Abstract
Background:
In type 1 diabetes (T1D) continuous subcutaneous insulin infusion (CSII) use has been associated with lower risks for mortality and diabetes-related complications when compared to multiple daily injections. There are limited data on the effect of CSII on blood pressure (BP). We aimed to evaluate the relationship between CSII initiation and BP changes.
Methods:
We performed a retrospective, single-center, observational study including all adults with T1D who initiated CSII between 2001 and 2014. Mean systolic BP levels were calculated from values obtained up to 15 months before and up 15 months after CSII initiation.
Results:
We studied 436 adults (46% male; mean [95% CI] age: 45 [36, 54] years, duration of diabetes: 20.3 [19.1, 21.5] years). CSII was associated with the following changes (post minus pre CSII levels) in mean (95% CI) SBP and DBP levels, respectively: whole cohort (N = 436): −2 (−1, −3),
Conclusions:
We showed BP reduction in association with CSII therapy particularly in those on antihypertensive medication. These original findings may partly explain the cardiovascular and mortality benefits associated with CSII therapy.
Keywords
CSII is considered the “gold standard” therapy for people with T1D. 1 CSII reduces both HbA1c and the incidence of severe hypoglycemia compared to multiple daily injections (MDI). 2
The commonest cause of death in adults with T1D is cardiovascular disease (CV). A large observational study has shown lower CV and total mortality in adults with T1D, treated with CSII compared to MDI. 3 Similarly, observational data suggest beneficial effects of CSII on the development and progression of albuminuria, retinopathy and neuropathy, independent of HbA1c.4-7 However, mechanisms of benefit beyond HbA1c lowering are unclear. No study has focused on the relationship between CSII and blood pressure levels and therefore we aimed to do this in a large cohort of adults with T1D.
Methods
This was a retrospective, single-center, observational study including all adults with T1D who initiated CSII between 2001 and 2014. We excluded those with <12 months follow-up; those without a documented CSII start date; those without information on BP levels or/and therapy; those receiving treatment for postural hypertension and those that were pregnant. Electronic and paper records were used for data collection. Mean systolic (SBP) and diastolic (DBP) blood pressure levels were calculated from all BP measurements obtained up to 15 months before and up to 15 months after CSII initiation. BP was measured using validated automated monitors used in hospital practice. BP was measured seated after a period of rest.
Data on the whole cohort are presented along with data from subgroups (1) not receiving antihypertensive treatment at baseline who remained off antihypertensive treatment (antihypertensive treatment-naïve patients) and (2) receiving stable antihypertensive treatment without any changes either to the kind or the doses of antihypertensive medications during the follow-up period.
Data presented are mean (95% CI). Changes in SBP and DBP (post minus pre CSII values) were assessed by paired samples Student’s
Results
Data from 436 adults were included: 46% male; mean (95% CI) age: 45 (36, 54) years, duration of diabetes: 20.3 (19.1, 21.5) years (Table 1). The proportions (numbers) in different treatment categories were: antihypertensive treatment-naïve 64% (n = 279); receiving stable antihypertensive treatment 27% (n = 118); and undergoing changes to antihypertensive treatment 9% (n = 39). Regarding the latter group of patients who underwent changes, 4 patients, who were not on any antihypertensive medication at baseline, started antihypertensive medication during follow-up; 3 patients on antihypertensive medication at baseline discontinued this medication during follow-up; in 13 patients on antihypertensive medication at baseline this medication was intensified during follow-up either by increasing the dose of existing medication or by adding a new drug; in 9 patients on antihypertensive medication at baseline this was deescalated during follow-up either by decreasing the dose of existing medication or by removal of one or more drugs; in 10 patients on antihypertensive medication at baseline, there were changes in the antihypertensive medication mostly due to intolerance or side effects. We analyzed 1663 BP readings; the mean number of readings per individual was 2.1 (2.0, 2.2) pre-CSII and 2.5 (2.4, 2.5) post-CSII.
Clinical Characteristics Prior to CSII Initiation.
