Abstract
Background:
Skin autofluorescence has been used to assess longer term glycemic control and risk of complications. There is however no agreed site at which autofluorescence should be measured. This study evaluated the within- and between-site agreement in measurement of skin autofluorescence using a noninvasive advanced glycation end product (AGE) reader.
Methods:
Overall, 132 participants were included: 16 with diabetes-related foot ulcers (DFU), 63 with diabetes but without foot ulcers (DMC), 53 without diabetes or foot ulcers (HC). Skin autofluorescence was measured using the AGE Reader (DiagnOptics technologies BV, the Netherlands). Three consecutive skin autofluorescence measurements were each performed at six different body sites: the volar surfaces of both forearms (arms), dorsal surfaces of both calves (legs), and plantar surfaces of both feet (feet). Within- and between-site agreements were analyzed with concordance correlation coefficients (CCC) and 95% confidence intervals (95% CI), absolute mean differences (±standard deviation), and Bland-Altman limits of agreement.
Results:
The agreement between repeat assessments at the same site was almost perfect (CCC [95% CI] ranging from 0.94 [0.91-0.96] for assessments in the right foot to 0.99 [0.99-0.99] for assessments in the left arm). The limits of agreement were narrow within ±0.5 arbitrary units for all sites. The between-site agreement in measurements was poor (CCC < 0.65) with large maximum absolute mean differences (±SD) in arbitrary units (DFU = 3.40 [±2.04]; DMC = 3.15 [±2.45]; HC = 2.72 [±1.83]) and wide limits of agreement.
Conclusions:
Skin autofluorescence measurements can be repeated at the same site with adequate repeatability but measurements at different sites in the same patient have marked differences. The reason for this variation across sites and whether this has any role in diabetes-related complications needs further investigation.
Keywords
Diabetes mellitus can result in both macro-vascular and micro-vascular complications including cardiovascular, eye, kidney and foot diseases.1-3 Diabetes-related foot disease (DFD) is one of the top ten global causes of disability and is associated with high mortality, frequent recurrence and substantial health care costs.4,5 Innovative assessment and management strategies are required to combat the global DFD epidemic. 6
Chronic hyperglycemia due to diabetes is thought to be an underlying cause of diabetic peripheral neuropathy (DPN) and peripheral artery disease (PAD), both of which are significant risk factors for DFD. 7 Hyperglycemia promotes accumulation of advanced glycation end products (AGEs), which stimulate secondary detrimental effects at a molecular and cellular level. 8 These include stimulation of the receptor for advanced glycation end products (RAGE) and promotion of inflammation, for example, by up-regulation of nuclear factor-κB and its target genes. 9 Glycation may also change the biomechanical properties of tissues via collagen cross-linking and thereby influence the mechanical response of tissues to load.10-12 This heightens the risk of tissue injury due to mechanical stress.13-16 Hence tissue glycation and repetitive mechanical loading, in the presence of loss of protective sensation due to DPN may promote the development of diabetes-related foot ulcers (DFUs). 17
Autofluorescence of the volar surface of the arm was previously correlated with biopsied tissue concentrations of some AGEs (
Skin autofluorescence has typically been measured on the volar surface of the forearm, 8 although measurements performed at the leg have also been reported.8,33 Despite this, it is currently unclear whether measurements of autofluorescence performed in the feet and legs agree with those performed in the arms. As fluorophores differ between body sites, 34 it is likely that skin autofluorescence measurements might also vary. 33 It is possible that the assessment of skin autofluorescence in the feet and legs could provide a more relevant estimate of the effect of glycation on lower limb biomechanics and risk of DFU development. The aim of this study was to investigate the within- and between-site agreement in autofluorescence measurements performed at different skin sites in people with diabetes-related foot ulcers, people with diabetes without foot ulcers and healthy controls.
Methods
Study Design and Setting
Data for this study are from 146 participants recruited for a larger original case-control study performed in North Queensland, Australia. 35 We investigated multiple hypotheses in the original study36,37 and a detailed study protocol exists. 35 In brief, there were three groups of eligible participants recruited: people with type 2 diabetes with an active plantar neuropathic foot ulcer (cases; DFU group); people with type 2 diabetes without a history of foot ulcers (diabetes controls; DMC group); and people with no history of type 2 diabetes or a foot ulcer (healthy controls; HC group). The study was approved by two local human research ethics committees. Written informed consent was obtained from all participants.
