Abstract
Hyperglycemia is common in the intensive care unit (ICU) both in patients with and without a previous diagnosis of diabetes. The optimal glucose range in the ICU population is still a matter of debate. Given the risk of hypoglycemia associated with intensive insulin therapy, current recommendations include treating hyperglycemia after two consecutive glucose >180 mg/dL with target levels of 140-180 mg/dL for most patients. The optimal method of sampling glucose and delivery of insulin in critically ill patients remains elusive. While point of care glucose meters are not consistently accurate and have to be used with caution, continuous glucose monitoring (CGM) is not standard of care, nor is it generally recommended for inpatient use. Intravenous insulin therapy using paper or electronic protocols remains the preferred approach for critically ill patients. The advent of new technologies, such as electronic glucose management, CGM, and closed-loop systems, promises to improve inpatient glycemic control in the critically ill with lower rates of hypoglycemia.
Keywords
Hyperglycemia is common in the intensive care unit (ICU) being present in approximately 80% of the patient population, and affecting both patients with and without a previous diagnosis of diabetes.1,2 It has been well established that hyperglycemia is associated with an increased rate of complications including infections, prolonged stay, and mortality in the ICU, with the higher risk observed in patients without a history of diabetes (new-onset and stress-induced hyperglycemia).1-6 In the decades of the 1990s and early 2000s, prospective randomized controlled studies showed that intensive insulin therapy used to achieve strict glycemic control in the ICU led to decreased morbidity and mortality compared to conventional therapy.7-10 These results prompted wide adoption of intensive insulin therapy and tight glycemic control protocols for the management of hyperglycemia in the ICU. However, since the NICE-SUGAR trial and several other studies that showed increased mortality risk associated with intensive glucose control (likely due to hypoglycemia),11,12 the approach of strict glycemic control in the ICU for all patients has been discouraged, and moderate control allowing for a certain degree of hyperglycemia has been promoted instead.13,14 To this date, challenges for achieving adequate glycemic control still exist. Despite having significant accuracy and interference errors, 15 point of care (POC) glucose monitoring remains the most common method for glucose monitoring in the ICU. In addition, multiple paper protocols for guiding the administration of intravenous insulin are available, yet risk of hypoglycemia is still an important limitation of their use. 16
Nonetheless, new technologies such as electronic glucose management, continuous glucose monitoring, and closed-loop systems, have shown promise to safely achieve adequate glycemic control with low rates of hypoglycemia.17-22
Insulin Therapy and Glycemic Targets
Several medical organizations, including the American Diabetes Association (ADA), Society of Critical Care (SCCM), American College of Physicians (ACP), and European Society of Intensive Care Medicine (ESICM) have published consensus statements for glycemic control in the ICU.23-25
The current recommendations include starting insulin management for two consecutive glucose readings >180 mg/dL with target levels of 140-180 mg/dL. Treatment should avoid hyperglycemia >180 mg/dL. Intensive insulin therapy (IIT) with stricter ranges of 110-140 mg/dL is only appropriate for selected patients if this can be achieved without significant hypoglycemia. 23 A meta-analysis of 26 trials involving a total of 13 567 patients, including the NICE SUGAR study, found that intensive insulin therapy was associated with a mortality benefit, but only in the surgical ICU population. 26
The optimal glucose range in the ICU population is still a matter of debate, and probably differs for different patient populations. 15 Certain patients likely benefit from stricter glucose control, such as those with traumatic brain injury, post cardiac surgery, myocardial infarction, liver transplant, cerebrovascular ischemic injury, aneurysmal subarachnoid hemorrhage, end stage liver and kidney disease, and high dose glucocorticoid management.16-22,27 However, most of the clinical studies showing morbidity or mortality benefit from IIT come from the cardiovascular surgical ICU population.7,10,28 Most recently, in a study conducted by Umpierrez et al, an electronic glucose management system was used to facilitate glycemic control, and patients with diabetes (n = 152) and without diabetes with hyperglycemia (n = 150) were randomized to an intensive glucose target of 100-140 mg/dL (n = 151) or to a conservative target of 141-180 mg/dL (n = 151) after coronary artery bypass grafting (CABG) surgery. 