Abstract
Islet cell transplantation has been limited most by poor graft survival. Optimizing the site of transplantation could improve clinical outcomes by minimizing required donor cells, increasing graft integration, and simplifying the transplantation and monitoring process. In this article, we review the history and significant human and animal data for clinically relevant sites, including the liver, spleen, and kidney subcapsule, and identify promising new sites for further research. While the liver was the first studied site and has been used the most in clinical practice, the majority of transplanted islets become necrotic. We review the potential causes for graft death, including the instant blood-mediated inflammatory reaction, exposure to immunosuppressive agents, and low oxygen tension. Significant research exists on alternative sites for islet cell transplantation, suggesting a promising future for patients undergoing pancreatectomy.
Keywords
Introduction
Islet cell transplantation has been limited most by poor graft survival, requiring multiple donor pancreases to achieve euglycemia. Optimizing the site of transplantation could minimize required donor cells and improve clinical outcomes. Potential sites should limit exposure to blood and immune cells to prevent inflammatory reactions and provide similar oxygen tensions to the native organ. An ideal site would also provide easy access for both transplantation and monitoring. This article reviews human and animal data of clinically relevant sites using these criteria (Table 1).
Comparison of Sites of Islet Cell Transplantation Based on Select Criteria.
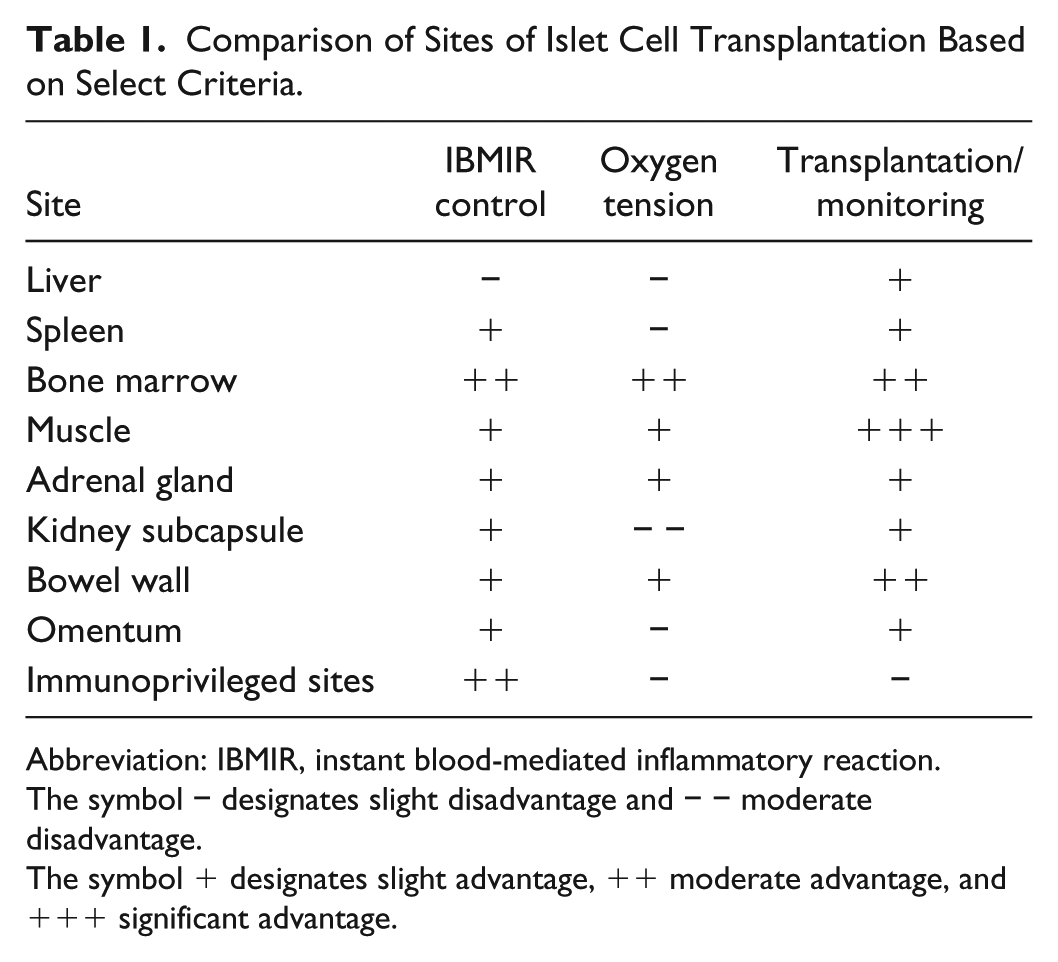
Abbreviation: IBMIR, instant blood-mediated inflammatory reaction.