Inclusion and Exclusion Criteria for Cases and Controls
Inclusion criteria for the DFU group were adults with a confirmed diagnosis of type 2 diabetes and a single active unilateral plantar DFU of longer than 3 months duration. 35 The DMC group comprised of adults with a confirmed diagnosis of type 2 diabetes without a history of DFU. 35 The HC group comprised of adults without a history of diabetes or DFUs. 35 Exclusion criteria included 36 (1) orthopedic, musculoskeletal, vestibular, visual or neurological problems affecting mobility (other than DPN); (2) previous orthopedic surgical intervention of the lower limb; (3) PAD defined as an ankle brachial pressure index (ABPI) of < 0.8; and (4) planned vascular reconstructions in the subsequent 12 months. 35
Description of Variables Collected
The procedures used for the collection of all variables have been described in detail previously.35,38 One trained investigator (MEF) conducted all assessments. Skin autofluorescence was measured noninvasively using the AGE Reader (DiagnOptics technologies BV, Groningen, the Netherlands). 20 Skin autofluorescence was defined as the average light intensity of the emission spectrum between 420 and 600 nm divided by the average light intensity of the excitation spectrum between 300 and 420 nm, multiplied by 100 and expressed as a percentage in arbitrary units. 20
Detailed methodology for collected variables are reported in the main study protocol. 35 These variables included age, sex, ethnicity, smoking status, and medical history in all participants and diabetes duration and insulin use in people with diabetes. Physical examination involved the assessment of height, mass, body mass index (BMI), body fat percentage, waist and hip circumference, resting systolic and diastolic blood pressure, 10g monofilament sensation assessed in 10 sites in each foot, and the Michigan Neuropathy score. 39 DPN was defined as a monofilament score of ≤8 in either foot. All methods used to obtain clinical and anthopometric measures in the study had good to excellent repeatability. 38 All participants with diabetes were referred for a fasting peripheral venous blood sample for the evaluation of glycated hemoglobin A1c (HbA1c), total cholesterol to HDL ratio, C-reactive protein, homocysteine, and estimated glomerular filtration rate (eGFR). 40
Skin Autofluorescence Measurement Protocol
The AGE Reader illuminates a skin surface of approximately 4 cm2, with an excitation light source with peak intensity at 370 nm.20,21 Guarded against surrounding light, emission light, and reflected excitation light from the skin is measured with a spectrometer. The device was rested on a stable bench top for performing arm measurements and on a raised wooden platform for performing lower limb measurements (see Figure 1). Three consecutive measurements of skin autofluorescence were each performed at six different body sites: arms—the volar aspect of both forearms (approximately 10 cm from the elbow); legs—the exact midpoint between the popliteal fossa and the gastrocnemius muscle belly of both lower legs (to lessen interference from muscle and adipose tissue); and feet—the plantar surface of both forefeet approximately 1 cm distal to the third metatarsal head. Measurements were performed on the same day in a random order. All measurements were performed in a dimly lit testing environment at room temperature to minimize interference from external light sources. 8
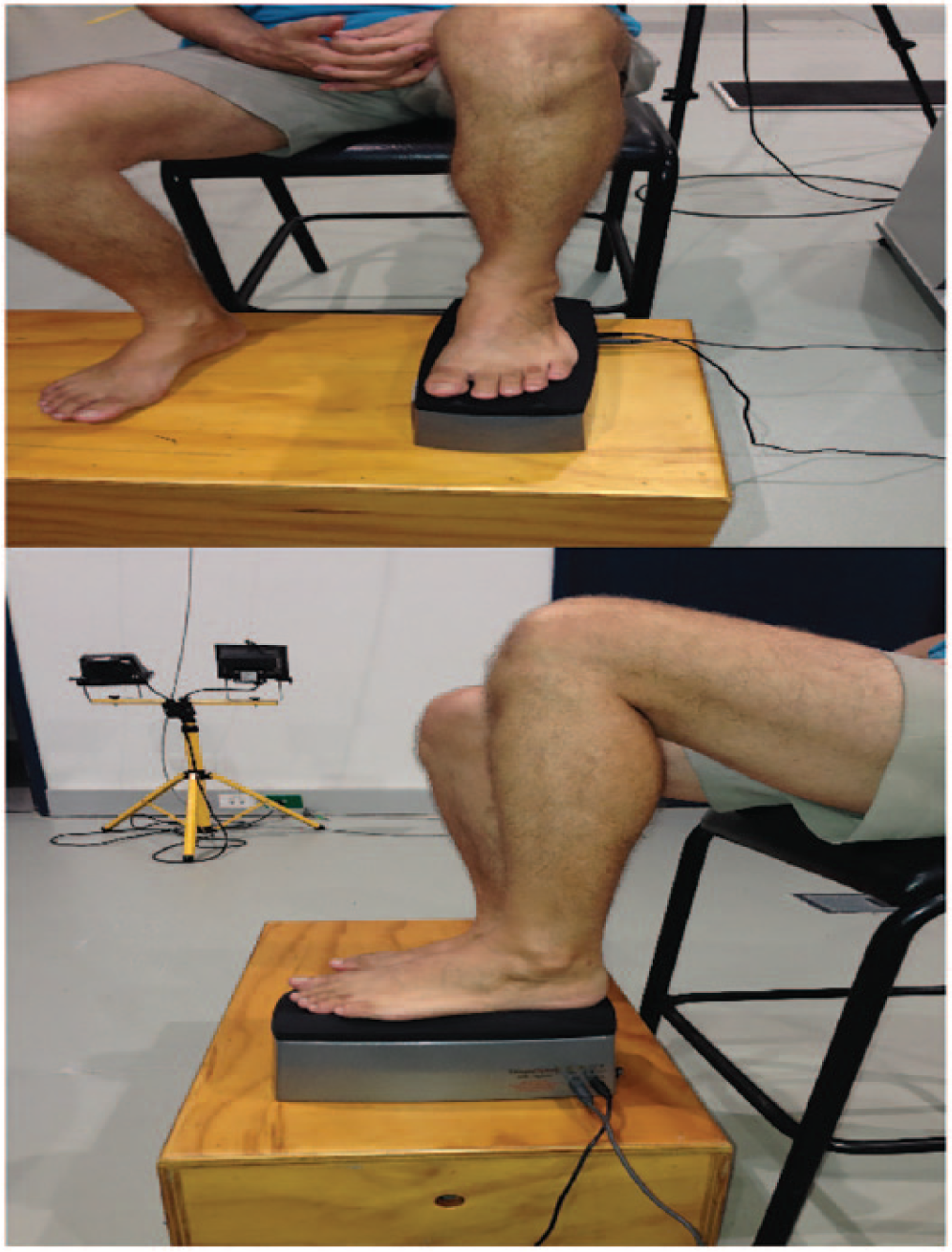
Skin autofluorescence measurement of the foot. Figure demonstrates the positioning of the foot over the top of the AGE Reader (DiagnOptics technologies BV, Groningen, the Netherlands) to obtain skin autofluorescence measurements. The room was lit for purposes of illustration. Important to note that minimal weight was placed over the top of the device during measurement.