29 Although results of this study showed a trend suggesting benefits of intensive glycemic control, no statistically significant differences were found in the composite outcome of perioperative complications between groups. Notably, in a subgroup analysis of the same study, a significantly lower number of complications were found in the group of patients without diabetes treated to the intensive target, but not in patients with diabetes. Importantly, rates of hypoglycemia were low and not significantly different between the intensive and conservative groups (8% vs 3%, P = .09). In agreement with these findings, a recent study of patients admitted to the ICU with a HbA1c <7% showed decreased mortality when glucose was controlled between 100-140 mg/dL, but not in patients with HbA1c >7%. 30 Similarly, other studies have also shown that patients without diabetes show increased morbidity and mortality with liberal glucose management strategies.31,32
Hypoglycemia and Glycemic Variability
Hypoglycemia and glycemic variability have been both identified as strong predictors of increased morbidity and mortality in the ICU. In the NICE-SUGAR study, intensive glucose control was significantly associated with increased risk for moderate and severe hypoglycemia, which were both associated with higher mortality. Although it was concluded that the association demonstrated a “dose response relationship,” a causal relationship could not be proved. 33 In accordance, other studies have shown that hypoglycemia is directly associated with poor outcomes in ICU patients. In a multicenter observational study, Krinsley et al showed the same dose response association between degree of hypoglycemia and risk of ICU mortality. However, a significant effect was found even for levels consistent with “mild” hypoglycemia. 34 Similar findings were reported in a retrospective database cohort study, where the authors concluded that after adjusting for a daily adjudicated measure of disease severity, the association between hypoglycemia and mortality remained significant indicating the possibility of a causal relationship. 35
Glycemic variability (GV) is a concept that has been more recently introduced and is less well understood. Several studies have reported the importance of GV as novel metric for glycemic control and its independent association with increased mortality in the ICU,36-38 however the best method for measuring GV in hospitalized patients has not been established.39,40 Although mean amplitude glycemic excursions (MAGE) or mean absolute glucose (MAG) have been used and proposed,38,41 these measures were initially applied in the pre-CGM era using available glucose values from POC, presenting challenges due to the differences in frequency and intervals of such measurements. 41 More recently however, GV has been studied using CGM in the critically ill. In a prospective study using CGM in patients admitted with myocardial infarction, higher levels of GV were found to be present in patients who developed major adverse cardiac events up to a year after the hospital admission. 42 Similarly, in a more recent prospective study, glucose levels of 76 patients who underwent elective cardiovascular surgery were monitored perioperatively using a CGM. Postoperative glucose management was obtained using intravenous insulin infusion while in the ICU, and by subcutaneous insulin injections once oral food intake was started. Results of this study also showed that patients who exhibited increased GV had a greater rate of surgical site infections and postoperative atrial fibrillation. 43
Glucose Monitoring
In the ICU, glucose sample sources include arterial, venous, capillary, and interstitial fluid. Processing of the sample can be done at the hospital central laboratory, or through POC glucose meters and blood gas analyzers which are available in most ICUs. 22 Most measurements in the ICU are probably done by sampling capillary blood and using a POC glucose meter. POC glucose meters became available in the 1970s, and despite the routine use in the ICU they were never developed nor intended for this population of critically ill patients. 44 However, just recently the FDA approved the first POC glucose meter for critically ill patients, the StatStrip® Glucose from Nova Biomedi1cal. 45