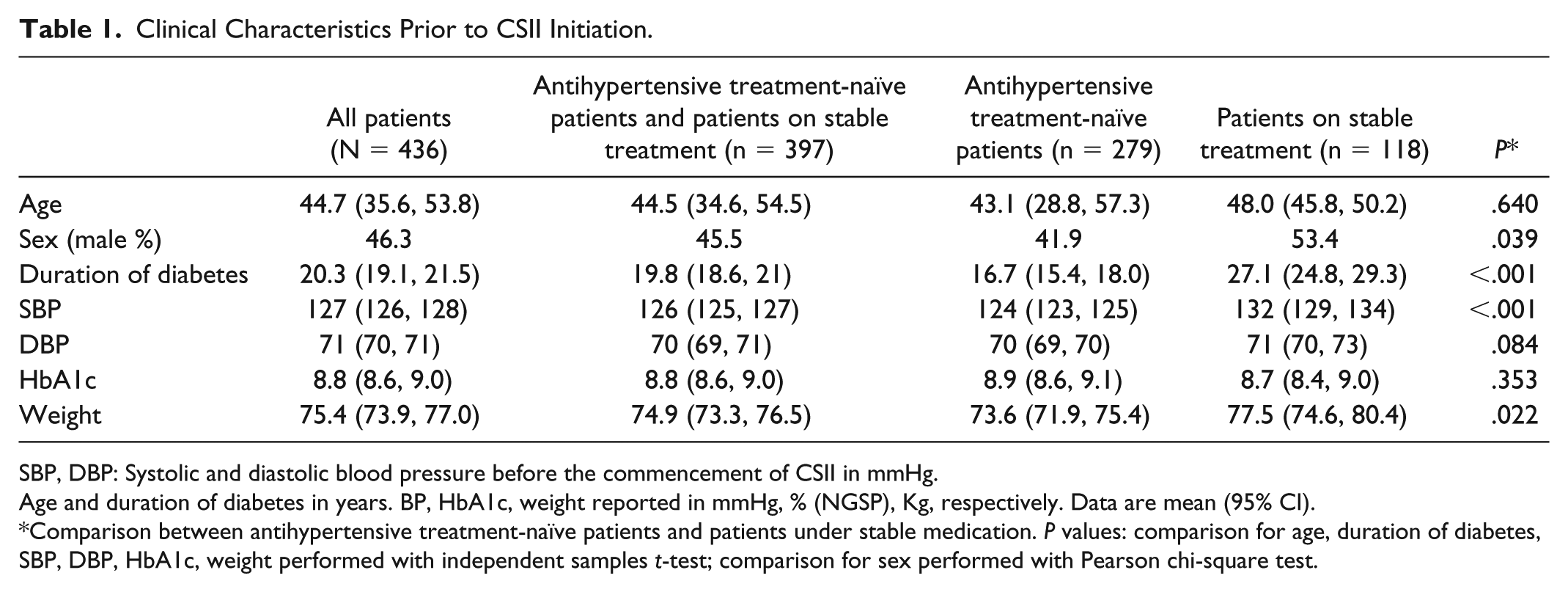
SBP, DBP: Systolic and diastolic blood pressure before the commencement of CSII in mmHg.
Age and duration of diabetes in years. BP, HbA1c, weight reported in mmHg, % (NGSP), Kg, respectively. Data are mean (95% CI).
Comparison between antihypertensive treatment-naïve patients and patients under stable medication.
Patients receiving stable antihypertensive medication had higher baseline SBP levels compared to treatment-naïve group (132 [129, 134] vs 124 [123, 125] mmHg,
Changes in BP in association with CSII initiation are summarized in Table 2. The most noticeable change in BP was observed in patients receiving stable antihypertensive treatment who exhibited reductions in SBP of 4 mmHg and DBP of 2 mmHg (both
Changes in Systolic and Diastolic Blood Pressure Levels in Association with CSII Initiation.
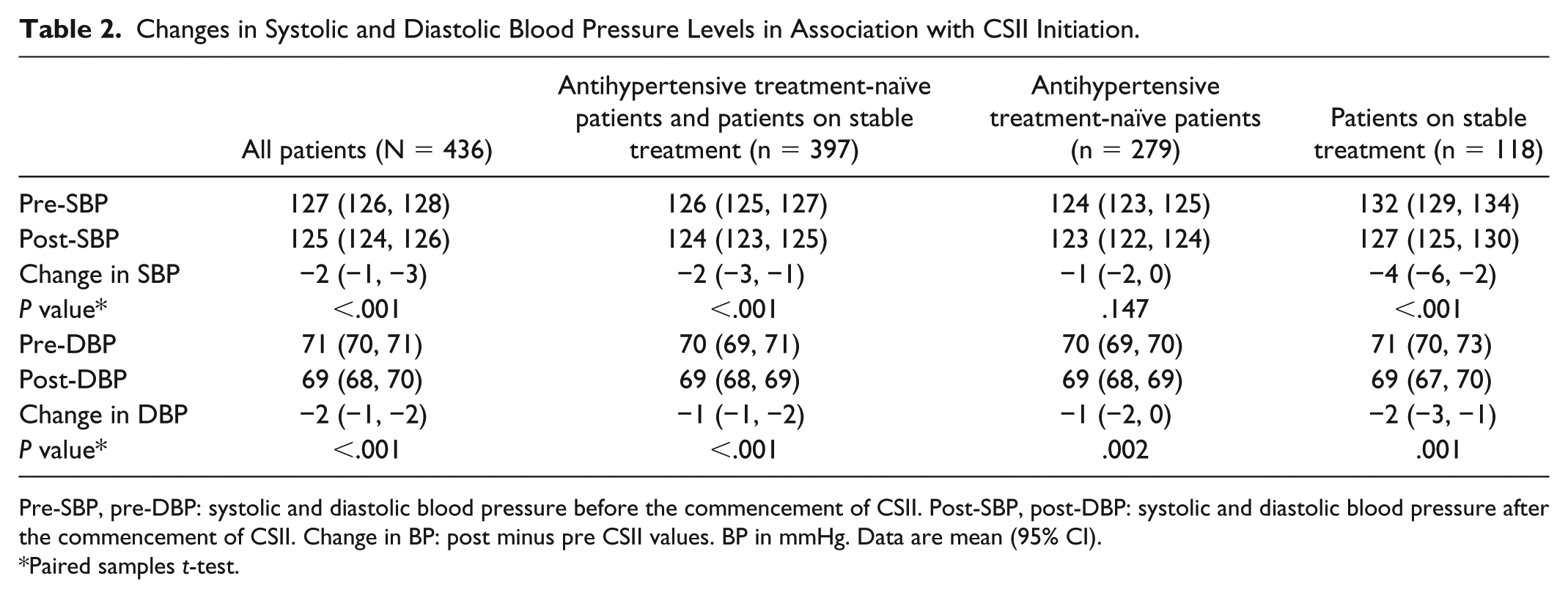
Pre-SBP, pre-DBP: systolic and diastolic blood pressure before the commencement of CSII. Post-SBP, post-DBP: systolic and diastolic blood pressure after the commencement of CSII. Change in BP: post minus pre CSII values. BP in mmHg. Data are mean (95% CI).