Measurements were performed at sites that were free of visible blood vessels, scars, lichenification, tattoos, or other skin abnormalities including DFUs and callus. 8 In people with DFUs, the plantar ulcers were located at the heel (n = 2), medial longitudinal arch (n = 3), first metatarsal head (n = 2), fifth metatarsal head (n = 1), apex of digits (n = 3), and hallux (n = 5). None of the DFUs were located at the site of skin autofluorescence measurement at the third metatarsal. Participants were advised to not use lotions, skin products or emollients on their skin at least two days prior to the assessment. 41 Most participants (n = 129) had relatively light skin pigmentation (Fitzpatrick Grades 1 to 3), 42 two participants had slightly pigmented skin (Fitzpatrick Grade 4), and one participant (n = 1) had deeply pigmented skin (Fitzpatrick Grade 5). 42 Despite this, measurements were obtained from all three sites in this participant. 43
Statistical Analyses
The Statistical Package for the Social Sciences (SPSS) for Windows (released 2013, IBM SPSS Statistics for Windows, Version 22.0, Armonk, NY, IBM Corp) and R (R Core Team, 2015) software (2015; RStudio, Inc., Boston, MA http://www.rstudio.com/) were used for all analyses. Continuous data were reported as mean and standard deviation (SD) if considered to be approximately normally distributed or median and interquartile range (IQR) if skewed. Categorical variables were reported as numbers and percentages (%). Differences in descriptive outcomes between the three groups were compared using a one-way analysis of variance (ANOVA) and Sidak’s post hoc tests or the Kruskal-Wallis test and post hoc Mann-Whitney
Lin’s concordance correlation coefficient (CCC) was calculated to examine the repeatability of measurements. 44 All CCCs were calculated using an online statistical calculator and reported with two-sided 95% confidence intervals (95% CI). 45 CCCs were interpreted as “almost perfect” (>0.90), “substantial” (>0.8-0.9), “moderate” (0.65-0.8), and “poor” (<0.65). 46
Absolute agreement between measures at each body site were assessed using the Bland-Altman limits of agreement (2 x the SD of the difference scores).
47
Absolute mean differences and SD were reported alongside CCCs for between-site comparisons. The median value of three measurements, the 95% CI of median measurement and range in skin autofluorescence measures were reported for the cohort and for each group. Pearson correlation coefficients for the differences in mean values against the mean, associated
Results
Participant Recruitment and Characteristics
A total of 146 recruited participants were invited to participate, but only 132 (90%) attended the additional study visit required for this study and had skin autofluorescence measurements performed. Multiple attempts were made to contact the participants that failed to attend. Some reasons for failure to participate (n = 14) included lack of available time, lack of interest in being involved, missed appointments, and failure to establish contact. Sixty-one participants (41.7%) could not have all autofluorescence measurements performed because no reading could be recorded, including from the right arm (n = 1), left leg (n = 36), right leg (n = 37), left foot (n = 10), and right foot (n = 29) despite multiple attempts to measure.
There were 16 participants in the DFU group, 63 in the DMC group, and 53 in the HC group (see Table 1). The average age of the participants was 61 years (SD ± 10.4 years), and 76 were male (57.6%) and 68 (51.5%) had a history of smoking. The average BMI was 31.6 kg/m2 (SD ± 19.9 kg/m2). In participants with diabetes (n = 79), 30 (38%) had DPN. Differences and similarities between the three groups of participants are detailed in Table 1.
Description of the Cohort Stratified by Group.
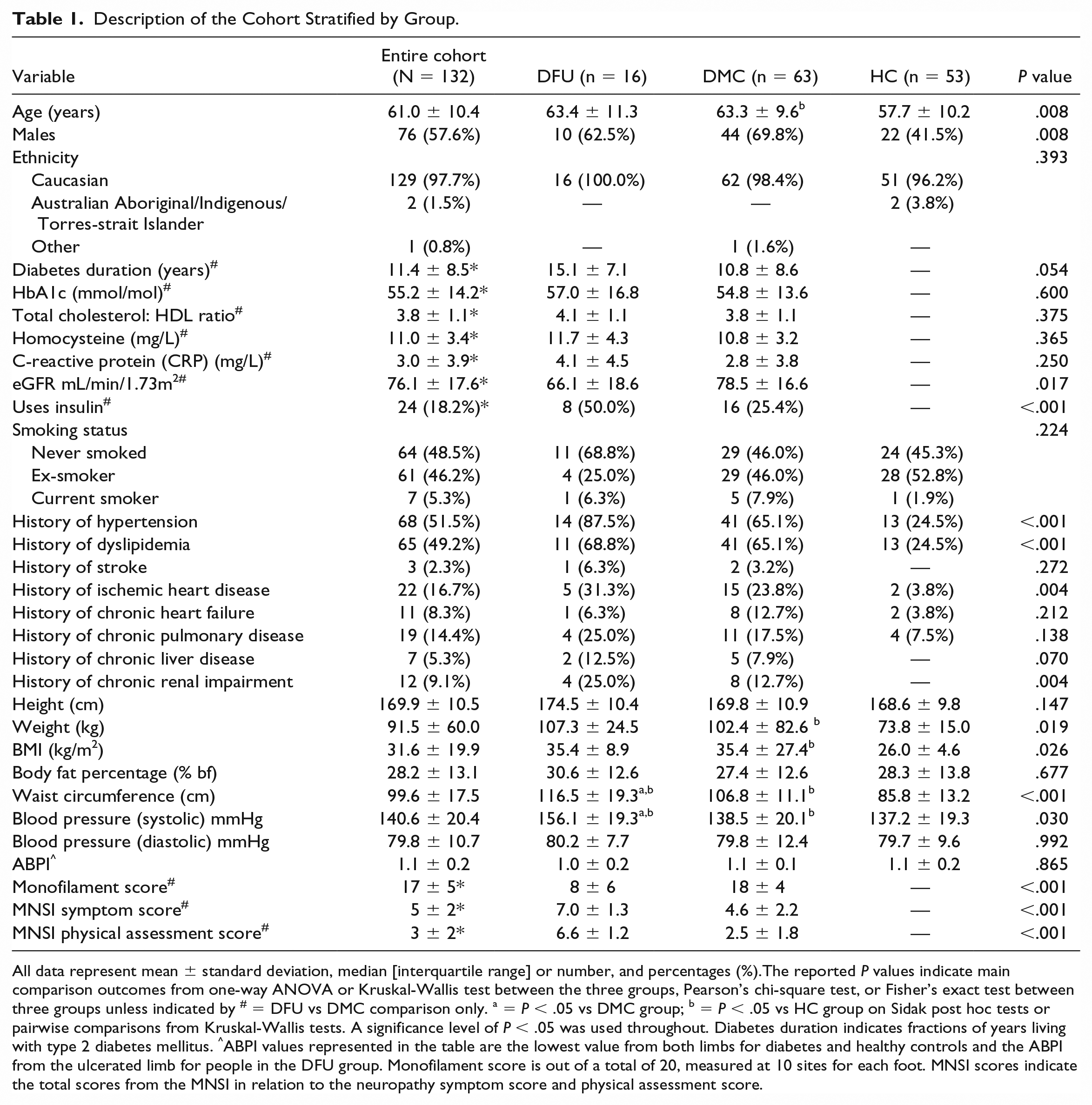
All data represent mean ± standard deviation, median [interquartile range] or number, and percentages (%).The reported
The descriptive characteristics of participants with missing autofluorescence measurements were evaluated (Supplementary Table 1). No obvious differences were noted between this subgroup and the entire cohort.