POC glucose meters are required to have 95% of their values to be within 20% of a reference method. The allowance of 1 out of 20 readings to be more than 20% of the reference value may be unacceptable for the critically ill patient on an insulin infusion. It is estimated that up to 15% of hypoglycemic events may go undetected by current bedside glucometers. 46 There are multiple sources of errors in POC glucose measurements: metabolic abnormalities in the critically ill population (ie, edema, dehydration), direct chemical interference (ie, ascorbic acid, acetaminophen), and inherent errors from sampling site (ie, venous vs arterial vs capillary). 15 Venous samples sent to the hospital central laboratory are more accurate, but the time delay render this impractical for titration of insulin in the ICU. Measurements from blood gas analyzers are accurate, and in one recent prospective study the sample difference between central laboratory and blood gas analyzer was only 8%, which falls below the International consensus. 47 When blood gas analyzer measurements were compared to the gold standard central laboratory measurements in a subset of critical hypoglycemic values, Liang et al showed that 98.1% of the paired values fell within the 95% limit. The FDA 2016 guidelines are more stringent and require results within 12% of the central laboratory values in 95% of the samples. 48
Electronic Glucose Management Systems
Given the very short half-life of circulating insulin, continuous intravenous insulin infusion is considered the most effective method for reaching and maintaining specific glucose goals in critically ill patients. 49 Despite the availability of multiple paper protocols for adjusting insulin doses, none in particular was shown to be superior or is particularly recommended. The chosen intravenous insulin protocol should lead to target-glucose values with consistency while minimizing risk of hypoglycemia. It has been proposed that the ideal protocol should also be easy to use, require minimal staff training, retain high adherence, and be adapted to the particular patient population. 16
The use of electronic glucose management systems (eGMS) to guide the administration of insulin therapy has been increasingly gaining ground for the management of diabetes and hyperglycemia in the hospital setting. In the case of critically ill patients, these systems provide decision support to health care professionals by performing complex calculations and providing standardized insulin dosing in a digital platform often integrated with the electronic health record (EHR) system. Currently there are four commercially available eGMS for the management of diabetes and hyperglycemia in the ICU with FDA approval in the United States: GlucoCare®, Glytec® (Glucommander), GlucoStabilizer®, and EndoTool®. 50 To date, there have been no head-to-head studies comparing differences among these eGMS. However, there have been studies assessing the safety and efficacy suggesting that these eGMS likely provide better glycemic control compared to paper-based algorithms (Table 1).
Electronic Glucose Management Systems Commonly Used in the ICU.
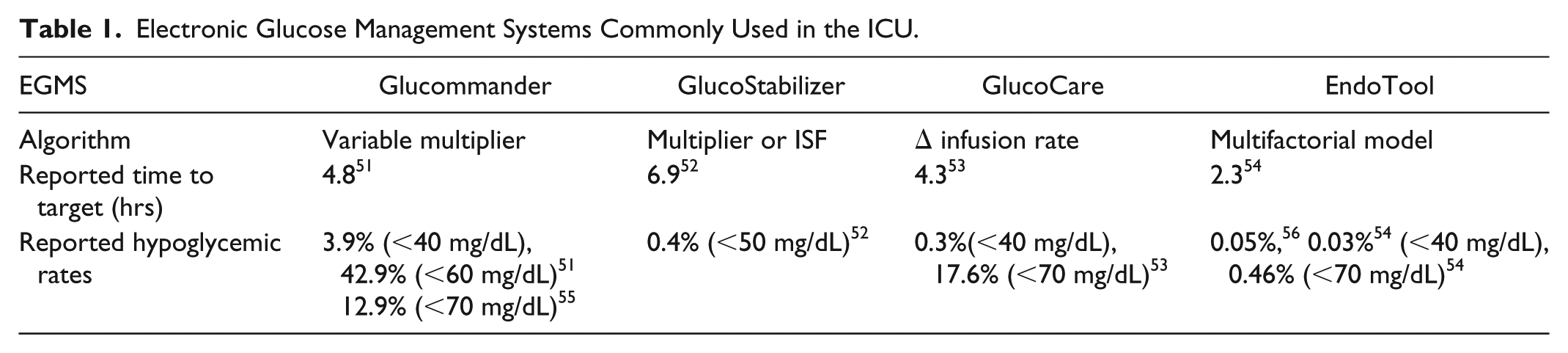
EndoTool
EndoTool is offered to institutions by Monarch Medical Technologies. This eGMS uses patient-specific factors to calculate insulin dosing and timing of both POC glucose determinations. Description of this eGMS specifies the use of patented technology that models, predicts, and adjusts insulin dosing recommendations based on 8 unique patient variables, including age, sex, blood glucose level, weight, type of diabetes, renal function, presence of steroid, and any estimated residual extracellular insulin (EREI) from previously administered insulin. This approach aims to rapidly achieve target glucoses while minimizing and preventing hypoglycemia and GV. EndoTool integrates with most EHRs and is available for pediatric patients (>2 years old and >12 kg). 57 The safety and efficacy of EndoTool was initially evaluated in a prospective study performed in four surgical ICUs and one progressive care unit where glycemic metrics of 1682 patients pre and postimplementation of EndoTool were compared. 