The symbol − designates slight disadvantage and − − moderate disadvantage.
The symbol + designates slight advantage, ++ moderate advantage, and +++ significant advantage.
Liver
After early success with direct injection of islets into the rat portal vein in 1973 1 and with the first trials into the human portal vein in 1980,2,3 the liver has been the de facto site of islet transplantation. Technically, this site offers ease of access; cannulation of a branch of the portal vein can be performed percutaneously in an outpatient setting. While portal drainage provides more physiologic insulin circulation, islets transplanted into the liver are exposed to high levels of immunosuppressants and variable levels of glucose. After isolation and transplantation, more than 60% of islet cell mass becomes necrotic, 4 likely due to a combination of damage to the cells during isolation, cold ischemia time, an instant blood-mediated inflammatory reaction (IBMIR) and low oxygen tension in the liver. Finally, alpha cells are less functional in the liver compared to other sites of transplantation, and there is a concern about the development of hepatic neoplasms.
Instant blood-mediated inflammatory reaction describes an innate immune response occurring when pancreatic endothelial cells encounter ABO-compatible blood and release tissue factor, activating the coagulation and complement cascades that result in leukocyte and macrophage-mediated islet cell death.5,6 Some inflammatory reaction to islets is beneficial. Islet cells become encased in fibrin and adhere to the walls of the portal vein, causing ischemia that promotes revascularization and integration of the islets into the vessel wall, eventually allowing the islets to receive blood from the vasa vasorum. 7 However, islet cell emboli and inflammation have caused significant morbidity and mortality to both the graft and the patient with reports of portal hypertension and fatal hepatic necrosis, 8 disseminated intravascular coagulation and massive intraoperative bleeding, 9 and esophageal varices requiring a mesocaval graft. 10 These complications may be avoidable. With improvements in isolation technique and by infusing packed cell volumes less than 5 mL, Shapiro’s group has reported a 3.7% occurrence of partial portal thrombosis with no complete thrombosis in 268 intraportal islet transplants. 11
After this initial immune response, islet cells must uptake oxygen and nutrients by diffusion until fully vascularized, which in a rat model took ten days post-transplantation. 12 Despite making up 1% to 2% of the organ’s mass, islet cells demand a disproportionate amount of blood supply, requiring 5% to 20% of pancreatic blood flow. 13 Compared to the islets’ native tissue oxygen tension and parenchymal oxygen tension of 40 and 30 mmHg, respectively, the liver provides significantly lower tensions of less than 10 mmHg for both. 14 However, when compared to the kidney and spleen as transplantation sites, only the parenchymal oxygen tension is significantly lower in the liver. 14 Therefore, inadequate revascularization and oxygen tension is not site-specific and instead is likely specific to intrinsic properties of the islets, such as inadequate production of nitric oxide post-transplantation.
The Edmonton protocol, a sirolimus-, tacrolimus-, and daclizumab-based glucocorticoid-free immunomodulatory regimen established in 2001, confers insulin independence for 90% of allograft islet cell transplant recipients at one year with the liver as its most-used site of transplantation. 15 However, more recent studies have raised concerns about the longevity of insulin independence after three to five years, with most patients requiring return to insulin after five years in the most recent study.16,17 Additionally, sirolimus has been shown to cause anti-angiogenesis and decreased production of vascular endothelial growth factor (VEGF), impairing islet neovascularization. 18 Newer immunosuppressive regimens and enzymatic blends of collagenase and neutral proteases have contributed to an increased rate of success. 19 Considering the anatomy of the portal vein as the common drainage for the gastrointestinal tract, oral immunosuppressants reach higher and potentially more toxic levels in the portal circulation compared to the systemic. 20 Of note, the disadvantage of the liver as a transplant site is less challenging in autologous transplantation since immunosuppressants are not required. Additionally, the portal vein would likely expose islets to other circulating toxins and variable levels of nutrients and gut hormones, particularly postprandial hyperglycemia, which may cause large variability in insulin release. The clinical significance of these findings is currently unknown and warrants further research. While delivery of insulin into the portal circulation has been considered an advantage, 21 comparisons among sites have not shown a clinical difference between portal and systemic drainage. 22
When evaluating alpha-cell function, failure of islets to release glucagon in response to hypoglycemia has been long known to be liver-site specific. 23 Removing vagal innervation during transplantation decreases the release of pancreatic polypeptide. Additionally, hepatocyte production of glucose during hypoglycemia causes normalized glucose levels surrounding transplanted islets and limits ability of islets to recognize systemic hypoglycemia. 24 This impaired alpha function and decreased symptom awareness in liver site transplants increases the risk of hypoglycemic coma. 25
In a group of 800 streptozotocin-diabetic rats, the vast majority developed hepatocellular neoplasms after 15 to 18 months, triggered by local hyperinsulinism and hyperglycemia via the Ras-Raf protein kinase pathway and insulin-like growth factor system. 26 While this process has not been demonstrated in humans, the preponderance raises concerns about the long-term safety of transplantation into the liver and suggests close monitoring of patients with ultrasound or magnetic resonance imaging.