Within-Site Agreement
Table 2 shows that the agreement between repeat readings performed at the same site was almost perfect, with CCC ranging from 0.94 [0.91-0.96] for the right foot to 0.99 [0.99-0.99] for the left arm. Supplementary Table 2 shows all three participant groups also had substantial to almost perfect within-site agreement at all six sites, except for the left leg in the DFU group CCC = 0.75 [0.31-0.93]. The Bland-Altman limits of agreement were within ± 0.5 arbitrary units for all sites (Figure 2).
Agreement in Measurements Performed at Each Body Site Stratified by Limb.
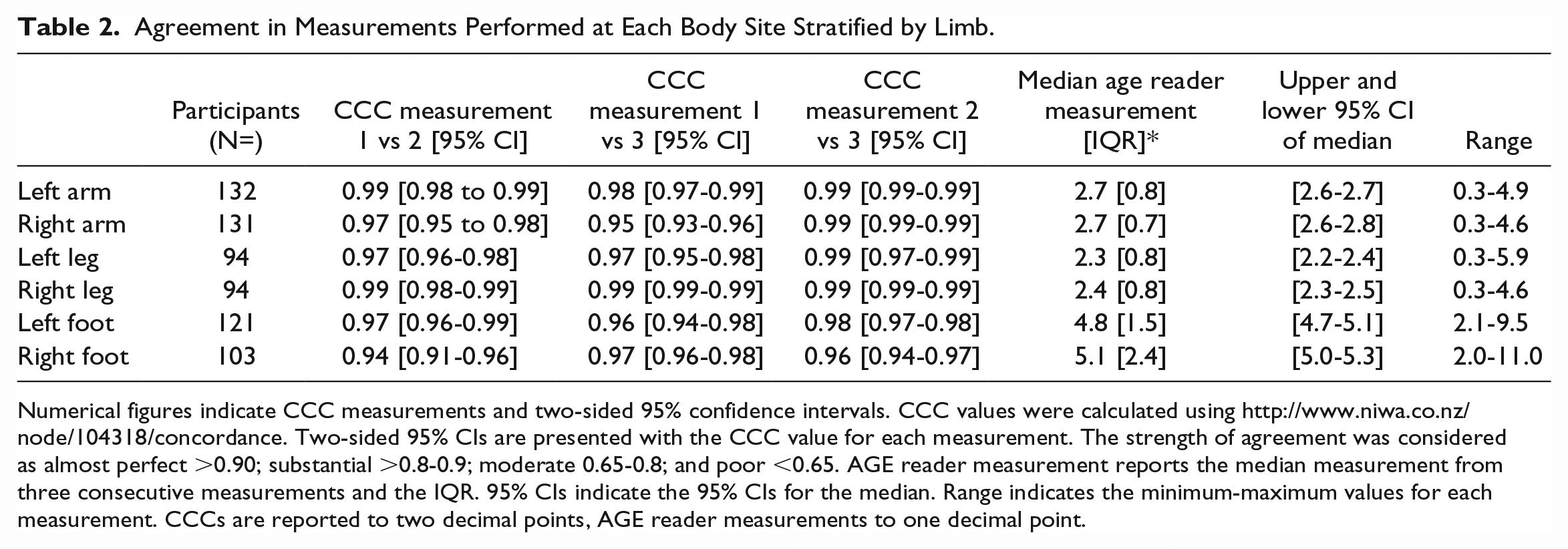
Numerical figures indicate CCC measurements and two-sided 95% confidence intervals. CCC values were calculated using http://www.niwa.co.nz/node/104318/concordance. Two-sided 95% CIs are presented with the CCC value for each measurement. The strength of agreement was considered as almost perfect >0.90; substantial >0.8-0.9; moderate 0.65-0.8; and poor <0.65. AGE reader measurement reports the median measurement from three consecutive measurements and the IQR. 95% CIs indicate the 95% CIs for the median. Range indicates the minimum-maximum values for each measurement. CCCs are reported to two decimal points, AGE reader measurements to one decimal point.