56 Before the implementation of EndoTool, authors reported that distinct paper protocols, including the Portland Protocol, 58 were being used. After 18 months of using the eGMS, a 45% to 57% reduction of glucose values over 150 mg/dL were reported while demonstrating a 95% risk reduction (from 1% to 0.05%, P < .001) of severe episodes of hypoglycemia (<40 mg/dL). Consistently, a more recent retrospective trial analyzed the seven year impact of using EndoTool in a 900-bed tertiary teaching hospital. The study included 492 078 BG readings from 16 850 patients. With the use of this eGMS, glucose levels were brought to target within an average of 1.5 to 2.3 hours (4.5 to 4.8 hours for cardiovascular patients). Minimal hypoglycemia was observed (BG values <70 mg/dL, 0.93%; <40 mg/dL, 0.03%), with further analysis showing significant reductions over time in hypoglycemia frequency (<70 mg/dL), from 1.04% in 2009 to 0.46% in 2015 (P < .0001). In addition, GV assessed using CV% was reported to be low at 26.5% (±12.9%), with only 4% of patients exhibiting glucose excursions (defined as BG levels >180 mg/dL once control was attained). 54
Glucommander
Glucommander is made available by Glytec Systems. This eGMS adjusts insulin dosing based on initial glucose target ranges and the use of a multiplier determined by the weight of the patient. Subsequently, insulin infusion rates and rechecks of the blood glucose are recommended by the software. The multipliers are adjusted depending on the glucose proximity to the target. 59 A number of studies have been published showing the safety and efficacy of Glucommander for the management of diabetes and hyperglycemia in critically ill patients. In a multicenter trial where ICU patients were randomized to continuous insulin infusion using Glucommander or a standard paper protocol, patients managed by Glucommander showed a lower mean glucose levels (103 ± 8.8 mg/dL vs 117 ± 16.5 mg/dL, P < .001) and a shorter time to target BG (4.8 ± 2.8 vs 7.8 hours ± 9.1 hours, P < .01), with higher percentage of BG readings within range (71.0 ± 17.0% vs 51.3 ± 19.7%, P < .001) compared to the standard protocol. The percentages of patients with more than 1 episode of severe hypoglycemia (<40 mg/dL) and hypoglycemia (<60 mg/dL) were 3.9% and 42.9% in the Glucommander and 5.6% and 31.9% in the standard respectively (P > .1). No significant differences were seen in length of hospital stay (P = .704), ICU stay (P = .145), or in-hospital mortality (P = .561) between groups. 51 More recently, the use of Glucommander for the management of patients presenting with diabetic ketoacidosis (DKA) was shown to be safe and effective in a retrospective multicenter study involving 2665 patients with DKA across 34 institutions in the United States. 55 Treatment with this eGMS was associated with lower rates of hypoglycemia <70 mg/dL during the time of the insulin drip (12.9% vs 35%, P = .001), faster time to normalization of blood glucose (9.7 ± 8.9 vs 10.97 ± 10.2 hours, P = .0001) and of resolution of metabolic acidosis (13.6 ± 11.8 vs 17.3 ± 19.6 hours, P = .0001)
GlucoStabilizer
The GlucoStabilizer insulin management software is a trademark of Indiana University Health. 60 This system provides systematic and standardized titration of intravenous insulin for glycemic control within a designated glucose target range. Similarly to the Glucommander, it uses a multiplier or insulin sensitivity factor (ISF). The multiplier initial value, typically set at 0.02, plays an essential role on the insulin dose calculation, and follows patterns set by the original tables for closed-loop insulin delivery. 61 The safety and efficacy of the GlucoStabilizer was initially evaluated in a retrospective study conducted at a large academic institution where glycemic control in the ICU was compared prior to and after implementation of this eGMS. 52 In terms of glycemic control, the authors reported that the percentage of BG values below the upper limit (110 mg/dL) in the ICUs significantly increased from 31.5% to 51.5% (P < .001) with the use of the GlucoStabilizer without observing an increase in hypoglycemia. In fact, the rate of hypoglycemia (BG <50 mg/dL) decreased slightly from 0.5% to 0.4% in the ICUs during the 3 months after introduction of the GlucoStabilizer program. In the obstetric population, a recent retrospective cohort study comparing laboring patients with diabetes requiring insulin infusion managed by standard insulin dosing chart versus the GlucoStabilizer software program was performed. 62 Results of this study showed that patients managed by GlucoStabilizer software program had higher percentage of values in target range at delivery (81.8% vs 9.1%; P < .001) compared with standard insulin dosing without increasing maternal hypoglycemia (0% vs 4.3%; P = .99). In addition, mean BG was also lower compared with the standard insulin infusion (102.9 ± 5.9 mg/dL [5.7 ± 0.33 mmol/L] vs 121.7 ± 5.9 mg/dL [6.8 ± 0.33 mmol/L]; P = .02).