Although the liver has historically been a successful and easily accessible site of islet cell transplantation, site-specific alpha cell dysfunction clearly encourages the search for a more optimal site. While there is serious necrosis of islet cell mass, IBMIR, and low oxygen tension in the liver, these problems affect other sites, including the kidney and spleen, to potentially similar extents. Further research needs to be performed to evaluate the clinical significance of higher levels of enterotoxins and variable levels of glucose, drugs, and enterohormones in the portal vein.
Spleen
Considering other organs that can provide insulin release into the portal system, the spleen has been studied in mice, 27 dogs, 28 monkeys, 29 and birds. 30 In a study in diabetic mice, islet cells were injected directly into the splenic parenchyma and compared to the portal vein and underneath the kidney capsule. The spleen required fewer islets to improve hyperglycemia, induced less acute inflammatory response measured by cytokine response and, most importantly, demonstrated islet cell growth after transplantation based on upregulation of T-cell leukemia homeobox 1 genes that regenerate insulin-producing cells. 27 Compared to the subcapsular space, the spleen has similar glycemic control, with no specific benefit for portal vs systemic drainage found. 31 While transplantation into the spleen does not confer a risk of portal hypertension, this site has been considered to have a high risk for hemorrhage similar to the liver. 32 Notably, descriptions of serious hemorrhage or other procedure-related complications are notably lacking in the literature. Injection into the splenic vein or parenchyma exposes islets to ABO-compatible blood and thus renders them theoretically vulnerable to IBMIR; however, this needs to be further studied before any definitive conclusions can be made.
Interestingly, the spleen may play an important role for islet transplantation not as a site but as a source. Hyperglycemia in mice with and without diabetes induces proinsulin production in extrapancreatic sites, including the spleen. 33 Data from quail have demonstrated that splenic cells can differentiate into insulin-producing cells, 30 suggesting that these could be harvested from the spleen for patients with an insufficient quantity of viable islets. Splenic mesenchymal stem cells have recently been shown to contribute to repair of damaged tissues and could possibly be used to repair native type 1 diabetic pancreases or ectopically transplanted islets in the future. 34
Bone Marrow
Avoiding IBMIR while still providing excellent oxygen tension requires a highly vascularized tissue such as bone marrow. The bone marrow can be easily accessed in a noninvasive ambulatory setting for implantation or monitoring and provides a large bed for transplantation. 35 Cantarelli et al performed the first comparison between the portal vein and the bone marrow, using the femur in streptozotocin-induced diabetic mice, finding that the bone marrow was 2.5 times more likely to induce euglycemia (95% CI 1.2-5.2) and did so faster, taking 85 days vs 180. 35 Considering potential side-effects, they found no effect on hematopoietic function and a normal proliferative response to virally induced pancytopenia. While there was a 4% to 6% loss of bone density, 35 choosing a nonweight-bearing site could avoid clinically significant fractures. Compared to the liver, the islet cell mass in the bone marrow had a normal ratio of alpha to beta cells 35 vs alpha cell paucity in the liver.
