Abstract
Background:
This comparative handling study investigated user satisfaction and insulin pump handling with a prefilled insulin cartridge versus a self-filled insulin reservoir in insulin pump users with type 1 diabetes (T1D).
Methods:
Adult (n = 105) and adolescent (n = 25) participants performed insulin pump preparations using a prefilled insulin cartridge and self-filled insulin reservoir. User satisfaction, insulin pump preparation time, and residual air in infusion set tubing were assessed for each insulin filling method. Post hoc analysis evaluated training time.
Results:
User satisfaction scores were statistically significantly different in favor of the prefilled insulin cartridge versus the self-filled insulin reservoir (mean [SD]: overall, 4.0 [0.5] vs 3.3 [0.9]; burden on the user, 1.8 [0.6] vs 2.9 [1.0]; user inconvenience, 2.0 [0.7] vs 2.8 [1.1]; device effectiveness, 3.9 [0.7] vs 3.6 [0.9]; all P < .001). Insulin pump preparation time and residual air measurements were significantly different and numerically lower for the prefilled insulin cartridge versus the self-filled insulin reservoir (mean [SD]: preparation time, 124.4 [30.3] vs 237.8 [64.2] seconds, P < .001; residual air, 2.3 [26.3] vs 10.0 [63.3] mm, P = .007). Training time was shorter with the prefilled insulin cartridge versus the self-filled insulin reservoir (mean [min; max]: 193.1 [36; 453] vs 535.8 [124; 992] seconds).
Conclusions:
Adult and adolescent insulin pump users were more satisfied with the prefilled insulin cartridge versus the self-filled insulin reservoir when preparing an insulin pump. The prefilled insulin cartridge was associated with reduced insulin pump preparation time and reduced training time versus the self-filled insulin reservoir.
An increasing number of people with diabetes, especially type 1 diabetes (T1D), use insulin pump technology to deliver continuous subcutaneous insulin infusion (CSII). Insulin pumps can be programmed to deliver basal rates of insulin around the clock, with on-demand bolus dosing functionality to cover meals and correct for high blood glucose levels. Furthermore, CSII treatment integrated with continuous glucose monitoring (CGM) can be associated with significant improvements in glycemic control, hypoglycemia risk, and quality of life.1,2
The majority of available insulin pumps deliver insulin from a self-filled reservoir inside the pump to a patient’s body via an infusion set and cannula. Many insulin pumps integrate an automatic insulin bolus calculator and communicate wirelessly with CGM. Improvements in insulin pump technology, such as tubeless patch pumps 3 and sensor-driven adjusted insulin delivery, 4 make insulin pump use more convenient, and help to reduce the burden of diabetes treatment.
However, patients with T1D perform many tasks related to their treatment, and complexities around insulin pump preparation largely remain unchanged. Patients need to change and fill the insulin reservoir and change the infusion set. These procedures involve manually filling the reservoir from a vial of insulin using a transfer adapter or a needle and syringe, inserting the reservoir into the insulin pump, and priming and attaching the infusion set. In a large survey of insulin pump users, 29% described the number of steps involved in changing the reservoir as “a few too many” to “entirely too many,” and 36% described the number of steps involved in filling the insulin reservoir as “a few too many” to “entirely too many.” 5
Prefilled insulin cartridges have been developed to simplify the insulin pump preparation procedure.6-8 In a comparison of two insulin pump devices by individuals treated with multiple daily injection therapy, the insulin pump with a prefilled insulin cartridge was rated as more usable and preferred for a number of pump tasks, including refilling the insulin. 9 In a small user experience study performed under everyday conditions, the majority of patients reported that the prefilled insulin cartridge was a clear advantage of a novel pump system (Figure 1). 7 Anecdotal evidence reported from users and health care professionals to the authors also suggests that patients are in favor of prefilled insulin cartridges. This study aimed to formally investigate user satisfaction and insulin pump handling with a prefilled insulin cartridge versus a self-filled insulin reservoir in insulin pump users with T1D.

Accessory equipment required for filling an insulin pump using the self-filled insulin reservoir (left) and the prefilled insulin cartridge (right). The insulin pump preparation procedure has traditionally included manually filling the insulin reservoir using an insulin vial and transfer adapter set, inserting the insulin reservoir into the pump, and attaching and priming the infusion set. Users are also required to wipe the top of the insulin vial with alcohol before filling the reservoir. The prefilled insulin cartridge is a one-step loading device that is inserted directly into the pump. Vial image used under license ©2018 Sveta / stock.adobe.com. Alcohol wipes image used under license ©2018 Duplass / Shutterstock.com. Insulin pump and prefilled insulin cartridge images reproduced with permission of Ypsomed AG, Burgdorf, Switzerland.
Methods
Study Design and Participants
In this multicenter, one-day handling study, participants with T1D compared the use of a prefilled insulin cartridge (PumpCart®, Novo Nordisk A/S, Søborg, Denmark) and a self-filled insulin reservoir (mylife™ YpsoPump® Reservoir, Ypsomed AG, Burgdorf, Switzerland) when preparing an insulin pump (mylife YpsoPump). The protocol was reviewed and approved by the Allendale Investigational Review Board (Old Lyme, CT, USA), and the study was conducted in the United Kingdom.
