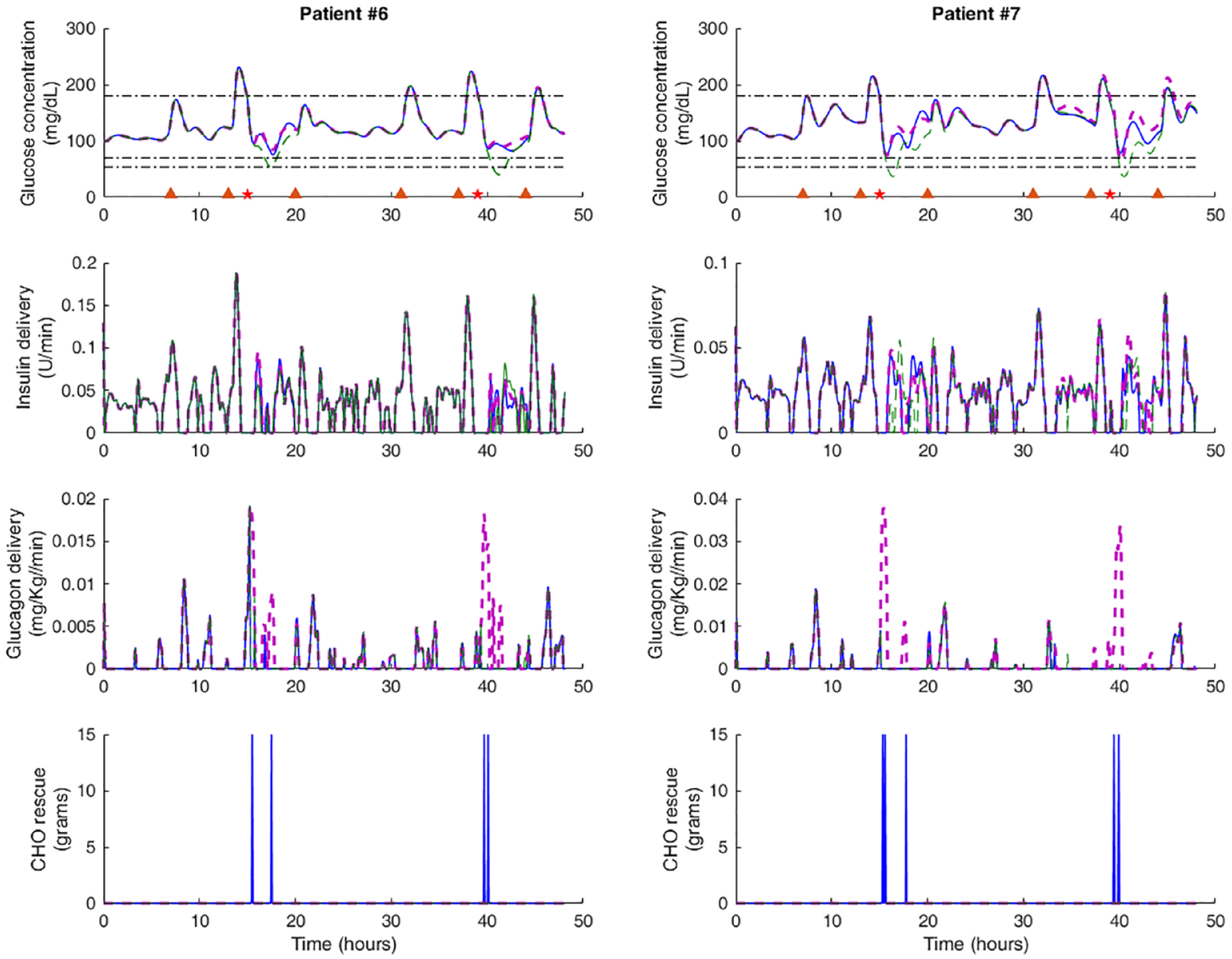
Comparison between different controller approaches: DH-CC (dashed purple line), DH-CC-CHO (solid blue line), and DH-CC (glucagon ≤1 mg/d) (dashed green line). Meals and exercise events are marked with triangles and stars, respectively.
This proof-of-concept study demonstrates (in silico) the flexibility of the parallel control strategy, where different divisor strategies can be devised attending to scenario particularities and user preferences, besides overcoming constraints on glucagon delivery. For instance, glucagon has shown to be more effective in exercise scenarios than postprandial periods, where rescue carbohydrate can be a better option for hypoglycemia mitigation. Additionally, other quantization strategies for practical carb intake by the patient could be derived. Clinical validation supporting the in silico results is needed since simulations might suffer from TIR overestimation, given current clinical results where TIRs of 75%-80% are achieved in closed-loop settings at the best.
Conclusions
A new control strategy for a DHAP system incorporating automatic suggestion of rescue carbohydrates in coordination with insulin and glucagon delivery was presented. The method allows to select among glucagon or rescue carbohydrate counterregulatory action, providing the same equivalent required control effort as computed by a master controller, following a parallel control structure. Application of this strategy to overcome constraints on daily glucagon delivery was successfully evaluated in a proof-of-concept in silico study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Spanish Ministry of Economy, Industry and Competitiveness (MINECO) through grant number DPI2016-78831-C2-1-R and the European Union through FEDER funds. Vanessa Moscardó was recipient of an FPU grant, FPU13/04253.