Abstract

Since continuous glucose monitors (CGMs) first became commercially available in 2000, numerous studies have shown that their use improves the management of diabetes for both pediatric and adult patients and both type 1 and type 2 diabetes.1-5 However, there remain little data on their efficacy in certain subpopulations such as patients with significant medical comorbidities, mental health conditions, or substantial social barriers, who were largely excluded from the studies of CGM’s efficacy. 6
Commonwealth Care Alliance (CCA) is a not-for-profit, community-based healthcare organization that offers comprehensive health plans to patients dually eligible for Medicare and Medicaid, a medically and socially complex patient population. In April 2017, CCA launched a pilot program to implement professional CGMs in patients with diabetes who had poorly controlled hyperglycemia or suspicion of asymptomatic hypoglycemia.
The device used in this pilot was the Freestyle Libre Pro System, a professional CGM that does not provide patients with real-time access to glucose readings. Patients continued their prior methods of glucose monitoring and control while using the device. The CGM was placed for a minimum of one week (range: 8-18 days). The data collected were reviewed by CCA clinicians and used to guide clinical decision making. Surveys were conducted with the patients and clinicians to determine the user and provider experience with the device.
Thirteen patients were enrolled in the study (Table 1). The cohort had a high burden of chronic disease (average of 14.5 chronic conditions), behavioral health conditions (100% carried a mental health diagnosis), functional needs (100% required assistance with activities of daily living or instrumental activities of daily living), and recent hospitalizations (62% were hospitalized in the last year). The average preCGM hemoglobin A1c (HbA1c) was 9.4% with a range of 6.2% to 14.5%. The average change in HbA1c was −0.5% (P = .38). Among the nine subjects with starting HbA1c values above 8% (enrolled for uncontrolled hyperglycemia), the average HbA1c change was −0.9% (P = .23).
Characteristics of Study Participants. Data on Demographics, Functional Status, Housing Security, Acute Care Utilization, and Medical History for Study Participants.
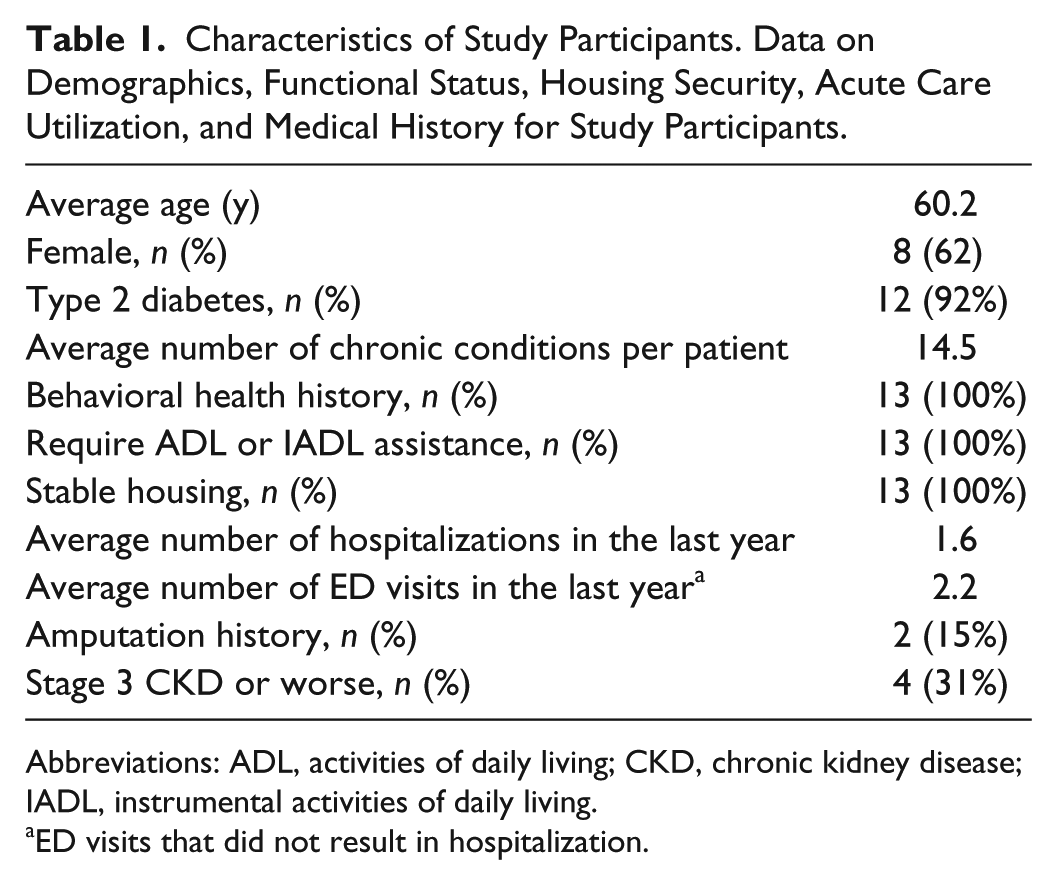
Abbreviations: ADL, activities of daily living; CKD, chronic kidney disease; IADL, instrumental activities of daily living.
ED visits that did not result in hospitalization.
Eight patient surveys were completed. A majority of respondents (six subjects) reported liking the device, citing that they felt more in control (three subjects). Similarly, seven respondents found the CGM helpful, five reported that it changed how they viewed their diabetes, and four reported that it changed how they treated their diabetes.
Eleven clinician surveys were completed. Ten respondents found that the CGM provided clinically useful data and seven felt that the information led to clinical improvement in diabetes control. Qualitative analysis of these surveys revealed five themes: (1) identification of asymptomatic hypoglycemia, (2) better characterization of hyperglycemia, (3) utility in patients who do not perform finger-stick glucose checks, (4) increased patient engagement in diabetes care, and (5) challenges with sensor displacement.
This study demonstrates that CGM device use in high-risk, complex patients can uncover episodic hypoglycemia and better trend hyperglycemia, which can provide clinicians with valuable information. These devices can also increase patient engagement and motivate better adherence to treatment regimens in a patient population that traditionally has barriers to engagement. More studies are needed to determine whether these benefits result in lasting improvements in glycemic control in this patient population.
Footnotes
Acknowledgements
The authors are grateful to the staff of Commonwealth Care Alliance for participating in and facilitating this study. They would also like to thank the health plan members of Commonwealth Care Alliance for inspiring this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data use statement
De-identified data will be provided upon request. For access to data, please contact