Participants with T1D (adolescents 12-17 years; adults ≥18 years) were required to have at least 6 months of experience self-administering insulin using an insulin pump, and be able to prepare their insulin pump cartridge and infusion set tubing independently. Participants were also required to be fluent in English, and be able to read and understand written English. All participants provided written informed consent.
Exclusion criteria included working in the field of human factors, market research, or medical device development; a personal or family connection to a pharmaceutical company; self-reported participation in market research or usability testing of insulin cartridges within the past year; mental or extreme physical incapacity; or a visual, hearing, or dexterity impairment (to the extent that the participant is unable to read a newspaper [corrected vision was acceptable], listen to the television at the same volume as others [correction with hearing aids was acceptable], or operate a mobile phone or TV remote).
Study Procedures
All participants received a one-on-one training session lasting up to 30 minutes with an experienced insulin pump trainer immediately prior to participating in a single test session. Training included instruction on basic pump functionality and insulin pump preparation practice using the self-filled insulin reservoir and the prefilled insulin cartridge.
During the test session, participants were asked to perform three insulin pump preparations with the prefilled insulin cartridge and three insulin pump preparations with the self-filled insulin reservoir, each up to the point that the infusion set was ready to be attached to the cannula base on the skin. Participants alternated between the two insulin-filling methods, with approximately half of the participants performing the first preparation with the prefilled insulin cartridge and the other half performing the first preparation with the self-filled insulin reservoir. When participants considered the preparation procedure to be complete, they handed the insulin pump and the attached infusion set tubing to a test administrator for evaluation.
Participants performed insulin pump preparations while seated at a table with a test administrator and data analyst. Lighting and noise levels were comparable to those of a relatively quiet home environment, or a physician’s office. Participants used insulin at room temperature (removed from the refrigerator at least 30 minutes before the test session). A parent or guardian could accompany adolescent participants and remain present during the training and test sessions. Training and test sessions were recorded using a video camera.
Assessments
The primary objective of the study was to investigate user satisfaction. Participants assessed satisfaction after the last preparation with each insulin filling method using the Insulin Delivery Satisfaction Survey (IDSS) for T1D. 10 The IDSS provided an overall satisfaction score based on 14 statements about the insulin delivery system, and three subscores based on statements relating to burden on the user (five statements), user inconvenience (four statements), and device effectiveness (five statements). Participants rated each statement on a 5-point scale (1 = “strongly disagree” to 5 = “strongly agree”).