GlucoCare
GlucoCare 140(B) is offered by Pronia Medical Systems. This system utilizes the Yale Protocol53,63 as the basis for insulin dosing recommendations. This protocol takes into account four different patient factors: current blood sugar, immediate past blood sugar, time between measurements, and current insulin rate rate/velocity of the change in glucose levels are then calculated. The original version of GlucoCare (100 to 140 mg/dl) was shown to be safe and effective in achieving glycemic control with low rates of hypoglycemia in a retrospective study encompassing 1657 patients and 55 162 BG readings. The authors reported that 92.4% of patients reached the target range (100-140 mg/dL) within an average of 4.3 hours. Overall rates of severe hypoglycemia (<40 mg/dL) were 0.01% of readings and 0.3% of patients. Hypoglycemia (<70 mg/dL) occurred in 1.1% of readings and 17.6% of patients. This eGMS latest version (GlucoCare 140(B)) has a single target of 140 mg/dl which was modified from the former versions (100-140 and 120-140) resulting in reduced incidence of hypoglycemia <70 mg/dL( from 0.998% to 0.256%, P < .00l). In addition, a feature that allows for insulin boluses to reduce overall continuous insulin infusion rates, was shown to further decrease rates of hypoglycemia to 0.04% (P < .00l). 64
Continuous Glucose Monitoring (CGM)
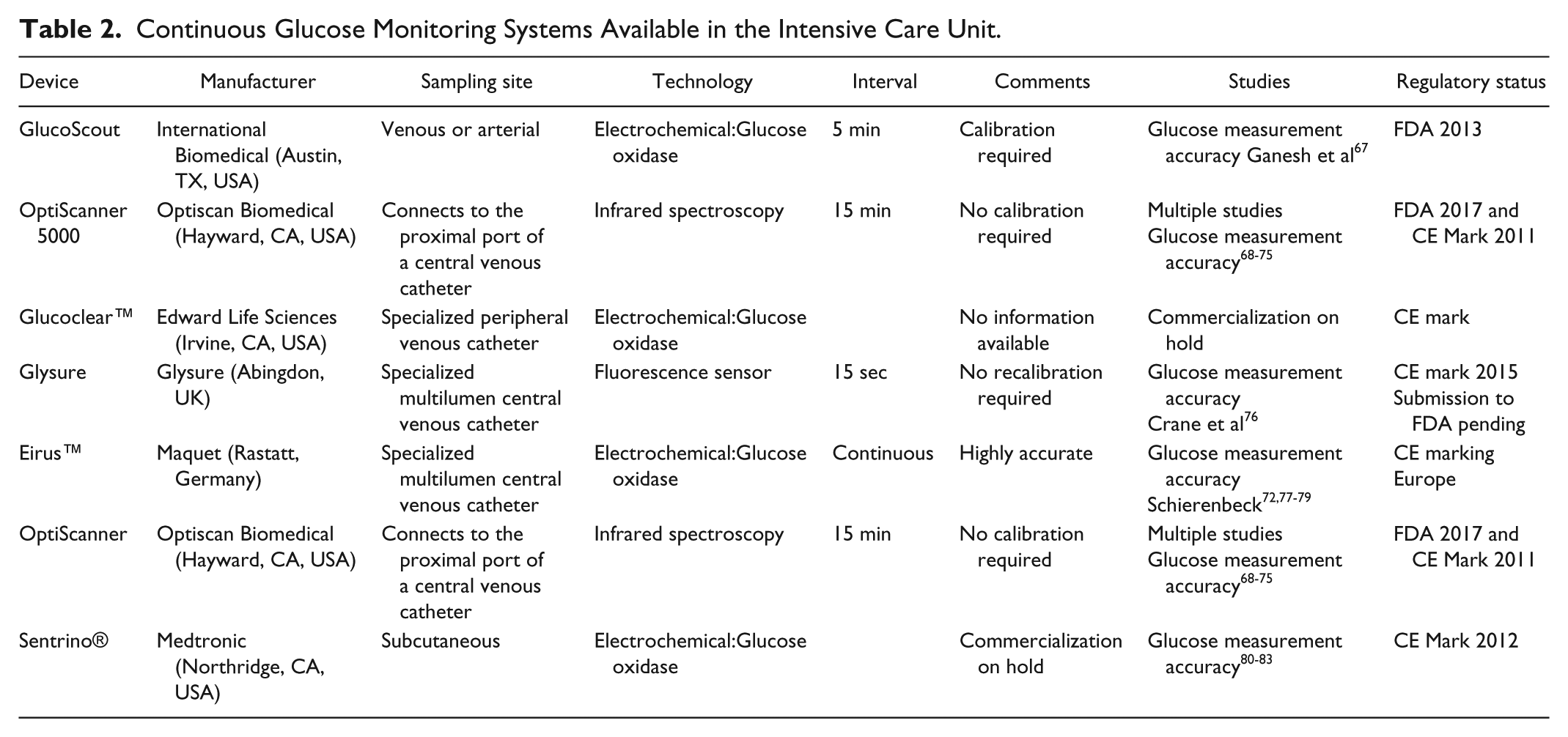
CGM can be achieved minimally invasive, such as with subcutaneous and transdermal devices, versus invasive intra-arterial or intravenous. CGM technology in the ICU has gained increased attention, as tighter glucose control increases the risk of hypoglycemia. 65 CGM technologies have been developed since 1970, and currently there are more than 15 CGM devices described in the outpatient setting. Just in 2018, six CGM systems have received FDA approval. The GlucoScout® and the OptiScanner® are the only two FDA-approved CGMs for hospitalized patients, and they require venous or arterial cannulation (Table 1). 22
CGM devices can continuously monitor blood glucose every 1-15 min, and most are limited by the need for recalibration after 12-24 hours, with the exception of OptiScanner and Glysure® that do not require recalibration (Table 2). Other specialized sensing techniques in the interstitial fluid include reverse iontophoresis, thermal emission, photoacoustic detection or sonophoresis. 66 Most devices on the market use the glucose-oxidase electrochemical method (Table 2).
Continuous Glucose Monitoring Systems Available in the Intensive Care Unit.
The most common minimally invasive sensor is placed subcutaneously in the abdomen or in the arm, sampling the interstitial fluid for glucose measurements. Frequent recalibrations are required to maintain sensor accuracy using POC glucose meters as a reference. There are discrepancies between blood and interstitial fluid concentrations due to several factors, such as delay in glucose equilibration from blood to interstitial space, measurement delays from using subcutaneous sensors, and glycemic dynamic profiles. 66