These results were replicated in tibial bone marrow of rhesus monkeys with no post-transplant hypoglycemia and no impact on bone marrow density. 36 In the first clinical study on four patients post-total pancreatectomy, Maffi et al transplanted autologous islet cells into the iliac crest using the procedure for cord blood transplantation in patients with acute leukemia. These patients required exogenous insulin but demonstrated C-peptide release and sustained pancreatic function for more than 900 days after transplantation, with no significant adverse effects linked to the procedure. 37 The bone marrow may avoid liver-site complications while providing superior islet cell function long-term, but these results need to be validated in larger trials.
Muscle
Striated muscle has proven to be a successful site for parathyroid tissue in patients undergoing parathyroidectomy, particularly in Japan, where transplanted tissue remains functional for more than ten years. 38 Early experimental models for islets have had limited success, 39 likely due to significant peri-islet fibrosis and early hypoxia with large transplant volumes and islet cell clumping. 40 Christoffersson et al injected small volumes of nonclumped islets in a pearls-on-a-string fashion, and in doing so, resulted in euglycemia within eight days with blood flow and vessel density, comparable to the native pancreas.41,42
Remarkably, in 2007, a seven-year-old child with hereditary pancreatitis underwent pancreatectomy with auto-islet cell transplantation into the brachioradialis muscle. In combination with low doses of insulin via a pump, her diabetes is well-controlled with no episodes of hypoglycemia or pancreatitis, resulting in a significantly improved quality of life at two years post-transplant. 43
While transplanted islet cells diffuse throughout the liver and its vasculature, islets transplanted into skeletal muscle remain more localized and are therefore more easily monitored with biopsies and PET scans.44,45
Adrenal Gland
The adrenal gland, with its dense vasculature 46 and anti-apoptotic microenvironment, 47 has been successful in a mouse model with normoglycemia at ten days post-transplant. 48 Interestingly, growth hormone-releasing hormone receptor agonists administered to the islet cell-adrenal cell culture prior to transplant have been shown to increase viability, likely due to the promotion of islet function and the reduction of apoptosis.48,49 However, significant work must be done before the adrenal gland can be a viable option for clinical testing.
Kidney Subcapsule
Since 1986, the kidney subcapsule has been studied extensively in rodent models. 50 This site has been superior to liver and spleen, 51 omentum, 31 and lung, liver and spleen 52 ; however, in other studies, the subcapsule has been shown to be inferior to the spleen, 27 demonstrating the difficulty of comparing successes across studies and species. When compared to implantation into the liver, omentum, and muscle, implantation into the subcapsular space had the shortest operative time, lowest mortality, and best glucose control. 53 There has been limited clinical success with this site, likely owing to the difficulty of stretching the subcapsular space for implantation, 54 risks of exocrine contamination, 55 and poor blood supply resulting in oxygen tension of about one-tenth that of the pancreas. 14 Moreover, the prevalence of diabetic nephropathy may limit the patient population able to withstand the surgery.
Bowel Wall
Early in organogenesis, foregut endoderm buds into mesoderm and an interaction between epithelium and mesenchyme results in differentiation into the exocrine and endocrine cells of the pancreas. 56 In vitro, culturing islets with foregut submucosa increases overall yield and doubles the insulin response, 57 suggesting that the epithelium-mesenchymal interaction within the gastrointestinal wall may provide a microenvironment hospitable for islet longevity and function. There are a few particular advantages to this site: it can be accessed via laparoscopy or endoscopy for transplantation and monitoring, and its dense vascular network provides oxygen tension comparable to that of the pancreas. 58 Submucosa, either gastric or small intestinal, has been evaluated as early as 1926 in dogs, 59 and more recently in pigs, 58 rats, 60 and hamsters, with the latter study demonstrating beta-cell regeneration after transplantation. 61 The hospitable microenvironment may be harnessed using a scaffold seeded with submucosa and mesenchymal stem cells to increase clinical outcomes for other sites as well. 62 Two initial procedures were performed in 2013 and 2014 to endoscopically implant autologous islet cells into the gastric mucosa; after three-year followup, both patients had improved C peptide from 0.1 ng/mL to 3.0 and 0.6 ng/mL with improvement in pain from chronic pancreatitis. 63 A third patient from the same Polish research group received an endoscopic islet transplant after failed pancreas transplant and due to persistent portal hypertension making the portal vein a dangerous site. After four-year followup, her insulin requirements decreased from 60 to 27 U/day, showing promise for the safety and efficacy of the gastric wall as a site for transplantation. 64