Secondary objectives were to measure the amount of time participants spent preparing each insulin pump, and the amount of residual air remaining in the infusion set tubing after each insulin pump preparation. Preparation time and residual air measurements were obtained for the second and third insulin pump preparations with each insulin filling method. Preparation time was measured from video recordings of the study. Test administrators visually inspected the infusion set tubing for the presence of residual air and assessed the size of air bubbles using measurement scales. Any adverse events or technical complaints were also recorded.
Post hoc exploratory analyses evaluated the duration of training the participants received. Measurements of total training time and time spent specifically training the participant on using the self-filled insulin reservoir and prefilled insulin cartridge were obtained from video recordings.
Statistical Analysis
Satisfaction scores, residual air measurements, and preparation and training time were analyzed using descriptive statistics. The paired satisfaction scores and residual air measurements were analyzed using a nonparametric Wilcoxon signed-rank test for paired data. Mean preparation time was analyzed using a paired t-test. A P value of less than or equal to .05 was considered statistically significant.
Results
Participant Demographics
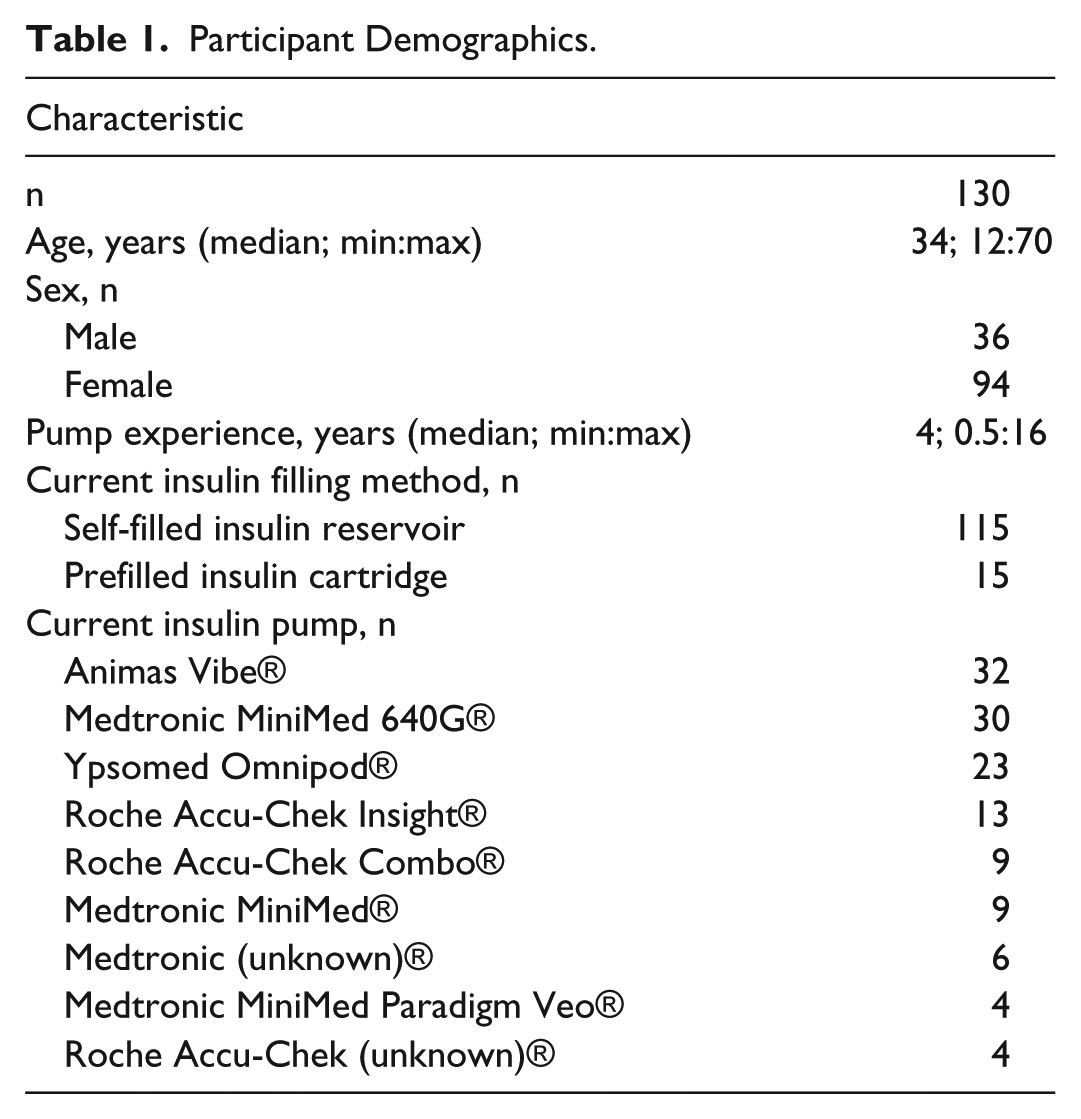
In total, 134 participants attended a training session and 130 participants completed the study. Four participants were disqualified for not meeting the inclusion criteria: two participants did not have any insulin pump experience, one participant had <6 months of insulin pump experience, and one participant was deemed unable to perform insulin pump preparations independently (based on the trainer’s professional judgement). Of the 130 participants who completed the study, 25 participants were adolescents (ages 12-17) and 15 participants were currently using prefilled insulin cartridges (with Roche Accu-Chek® insulin pump systems [Roche Diabetes Care GmbH, Basel, Switzerland]). Participant demographic information is presented in Table 1. Two participants did not perform all six insulin pump preparations due to time limitations. As such, the analysis only included data obtained after these participants’ second insulin pump preparation with each insulin filling method.
Participant Demographics.
Satisfaction Scores
Overall satisfaction scores were statistically significantly different and numerically higher for insulin pump preparations using the prefilled insulin cartridge versus the self-filled insulin reservoir (mean [SD]: 4.0 [0.5] vs 3.3 [0.9]; P < .001) (Figure 2, Table 2). The three subscores related to burden on the user, user inconvenience, and device effectiveness were all statistically significantly in favor of the prefilled insulin cartridge versus the self-filled insulin reservoir (burden on the user, 1.8 [0.6] vs 2.9 [1.0]; user inconvenience, 2.0 [0.7] vs 2.8 [1.1]; device effectiveness, 3.9 [0.7] vs 3.6 [0.9]; all P < .001) (Figure 3, Table 2).