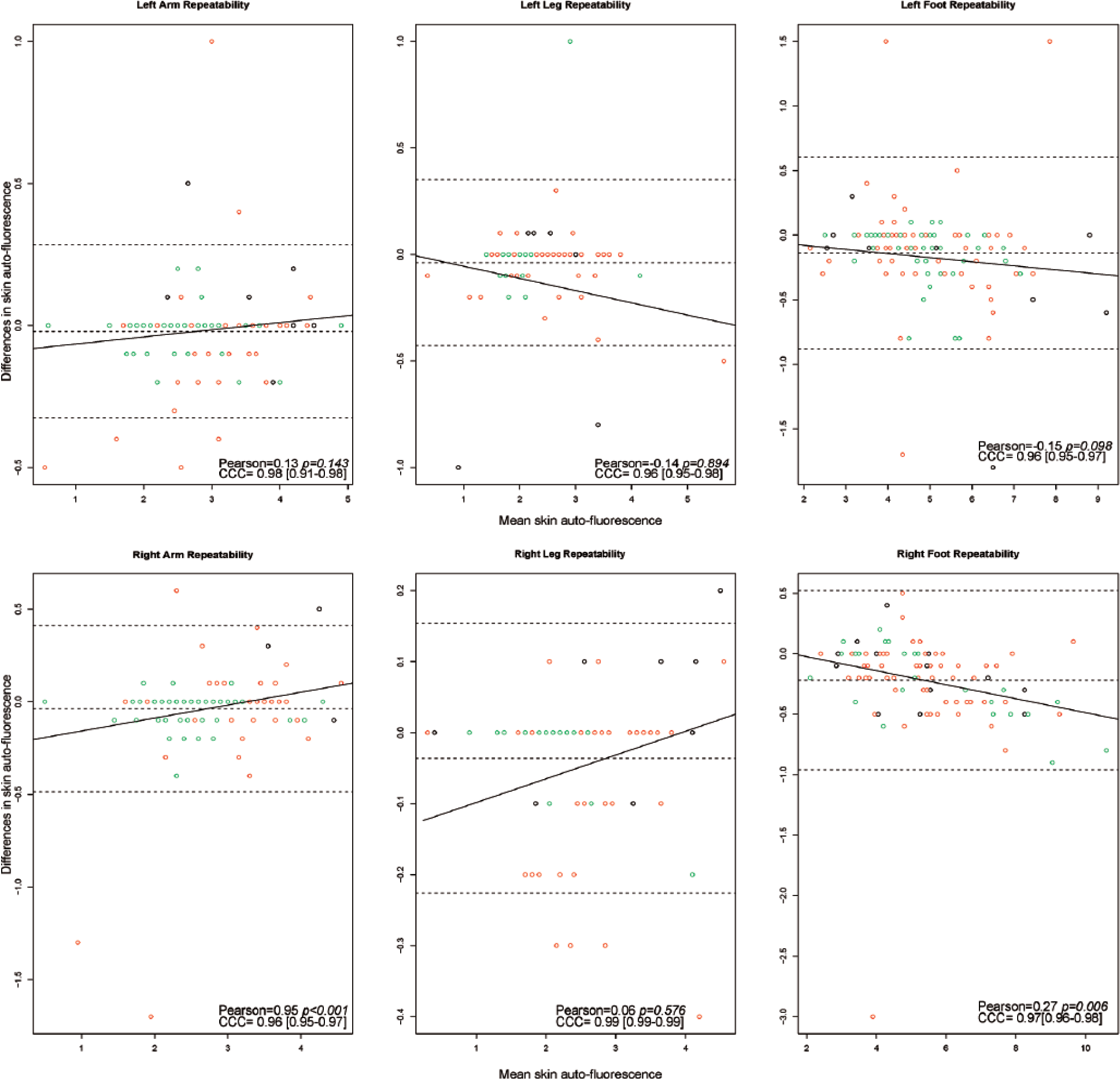
Bland-Altman plots for repeated measures at all six sites. All measurements represent skin autofluorescence measurements in AU. Figure outlines the Bland-Altman plots as represented by means on the x-axis and mean differences on the y-axis between measurement 1 and measurement 3 at each site. The black circles represent measurements in the DFU group (cases), the red circles indicate measurements in the DMC group (control), and the green circles represent measurements in the HC group. The middle dotted line represents the mean difference; the upper and lower dotted lines indicate ± 2 standard deviations from the mean difference (Bland-Altman limits of agreement). The regression line represents a linear regression of the differences and means. Pearson = pearson correlation coefficient; the reported
Agreement Between Left- and Right-Sided Measurements
The agreement based on CCC [95% CI] for each group is reported in Supplementary Table 3 and was moderate to substantial for the arms (0.80 [0.56-0.92] DFU to 0.86 [0.78-0.92] HC), poor to substantial for the legs (0.64 [0.25-0.85]) DFU to 0.82 [0.69-0.90]) HC), and poor to moderate for the feet (0.43 [0.23-0.59] HC to 0.71 [0.33-0.89] DFU). Post hoc analyses suggested that the presence of a DFU and the presence of DPN had minimal impact on right and left side between-site agreement (Supplementary Table 4). The Bland-Altman limits of agreement between measurements performed at right and left limbs are shown in Figure 3 stratified by group. The limits of agreement were larger for the feet (±2.5 arbitrary units) compared to the arms (±0.5 arbitrary units). The absolute mean differences (±SD) ranged from 0.03 (±0.56) to 0.13 (±0.33) arbitrary units for the DFU group and HC group for arm measurements and 0.13 (±2.54) to 0.79 (±2.83) arbitrary units for the HC group and DMC group for feet measurements.