In patients with septic shock and on insulin infusion, CGM (Medtronic MiniMed Soft-Sensor™) correlates with intermittent capillary blood glucose measurements. 84 Other RCTs (randomized controlled trials) have yielded same correlation between POC glucose measurements and CGM in ICU patients. 85 The literature is mixed in regards to CGM and detection of hypoglycemia, while Holzinger et al showed that CGM decrease the episodes of hypoglycemia, 86 Boom et al failed to demonstrate increase detection of hypoglycemic events. 85 Newer CGM may offer increased accuracy and less need for recalibration. In a recent clinically validated virtual trial, CGM reduced the amount of blood work required by 74% without compromising safety. 87 A recent systematic scoping review by van Steen et al identified 32 studies that addressed the accuracy of CGM, but only five RCTs in the ICU population that studied the efficacy of CGM. The author concluded that subcutaneous CGM doesn’t improve glycemic control, however no formal meta-analysis could be done due to the few number of studies (five), small group sample size, group heterogeneity, and difference in target glucose levels. 88 Of note, all RCTs that addressed efficacy used subcutaneous CGM, which is considered less accurate than intravascular devices. Differences in the accuracy of subcutaneous and intravascular devices still present a challenge. Schierenbeck et al showed a MARD (mean absolute relative difference) of 30.5% versus 6.5% between FreeStyle Libre® (subcutaneous) and Eirus (intravascular), with lower MARD corresponding with higher accuracy. 89 Even though a definitive answer has not been reached, expert consensus is that CGM could offer improved glucose control with less risk of hypoglycemic events in ICU patients.19,90
Closed-Loop Systems
The artificial pancreas or closed-loop insulin delivery system is the most promising advance in glycemic control in the critical ill patient. This feedback system combines an insulin infusion with a CGM device. The only full closed-loop system in the market is the STG-22 and STG-55 made by Nikkiso, and only available in Japan. The Japanese Health Care System has approved this full-closed loop system for perioperative glycemic management. 91 In the rest of the world, the clinical implementation is currently unavailable. For outpatient management of type 1 diabetes, systems such as the Medtronic MiniMed 670G™, offers its closest version to a true “artificial pancreas,” showing improved management of hyperglycemia while preventing hypoglycemic events. The use of subcutaneous insulin delivery and interstitial glucose sampling remains a major limitation in the ICU population. Many critically ill patients require intravenous delivery of insulin and interstitial fluid has many limitations that may render the glucose levels inaccurate (ie, edema, anemia, vasoconstriction, lag time).