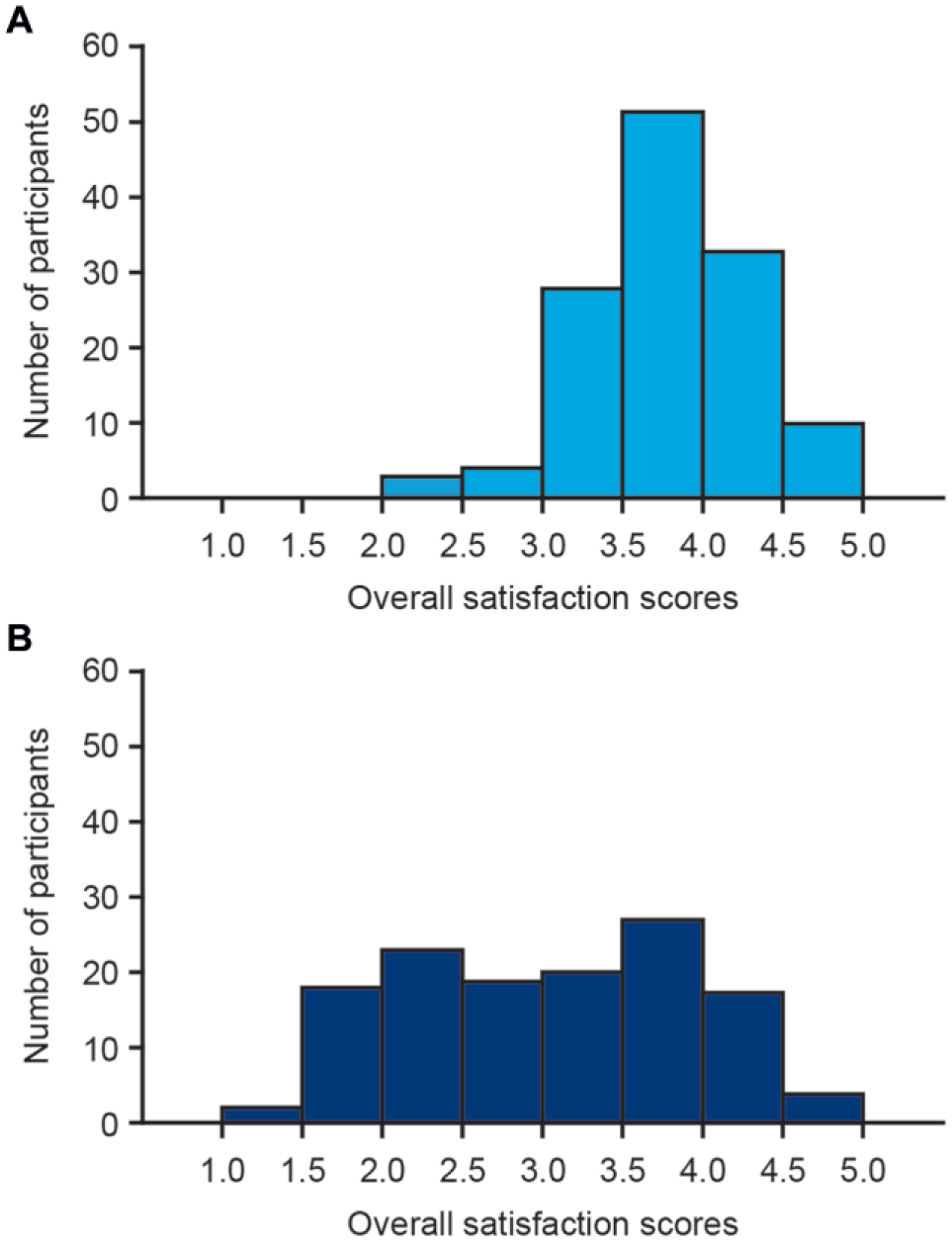
Frequency distribution of participants’ overall satisfaction scores for insulin pump preparation using the prefilled insulin cartridge (A) and the self-filled insulin reservoir (B). Overall satisfaction scores were calculated based on the rating of all 14 statements in the IDSS completed after the last insulin pump preparation with each insulin filling method. A score of 1 reflects the lowest reportable level, and a score of 5 reflects the highest reportable level. Two participants completed the IDSS after performing the second preparation with each insulin filling method.
Overall Satisfaction Scores and Scores Relating to Device Effectiveness, Burden on the User, and User Inconvenience.
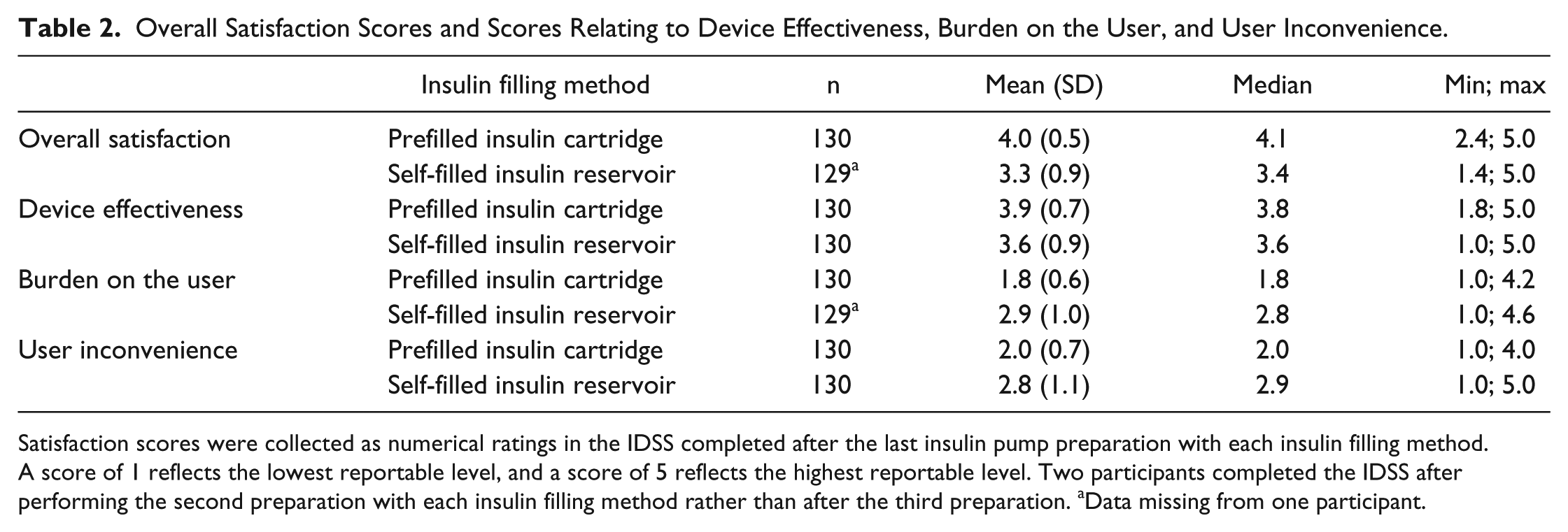
Satisfaction scores were collected as numerical ratings in the IDSS completed after the last insulin pump preparation with each insulin filling method. A score of 1 reflects the lowest reportable level, and a score of 5 reflects the highest reportable level. Two participants completed the IDSS after performing the second preparation with each insulin filling method rather than after the third preparation. aData missing from one participant.
