Abstract
Insulin-dependent diabetes mellitus is treated with intensive insulin therapy using multiple daily injections or continuous subcutaneous insulin infusion with insulin pumps. For people with diabetes who cannot achieve acceptable glycemic control despite the use of intensive insulin therapy and continuous glucose measurement, there exists the possibility of continuous intraperitoneal insulin delivery via an implantable pump or a percutaneous port system that is connected to an external insulin pump. In this article, the current second generation of the Accu-Chek® DiaPort system for continuous intraperitoneal insulin delivery with its improvements over the former generation is presented and discussed.
Keywords
Continuous intraperitoneal insulin infusion (CIPII) is a particular type of insulin pump therapy characterized by the administration of insulin directly into the peritoneal space. This therapy enables more physiological insulin administration than subcutaneous delivery (rapid resorption to the portal venous system, avoiding the unphysiological distribution in the peripheral circulation) and provides therefore an option for better glucose control with lower HbA1c and less hypoglycemia. It is absolutely indicated when subcutaneous application of insulin is not possible or does not lead to adequate insulin levels in the blood.1,2 In addition to implantable pumps, percutaneous port systems are an alternative option to enable CIPII. In 2000, a percutaneous port system, the Accu-Chek® DiaPort (Roche Diabetes Care GmbH, Mannheim, Germany) was introduced and since then has been used for CIPII. It consists of a small port body which is implanted into the subcutaneous tissue of the abdomen, a catheter entering the peritoneal cavity, and an ordinary external insulin pump that is connected to the port via a specific infusion set. While the implantation and possible complications (eg, ingrowth of the abdominal catheter) require contribution of a surgeon, membrane and catheter replacements in Seldinger technique are usually performed by trained diabetologists. The external pump and infusion set can be handled and maintained by the patients themselves. For best long-term outcomes strict hygiene and correct handling by the patients are required.
Even though the first generation of DiaPort was shown to be safe and effective for CIPII, 3 complications in the tissue around the implantation site, and insulin underdelivery due to catheter occlusions occurred during the years of use. Subsequently, the manufacturer decided to apply experience from practical use and to develop a second generation with the purpose of overcoming these problems and increasing convenience. The improvements in design will be described in this article.
The New Design
Figure 1 shows the first and second generation of the Accu-Chek DiaPort system, and the principal components of the current second generation. Individual parts and design changes compared to the first generation are described below.
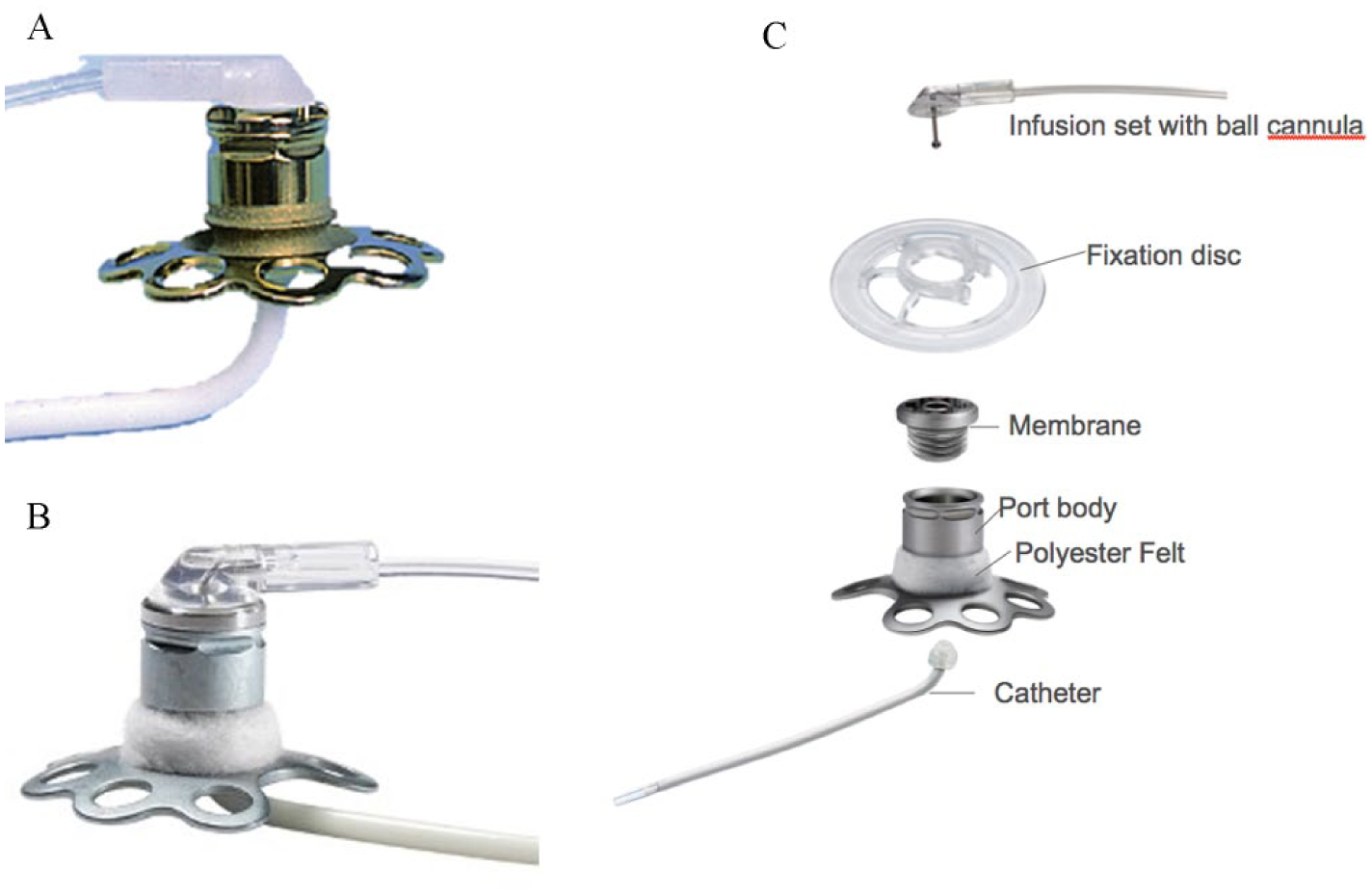
(A) DiaPort first generation. (B) DiaPort second generation. (C) Components of the DiaPort second generation.