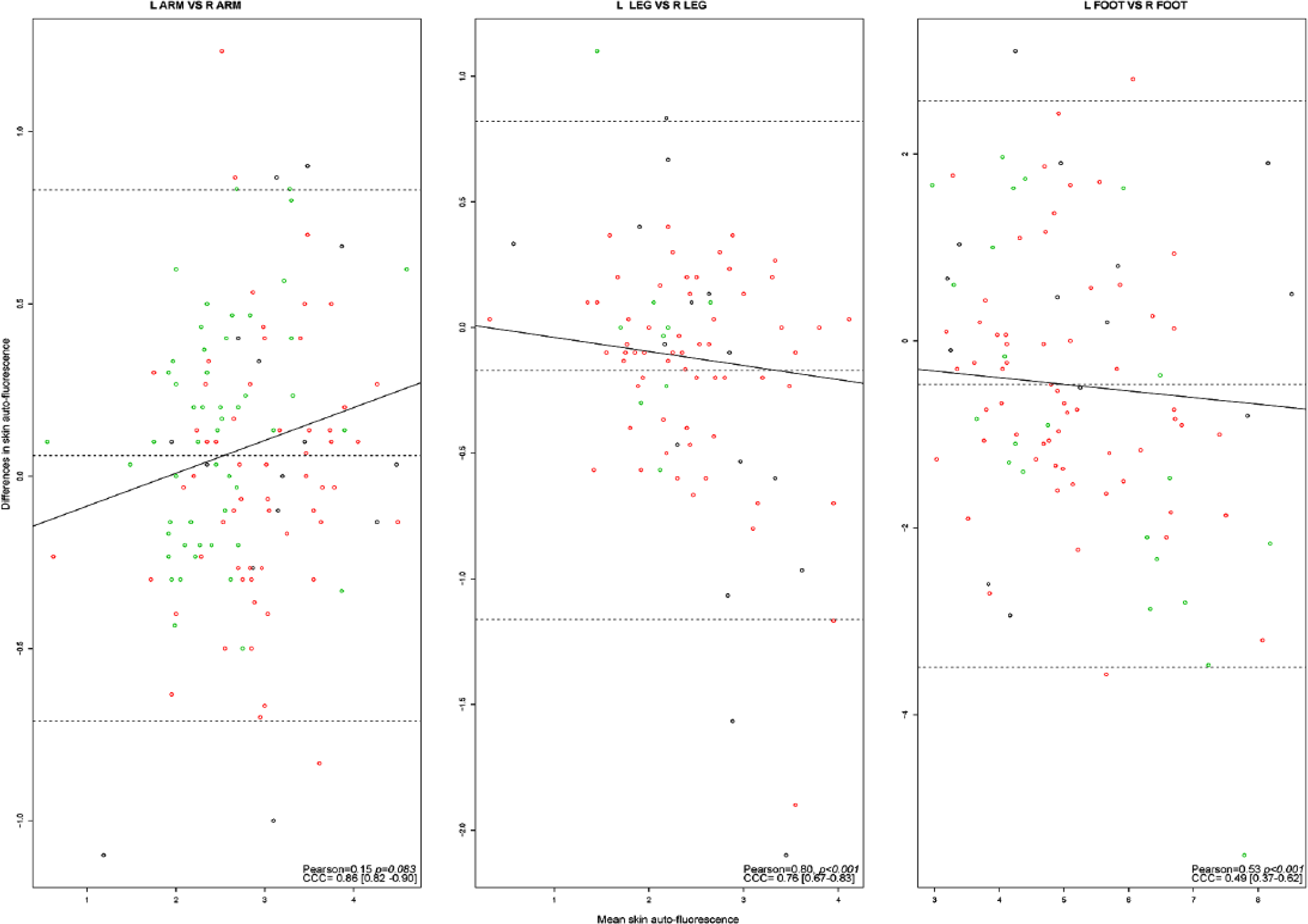
Bland-Altman plots for left and right measurement agreement. All measurements represent skin autofluorescence measurements in AU. Figure outlines the Bland-Altman plots as represented by means on the x-axis and mean differences on the y-axis between left- and right-sided measurements using averaged values from three measurements. The black circles represent measurements in the DFU group (cases), the red circles indicate measurements in the DMC group (control), and the green circles represent measurements in the HC group. The middle dotted line represents the mean difference; the upper and lower dotted lines indicate ± 2 standard deviations from the mean difference (Bland-Altman limits of agreement). The regression line represents a linear regression of the differences and means. Pearson = pearson correlation coefficient; the reported
Agreement Between Different Body Sites
Mostly there was poor between site agreement based on CCC [95% CI] and absolute mean differences (±SD) (Figure 4, Table 3). The Bland-Altman limits of agreement are shown in Figure 4 and indicated wider ranges for measurements performed in the feet compared to arms and measurements performed in the feet compared to the legs. There was moderate agreement between measurements performed in the left arm and right leg, and the right arm and right leg in the HC group (Table 3). The absolute mean differences were largest for measurements performed in the left leg and left foot in all three groups (3.40 [±2.04] arbitrary units DFU group; 3.15 [±2.45] arbitrary units DMC group; and 2.72 [±1.83] arbitrary units HC group).
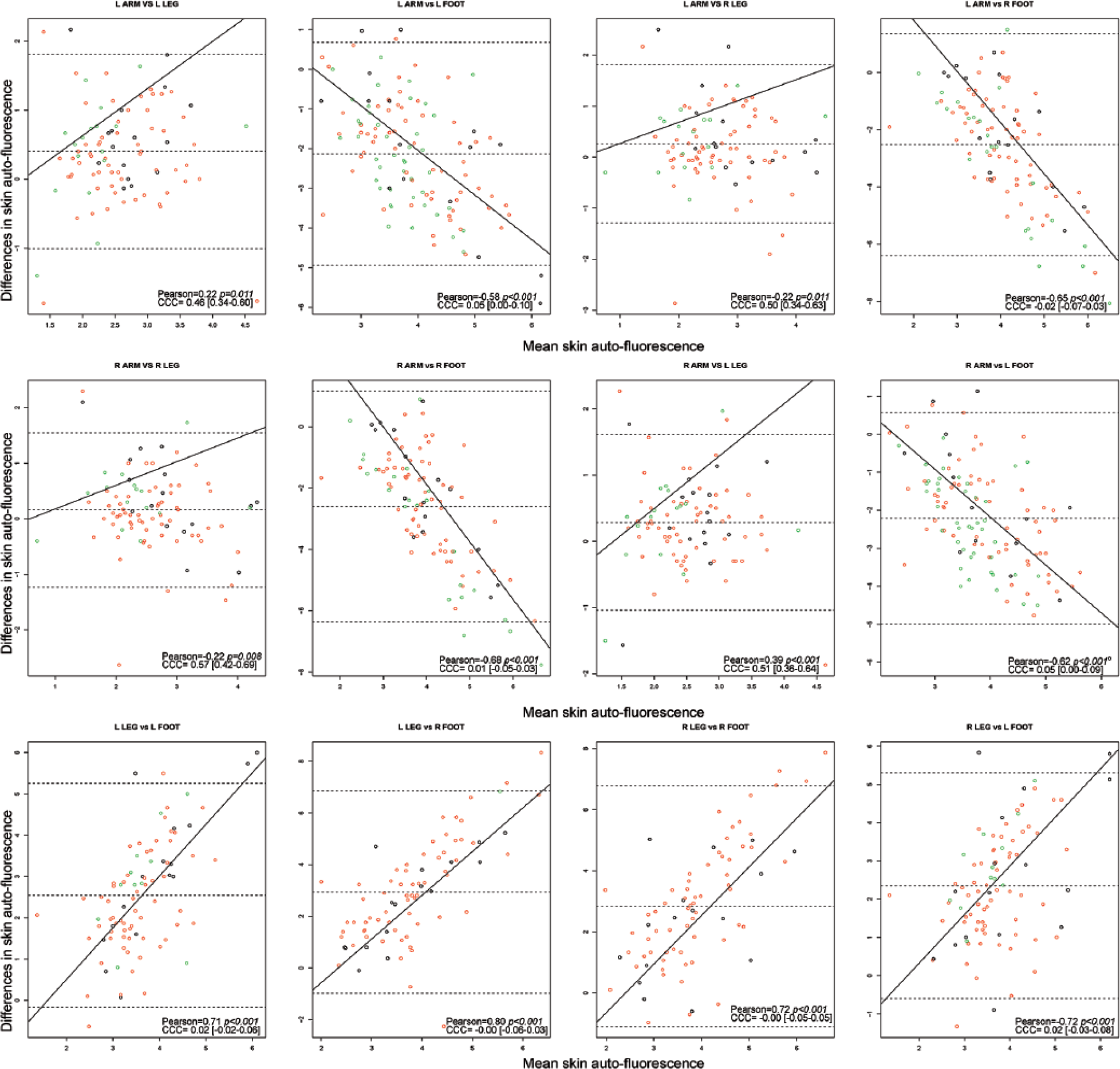