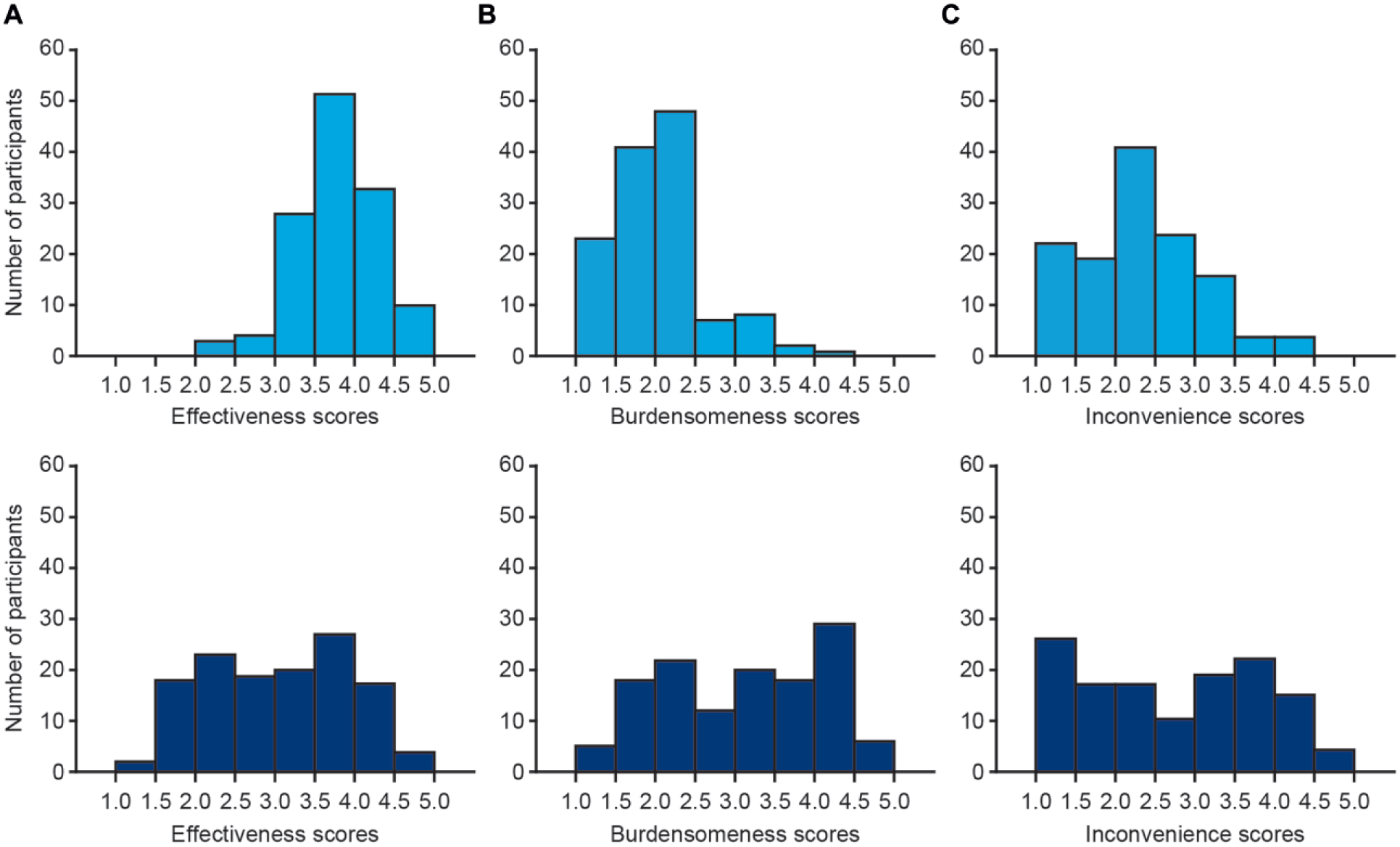
Frequency distribution of participants’ satisfaction scores relating to device effectiveness (A), burden on the user (B), and user inconvenience (C) for insulin pump preparation using a prefilled insulin cartridge (light blue, top) and a self-filled insulin reservoir (dark blue, bottom). Subscores were calculated based on the rating of statements relating to burden on the user, user inconvenience, and device effectiveness in the IDSS completed after the last insulin pump preparation with each insulin filling method. A score of 1 reflects the lowest reportable level, and a score of 5 reflects the highest reportable level (for burden on the user and user inconvenience, a score of 1 reflected the “best” response and 5 reflected the “worst”). Two participants completed the IDSS after performing the second preparation with each insulin filling method.