Port
Port Body
The port body, that is, the part that is placed in the subcutaneous tissue allowing access into the abdominal space, is made of titanium. While the first generation was 10.8 mm high with a diameter of 26 mm, the second generation was enlarged to a height of 12.8 mm. In addition, the number of circles stabilizing the port body on the inner side of the subcutaneous tissue was reduced from 8 to 7. The gap between titanium and the human tissue is prone to superficial infections that might result in severe inflammation and even peritonitis. 4 Therefore, the updated version is partially covered with a polyester felt that allows a stronger connection between the port and surrounding tissue, and consequently an enhanced barrier to infections.
Catheter
The first port generation combined two different catheters: an outer guide catheter connected to the port body, and an inner membrane-containing catheter that was introduced through the guide catheter. The guide catheter was very stiff and therefore easy to place but did not easily move in the peritoneal cavity causing irritations and pain. The second port generation has only one catheter that is not fixed to other components facilitating catheter exchanges. Because of changes in the material the single catheter is now soft and flexible leading to reduced overgrowth and less pain. The inner diameter was enlarged from 0.25 mm to 0.74 mm to reduce occlusions by crystallization of insulin. In addition the catheter tip was rounded and trumpet-shaped to further reduce occlusion as well as tissue irritation. 5
Membrane
The membrane is the barrier between the internal and external parts of the port system. As such it has to be changed regularly to maintain integrity and to ensure proper function of the system. Since in the first port generation it was fixed to the catheter, membrane and catheter had to be exchanged together. The second generation provides the membrane premounted only within the lid that is screwed onto the port body. Regular replacements of the membrane catheter were necessary every 3 months for the first generation, and this interval could be prolonged to 6 months for the membrane of the second generation.
Infusion Set
The infusion set connects the external pump to the port via a ball cannula. Materials for the second generation were exchanged to make the connection more robust and provide a more skin-friendly contact. The handling aid to connect and disconnect infusion set and port was enlarged which makes it easier to use. In order to adapt to patient requirements, other tubing lengths (40 cm and 70 cm) were made available. The infusion set has to be changed at least every 6 days.
Fixation Disc
The external part of the port body has to be fixed with a fixation disc to avoid dislocation of the port during ingrowth, and the following period, as well. The updated version of this fixation disc consists of a more flexible material to improve wearing comfort and to facilitate its application. In addition the new fixation disc is transparent instead of white for more discretion.
Implantation Tools
The port system is routinely supplied with a complete set of tools that are required for its implantation and further handling. Design changes were implemented to these tools as well to simplify and accelerate the implantation and the change of catheter or membrane. These tools include a layer scalpel to cut the subcutaneous pocket exactly in the recommended depth of 6 mm, where the port body will be placed, and a skin punch to create a circular incision above the pocket through which the port crosses the skin. A punch plate can be inserted into the subcutaneous pocket and helps creating the circular incision.
While for the first port generation the blade had to be mounted to the layer scalpel, it is premounted in the second generation in order to increase ease of use as well as safety of the surgeon.
For membrane replacements the first DiaPort was provided with a torque wrench to unscrew the lid with the membrane catheter from the port body and to screw in the new one. The second generation offers two separate instruments: a screw-off screwdriver for removing the lid with the membrane, and a screw-in screwdriver that already contains the new lid, therefore reducing handling steps. Both tools are considerably smaller than the former torque wrench so that trained diabetologists have better sensitivity and there is no more need for a torque.
Because in the second generation the catheter is not fixed to the lid any more, an additional tool, the catheter extractor, is required to remove the catheter out of the body. The implantation of the port system is normally performed under general anesthesia. Experienced medical teams can perform implantations under local anesthesia, as well.
Design Validation
The new device was investigated in a design validation study including 12 patients. Regular human U100 insulin with Genapol® stabilization (Insuman Infusat, Sanofi, Paris) showed to be the most compatible pump insulin for use in DiaPort. In addition to an evaluation of safety and effectiveness, patients, surgeons and diabetologists filled out questionnaires regarding the new design of the port and tools. Although it was not directly compared to its predecessor, a successful implementation of the design improvements in terms of patient safety could be documented. Considering handling of the devices, the surgeons rated the usability of the implantation tools and tools for replacement of the catheter or membrane as easy and safe. Catheter replacements could be performed within a mean duration of 9 minutes, usually as an outpatient procedure by trained diabetologists. Patients did not have problems with the handling of the infusion set and the fixation disc, nor with the daily care of the insertion site.
Footnotes
Acknowledgements
The authors thank Delia Waldenmaier (IDT, Ulm, Germany) for writing support.
Abbreviation
CIPII, continuous intraperitoneal insulin infusion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CR, KK, and WMH are full-time employees of Roche Diabetes Care GmbH. BG and AL declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. BG and AL received from the following companies fees or financial support for the participation in advisory boards, general advice, untied research support, or other medical-scientific services: AstraZeneca, Bayer, Becton Dickinson, Boehringer Ingelheim, Bristol-Myers Squibb, DexCom, Lilly, Medtronic, MSD, Novo Nordisk, Roche, Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Roche Diabetes Care GmbH.