However, in the noncritical hospitalized patient, closed-loop insulin delivery was recently shown to be feasible and safe. Bally et al conducted a randomized open-label study that showed the control closed-loop group to be in target range 65.8% versus 41.5% in the control group without any significant difference of hypoglycemic events. 92 Multiple small studies have shown that a closed-loop insulin delivery system improves glucose control and prevents hypoglycemic events in mixed medical ICUs and in very specific surgical populations.93-95
Summary
In the critically ill patient, insulin resistance and marked fluctuations in glycemia are an inherent part of the hormonal stress-axis activation, cytokine driven inflammatory state, common steroid use, and interaction with the nutritional therapy modality and macronutrient composition. The Surviving Sepsis Campaign of 2016 recommends blood glucose monitoring every 1 to 2 hours until glycemic levels are stable and then every 4 hours. Furthermore, for most critically ill patients, blood glucose levels should be targeted to <180 mg/dL, and POC capillary blood glucose measurements are to be used with caution 24
The pendulum has swung away from tight glucose control, mostly as a result of increased hypoglycemic events that increase mortality. 33 The optimal glucose target is debatable, but some specific surgical populations possibly benefit from IIT of 110-140 mg/DL as long as hypoglycemia is avoided. POC glucose monitors are inaccurate in the ICU population, but their clinical use is universal, with many studies using POC capillary blood glucose as the gold standard reference. 96 Subcutaneous CGM sensors also have limitations, including lag time and inaccuracy in conditions associated with critically ill patients, such as anemia, peripheral edema, vasoconstriction, use of vasopressors, and high acuity.80,88,97 Intravascular continuous glucose monitors are more accurate as they reflect fluctuations more precisely in blood glucose, less prone to interference by patient condition (ie, vasoconstriction, peripheral edema). In addition, many don’t require recalibration. The downside is the invasive nature and the complications associated with central vascular catheters.
The literature of efficacy with CGM is still limited in the ICU. The discrepancy in the results of these studies is derived from the use of different subcutaneous sensors, patient population, and differences in glucose management protocols. However, as recently proven in non–critically ill patients, 92 CGM together with a subcutaneous continuous insulin delivery system has proven safe and effective in non–critically ill patients. Therefore it is possible that as glucose management technologies continue to improve, CGM with an integrated eGMS or fully automated closed-loop system will come to be the standard practice for the management of diabetes and hyperglycemia in critically ill patients. Decision support software that would allow safe and effective delivery of subcutaneous insulin rather than intravenous insulin may provide significant cost savings and a more cohesive management during transitions of care. It is also possible that some critically ill patients may require continuous intravenous sensors and insulin delivery systems in order to adequately manage their hyperglycemic state. Prospective studies are thus needed in order to establish the role of CGM, eGMS, and closed-loop system in managing the critically ill population.
Footnotes
Abbreviations
ADA, American Diabetes Association; CABG, coronary artery bypass grafting; CGM, continuous glucose monitoring; DKA, diabetic ketoacidosis; EHR, electronic health record; EREI, estimated residual extracellular insulin; eGMS, electronic glucose management systems; GV, glycemic variability; ICU, intensive care unit; IIT, intensive insulin therapy; ISF, insulin sensitivity factor; MACE, major adverse cardiac events; MAG, mean absolute glucose; MAGE, mean amplitude glycemic excursions; MARD, mean absolute relative difference; POC, point of care; RCT, randomized controlled trial.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Carlos E. Mendez is part of the medical advisory board for Monarch Medical Technologies. Other author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