Preparation Time
Preparation time was statistically significantly different and numerically lower with the prefilled insulin cartridge versus the self-filled insulin reservoir (P < .001). The mean (SD) preparation time was 124.4 (30.3) seconds (median 119.3 seconds [min 70.5; max 296.0]) or 2.1 [0.5] minutes (median 2.0 minutes [min 1.2; max 4.7]) with the prefilled insulin cartridge versus 237.8 (64.2) seconds (median 229.8 [min 118.5; max 534.0]) or 4.0 (1.1) minutes (median 3.8 [min 2.0; max 8.9]) with the self-filled insulin reservoir (Figure 4).
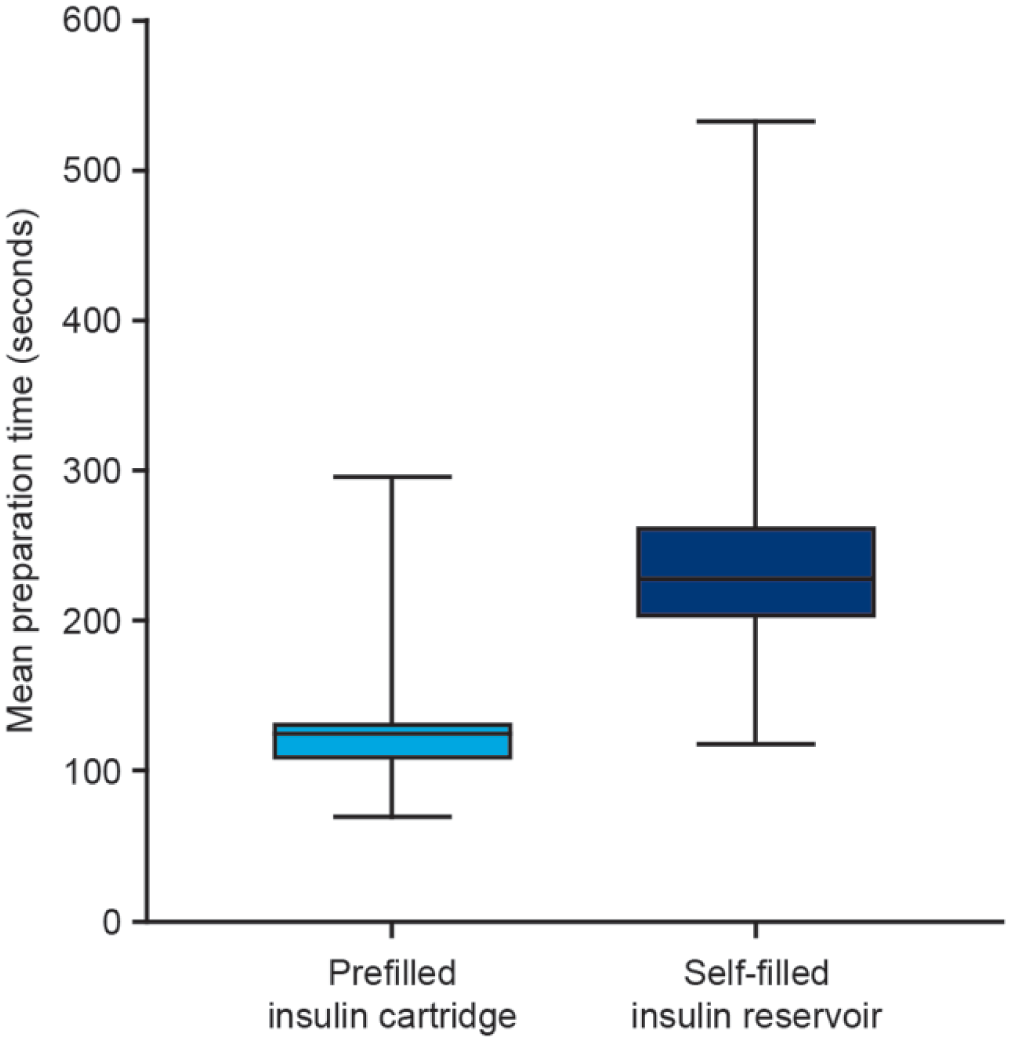
Preparation time for the prefilled insulin cartridge and self-filled insulin reservoir. Preparation time is based on the second and third insulin pump preparations with each insulin filling method. Box = 25th and 75th percentiles; bars = minimum and maximum values.
Residual Air in Infusion Set Tubing
The majority of participants had no residual air in the infusion set tubing after insulin pump preparations with either insulin filling method (129/130 with the prefilled insulin cartridge and 122/130 with the self-filled insulin reservoir). While the number of participants with residual air in the infusion set tubing was low, overall there was a significantly different and numerically lower amount of residual air in the insulin set tubing after insulin pump preparations with the prefilled insulin cartridge than with the self-filled insulin reservoir (P = .007). The mean (SD) amount of residual air was 2.3 (26.3) mm (median 0.0 mm [min 0.0; max 300.0]) with the prefilled insulin cartridge versus 10.0 (63.3) mm (median 0.0 mm [min 0.0; max 587.5]) with the self-filled insulin reservoir.