Bland-Altman plots for between site agreement. All measurements represent skin autofluorescence measurements in AU. Figure outlines the Bland-Altman plots as represented by means on the x-axis and mean differences on the y-axis between body sites. The black circles represent measurements in the DFU group (cases), the red circles indicate measurements in the DMC group (control), and the green circles represent measurements in the HC group. The middle-dotted line represents the mean difference; the upper and lower dotted lines indicate ± 2 standard deviations from the mean difference (Bland-Altman limits of agreement). The regression line represents a linear regression of the differences and means. Pearson = pearson correlation coefficient; the reported
Between Site Agreements in Measurements Stratified by Group.
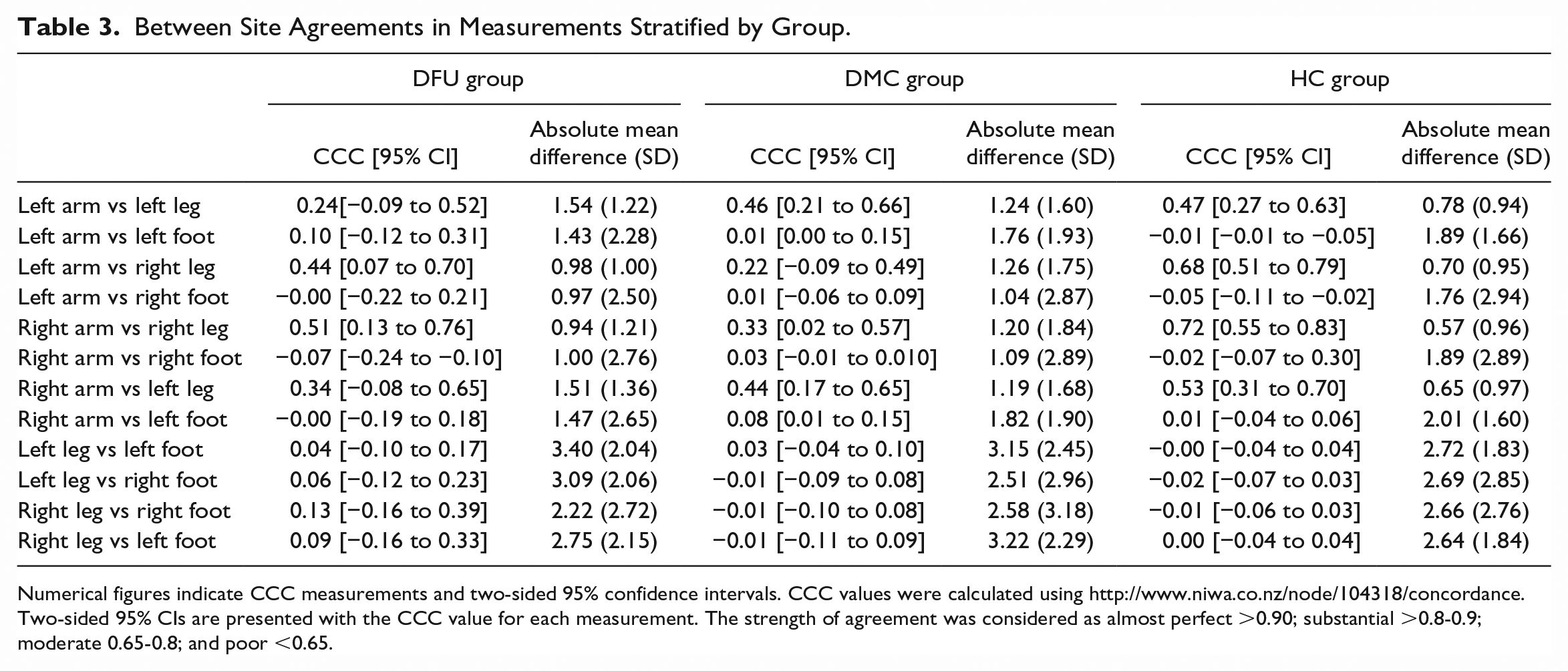
Numerical figures indicate CCC measurements and two-sided 95% confidence intervals. CCC values were calculated using http://www.niwa.co.nz/node/104318/concordance. Two-sided 95% CIs are presented with the CCC value for each measurement. The strength of agreement was considered as almost perfect >0.90; substantial >0.8-0.9; moderate 0.65-0.8; and poor <0.65.
Discussion
This is the first study to report the agreement between repeat skin autofluorescence measurements at the same and different body sites in a heterogeneous cohort. The agreement in measurements repeated at the same site were substantial to almost perfect in each group. The agreement between left- and right-sided measurements ranged from substantial to poor in the three groups, and the limits of agreement in measurements and absolute mean differences were larger for the feet compared to the arms. Overall there was poor agreement between measurements performed at the six different sites and the lack of between-site agreement seemed to increase with increased distance between sites. These findings suggest that there may be variation in glycation at different sites in the body, although other factors, such as skin translucency, may also explain the results. It is possible but unproven that the assessment of skin autofluorescence in the feet could provide a more accurate prediction of the risk of DFD.