Omentum
Since Graham described the omental patch in 1937, this site has been used to facilitate repair and regeneration of tissue. 65 Once this sheet of adipose tissue and immune cells recognizes injury, the proportion of immune cells rapidly increases to provide injured tissues with VEGF for angiogenesis, chemotaxins to attract progenitors from the bone marrow, and growth factors for a supportive local milieu. 66 Technically, a pouch can be created between the dual layers of the omentum, facilitating large cell volumes or bioengineered materials. 67 The omentum can also be mobilized and implanted subcutaneously, allowing monitoring with ultrasonography or percutaneous biopsies. 68 This site has been studied in rodents, 69 dogs, 70 and nonhuman primates, 68 outperforming the kidney capsule, 69 spleen, 71 and liver. 72 In a study with immature beta-cells, revascularization in the omentum exceeded that in the kidney capsule with larger vessels and proliferation of beta-cells with insulin levels twice as high in the omentum than the kidney capsule. 69 However, this study 69 and others 70 have demonstrated no alpha cell proliferation and abnormal alpha cell response to hypoglycemia. Additionally, the immune response and revascularization takes about a week longer for glucose normalization in the omentum than the portal bed 72 or subcapsule 67 and results in 10% graft failure. 72 While more work needs to be done for early graft ischemia, the recent clinical trial with bioengineered scaffolds is promising, with preliminary results in a middle-aged woman with type 2 diabetes mellitus showing at least 12 months of glycemic control without exogenous insulin. 73
Immunoprivileged Sites
While the development of the Edmonton protocol significantly improved outcomes in allografts, choosing an immunoprivileged site may allow reducing or foregoing an immunosuppressive regimen and its resultant side-effects. The testis, thymus, and the anterior chamber of the eye have been studied as possible sites for allografts or xenografts with varying levels of success. While immunoprivileged sites generally may not accept large enough numbers of islets to result in euglycemia, 74 a small number of islets could theoretically be transplanted into these sites to induce peripheral tolerance and support a second transplant site that can accept an adequately sized graft. Significant work needs to be done before this strategy could be employed in clinical practice.
Sertoli cells in the testis confer immune privilege by modulating the balance of cluster of differentiation 8 positive (CD8+) memory T cells and more antigen-specific CD4+CD25+ Treg cells, thereby stimulating immune tolerance. 75 Sertoli cells can also be transplanted with islets, modulating the local immune environment to benefit islet cell survival, as demonstrated in a rat model with greater rates of euglycemia for rats transplanted with Sertoli cells (90%) vs without (75%) into the kidney subcapsule. 22
As the thymus houses T cell negative selection, a process during which maturing T cells that bind too strongly to self-antigens die by apoptosis, transplanting into this site theoretically induces tolerance to alloantigens. A rat model demonstrated survival of islets in the kidney subcapsule after transplantation of few islets into the thymus with a dose of antilymphocyte serum without further immunosuppression. 76
While corneal transplantation has been successful, 77 islets transplanted into the anterior chamber of the eye are vulnerable to autoimmunity, suggesting that immunomodulation may be required. 78 This site has been studied in baboons 79 and mice, 78 demonstrating that as few as 50 islet cells can reverse hyperglycemia vs double for the kidney subcapsule. 78 After transplant, baboons did not demonstrate obvious signs of visual impairment or abnormalities in their ophthalmological exams. 79 Slit-lamp examination and tonometry allow noninvasive monitoring of the islet environment.
Conclusion
Islet cell transplantation via isolated islets or whole pancreas transplantation is the only method to prevent diabetes in patients who undergo total pancreatectomy and the only cure for type 1 diabetes mellitus. Compared to whole pancreas transplants, islet transplants have less insulin independence (59% vs 75%) but have less invasive surgical procedures and a better safety profile. 80 Islet cell transplantation is currently successful: the five-year survival rate of grafts ranges between 53% and 73% and the ten-year rate between 36% and 56%. 81 Due to the challenges posed by the liver site, including IBMIR, low oxygen tension, and alpha cell dysfunction, alternative sites have been increasingly studied; between 2008 and 2012, over 70% of published studies used the kidney capsule and only 23% used the liver. 82 With many new avenues for research, including bioengineered scaffolds and islet microencapsulation, the future of islet cell transplantation is promising.
Footnotes
Acknowledgements
We would like to acknowledge the efforts of Renee Cercone in editing this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