Technical Assistance
Five of the 130 participants required test administrator assistance while performing the insulin pump preparations: one with the prefilled insulin cartridge and four with the self-filled insulin reservoir.
Technical Complaints
No technical complaints were reported.
Training Time
In the post hoc analysis, training time was assessed for the 118 participants who were allowed to practice the insulin pump preparation procedure as many times as required within their 30 minutes of allotted time (training as needed). Training time was also assessed for the first 10 participants who received only one opportunity to practice with each filling method. Two participants were excluded from the post hoc analysis due to missing training videos.
For participants who received training as needed, the mean (SD) training time spent orienting the participant to the session, discussing goals of the training, and training on basic pump functionality and use of the self-filled insulin reservoir and the prefilled insulin cartridge was 1278.0 (282.0) seconds (min 480; max 1860) or 21.3 (4.7) minutes (min 8; max 31). The mean (SD) time spent specifically training participants in each insulin filling method was 342.7 seconds (5.7 minutes) shorter with the prefilled insulin cartridge versus the self-filled insulin reservoir (193.1 [72.4] seconds [min 36.0; max 453.0] or 3.2 [1.2] minutes [min 0.6; max 7.6] versus 535.8 [143.6] seconds [min 124.0; max 992.0] or 8.9 [2.4] minutes [min 2.1; max 16.5]). Individual participants’ training times for the two filling methods are shown in Figure 5.
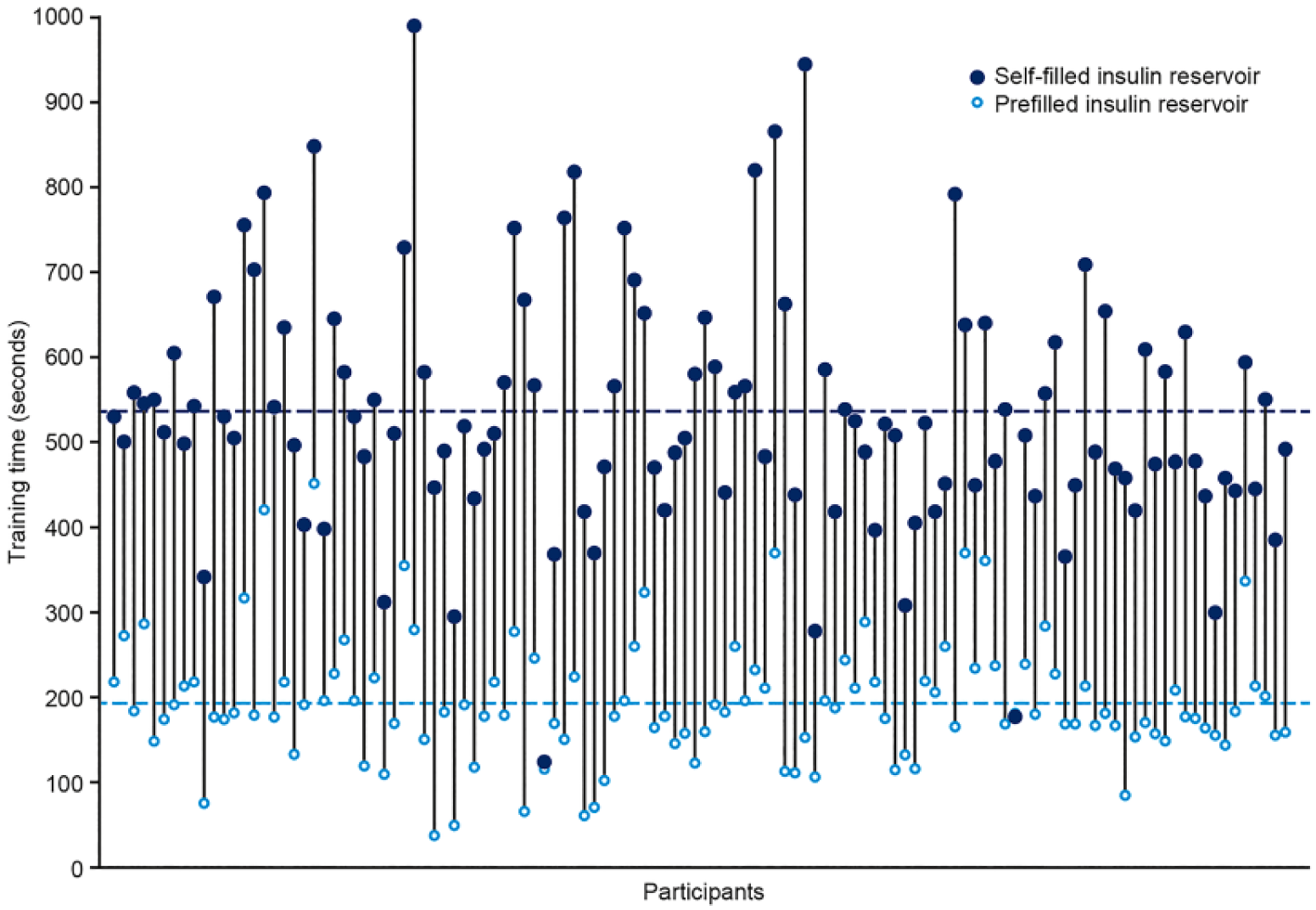
Training times for individual participants being specifically trained in the use of the prefilled insulin cartridge and the self-filled insulin reservoir. Training time for participants who were allowed to practice the insulin pump preparation procedure as many times as required within their 30 minutes of allotted time. Dashed lines represent mean training time with each insulin filling method.