Most previous studies have measured autofluorescence in the upper arm.32,41,48,49 The differences in measurements performed at different body sites were noted to be larger than differences previously reported between different patient populations with and without diabetes or DFD.19,22,32,50 This highlights the need for a consistent measurement site. Previous studies have also reported excellent levels of repeatability in skin autofluorescence readings consistently performed at the same site in the arm.8,51 Low coefficient of variations (6.9% and 10.9%) have previously been reported for skin autofluorescence measurements repeated 6 and 12 weeks apart in the arms of people with type 2 diabetes. 52 Variations in measurement technique, lack of consistency in the site of measurement, and biological variation over time were suggested to contribute to the measurement variability. 52 The current study expands on prior research by evaluating Bland-Altman plots and absolute agreement 53 and including the legs and feet as additional sites of measurement in both people at risk of developing DFD and people with active DFUs.
Skin autofluorescence readings in the feet were consistently higher than measurements in the legs and arms in all three groups. This finding is consistent with higher levels of glycation in the feet compared to more proximal sites, which could in part explain why DPN mostly affects the lower not upper limbs. 54 Since our findings indicate autofluorescence values differ substantially between different sides of the body, averaging values from left and right sides of the body is not recommended. The results of this study also suggest that the skin autofluorescence measured in the arm (the commonest site of measurement)19,26,28,55-58 are not necessarily representative of measurements at other body sites.
Different body sites have been reported to have different levels of fluorophores which may in part explain the findings of this study. Na and coworkers suggested a poor agreement between skin autofluorescence measurements performed at different sites could be due to differences in the levels of fluorophores within the skin. 34 Fluorophores such as nicotinamide adenine dinucleotide (NADH), flavin adenine dinucleotide (FAD), and porphyrins have an excitation spectrum similar to AGEs and differences in their concentration at different body sites may affect skin autofluorescence. 50 Although up to 76% of the variance in the skin autofluorescence signal has previously been correlated with different AGE concentrations, the AGE reader cannot distinguish between AGEs and other fluorophores in the same excitation spectrum. 41
Yamanaka and colleagues suggested that the presence of veins increases the skin autofluorescence measurements by 1.5-fold and therefore it is possible that variation in the distribution of veins at different body sites may explain some of the measurement variation. 49 Readings also likely depend on the depth of penetrance of light and the density of skin. 49 The skin on the plantar surface of the feet has a different tissue composition and thickness to the skin in the arms or legs. This likely affects the penetrance of the laser used to measure skin autofluorescence and may have contributed to the variation in measurements.
The use of multiple analysis methods including Bland-Altman plots with limits of agreement and absolute mean differences are a strength of this study.47,53 Another strength was the inclusion of a heterogeneous group of people considered to have a wide spectrum of skin autofluorescence measurements and the stratification of the cohort based on group. The age of participants in the DMC group and HC group and the duration of diabetes in the DFU and DMC groups differed and both are known to impact skin autofluorescence readings. 21 Therefore we evaluated measurement variation in the different participant groups.
The main limitation of this study was the convenience and heterogeneous sample. Skin autofluorescence readings for the lower limb could not be obtained from up to 37% of our cohort and the reasons for this were unclear. Patients with missing data did not have unique characteristics as compared to the overall cohort and the AGE reader data obtained in our study was comparable to other studies in the field.19,32,50 The study was cross-sectional, and the investigation did not evaluate between-day measurement variability or the relationship between skin autofluorescence and circulating AGEs or HbA1c.56,59 Last, the focus of this study was not to compare skin autofluorescence between patients that did or did not have DFD. The study was not adequately powered for this comparison that has been previously reported in prior studies.19,57 Furthermore, this study measured skin autofluorescence, not AGE concentrations, and therefore it is uncertain how the variation of autofluorescence reported here relates to AGE distributions throughout the body.
Conclusion
Skin autofluorescence measurements repeated at the same body site had good agreement whereas agreement between measurements performed at different body sites was poor. Further studies are required to determine the reasons for this variation and the ideal site to perform measurements in people with DFD.
Supplemental Material
Supplementary_Files – Supplemental material for Within- and Between-Body-Site Agreement of Skin Autofluorescence Measurements in People With and Without Diabetes-Related Foot Disease
Supplemental material, Supplementary_Files for Within- and Between-Body-Site Agreement of Skin Autofluorescence Measurements in People With and Without Diabetes-Related Foot Disease by Malindu E. Fernando, Robert G. Crowther, Peter A. Lazzarini, Kunwarjit S. Sangla, Scott Wearing, Petra Buttner and Jonathan Golledge in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors wish to thank all the participants of the study and the administrative and academic staff at the Movement Analysis Laboratory, Sports and Exercise Science, James Cook University, Townsville, Australia, for assistance with study visits and the utilization of space to conduct the study.
Abbreviations
ABPI, ankle brachial pressure index; AGE, advanced glycation end product; BMI, body mass index; CCC, concordance correlation coefficient; CI, confidence interval; DFD, diabetes-related foot disease; DFU, diabetes foot ulcer; DMC, diabetes mellitus control; DPN, diabetic peripheral neuropathy; eGFR, estimated glomerular filtration rate; HC, healthy control; IQR, interquartile range; MNSI, Michigan Neuropathy Screening Instrument; PAD, peripheral artery disease; RAGE, receptor for advanced glycation end products; SD, standard deviation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding from the Department of Health, Queensland Government under the Health Practitioner Research Grant Scheme (2013-2014), funding from the Graduate Research School and Strategic Research Investment Fund for The Ulcer and Wound Healing Consortium (UHEAL) at James Cook University, and funding from the National Health and Medical Research Council supported this work. JG holds a Practitioner Fellowship from the National Health and Medical Research Council, Australia (1117061). JG holds a Senior Clinical Research Fellowship from the Queensland Government. MEF was supported by an Australian Postgraduate Award Scholarship at James Cook University and a scholarship from College of Medicine, James Cook University at the time of the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