The mean training times for the 10 participants with limited training opportunities were considerably lower than for participants receiving training as needed. However, the results followed the same pattern, with a shorter mean training time with the prefilled insulin cartridge (mean [SD]: 38.5 [22.9] seconds [min 10.0; max 72.0] or 0.6 [0.4] minutes [min 0.2; max 1.2]) versus the self-filled insulin reservoir (mean [SD] 296.7 [97.4] seconds [min 195.0; max 480.0] or 4.9 [1.6] minutes [min 3.3; max 8.0]).
Discussion
Adult and adolescent insulin pump users with T1D reported greater satisfaction with the use of a prefilled insulin cartridge versus a self-filled insulin reservoir when preparing an insulin pump. This is consistent with a recently published study, in which experienced insulin pump users reported that the prefilled insulin cartridge was an advantage of a new insulin pump system. 7
Participants spent less time preparing the insulin pump with the prefilled insulin cartridge versus the self-filled insulin reservoir. While the majority of participants had no residual air in the infusion set tubing after insulin pump preparations with either insulin filling method, overall participants had marginally less residual air in the infusion set tubing with the prefilled insulin cartridge versus the self-filled insulin reservoir. In addition, post hoc analysis revealed that a shorter time was spent training participants with the prefilled insulin cartridge versus the self-filled insulin reservoir. Together, these findings suggest that the prefilled insulin cartridge is easier to handle and learn to use compared with a self-filled insulin reservoir, and simplifies the insulin pump preparation procedure.
Modifications to insulin pump systems that improve user satisfaction and ease of use, such as prefilled insulin cartridges, may impact treatment adherence and outcomes. Indeed, higher IDSS scores are significantly associated with better glycemic control and greater adherence to insulin treatment and insulin pump use, 10 and perceptions of usability have been shown to positively influence attitudes toward insulin pump therapy. 9
Systems that are easy to handle, with fewer preparation steps, may limit use errors and improve patient safety. 11 In addition, without the need for an insulin vial and a transfer adapter, or needle and syringe, to fill the insulin reservoir, the use of prefilled insulin cartridges reduces the need to carry accessory equipment as well as the risk of needle-stick injuries. However, prefilled insulin cartridges are made from glass and are more fragile than the plastic self-filled insulin reservoirs used in most insulin pumps. Insulin may stay more stable in glass rather than plastic cartridges, although there may be a higher risk of glass cartridges breaking in the pump.
One limitation of the study was the artificial setting. Insulin pump preparations were performed in a research facility that simulated a real-world setting as closely as possible. The insulin was used at room temperature to reduce air bubble formation, and the insulin pump was prepared under light and noise conditions similar to those in a home or physician’s office. However, the test environment may have had an impact on participants’ handling performance. Participants may also have a preference bias toward the insulin filling method they currently use. The crossover design of the study aimed to minimize this bias. However, any remaining bias would favor the self-filled insulin reservoir over the prefilled insulin cartridge as only 15 participants were currently using prefilled insulin cartridges.
It should also be emphasized that the study investigated the impact of the different insulin filling methods up until mounting the insulin pump system to a cannula base. The study did not assess the clinical implications of greater patient satisfaction, reduced user burden/inconvenience, or shorter insulin pump preparation time, and did not monitor for use errors. Further trials are needed to assess whether the benefits related to the use of a prefilled insulin cartridge are translated into continued insulin pump use, improved treatment adherence, and better blood glucose control.
Conclusion
This comparative handling study demonstrates that adult and adolescent insulin pump users with T1D were more satisfied with the use of a prefilled insulin cartridge versus a self-filled insulin reservoir when preparing an insulin pump. Compared with a self-filled insulin reservoir, the use of the prefilled insulin cartridge was also associated with reduced insulin pump preparation time and reduced training time.
Footnotes
Acknowledgements
The authors would like to acknowledge Ypsomed AG, Switzerland, for the use of the mylife YpsoPump in the study, Imke Silderhuis (UL International [Netherlands] BV), Tricia Gibo (UL International [Netherlands] BV), and Limor Hochberg (former UL LLC) for data analysis. Medical writing and editorial support were provided by Helen Parker PhD and Erin Slobodian of Watermeadow Medical, an Ashfield Company, part of UDG Healthcare plc, funded by Novo Nordisk A/S.
Abbreviations
CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; IDSS, Insulin Delivery Satisfaction Survey; min, minimum; max, maximum; SD, standard deviation; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JG, GSF, MLJ, and TS are employees of and hold shares in Novo Nordisk. MJ is an employee of UL LLC, and BG is an employee of UL International (Netherlands) BV.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Novo Nordisk A/S.
Prior Publication
Some of the data in this manuscript have been presented in poster form at the International Society for Pediatric and Adolescent Diabetes, 44th Annual Meeting (ISPAD 2018), 11-14 October, Hyderabad, India.