Paired samples
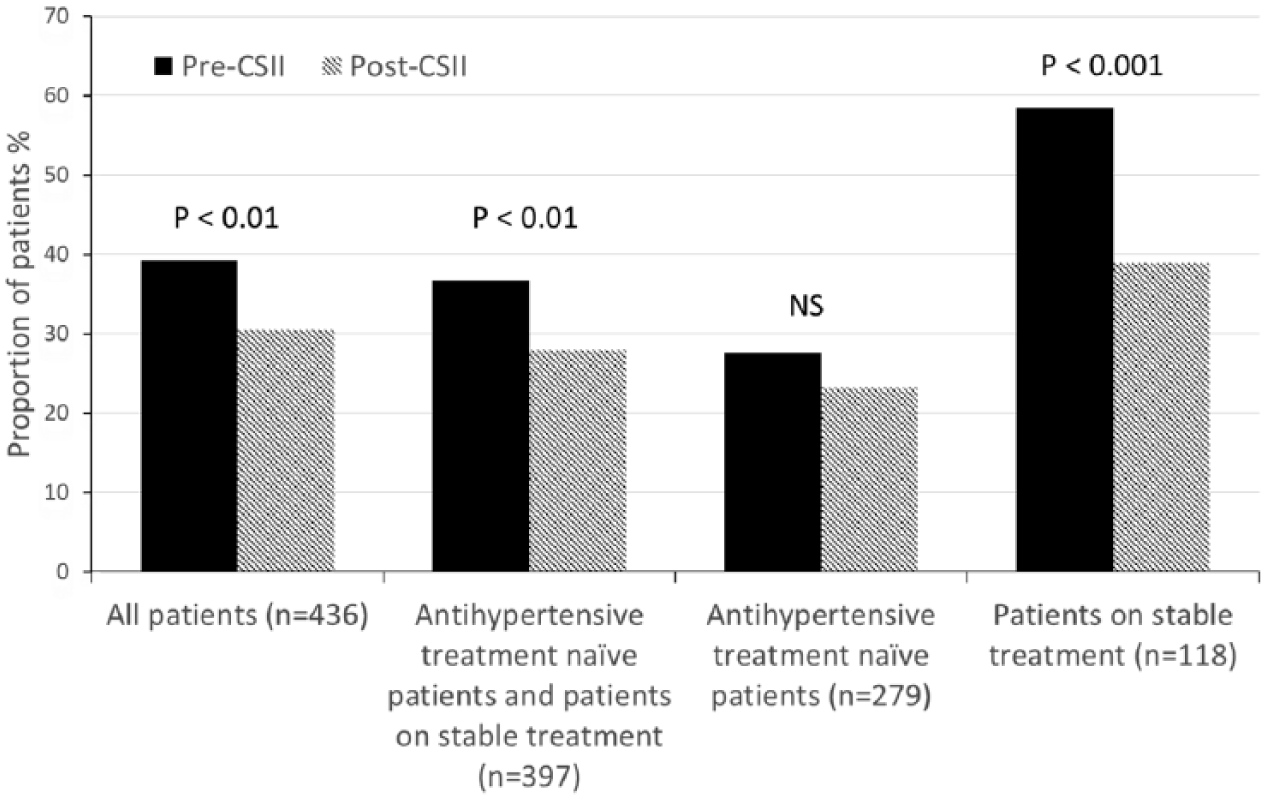
Proportion of patients with SBP above 130 mmHg and/or DBP above 80 mmHg prior and post the commencement of CSII.
Relationship of CSII Initiation With HbA1c and Weight.
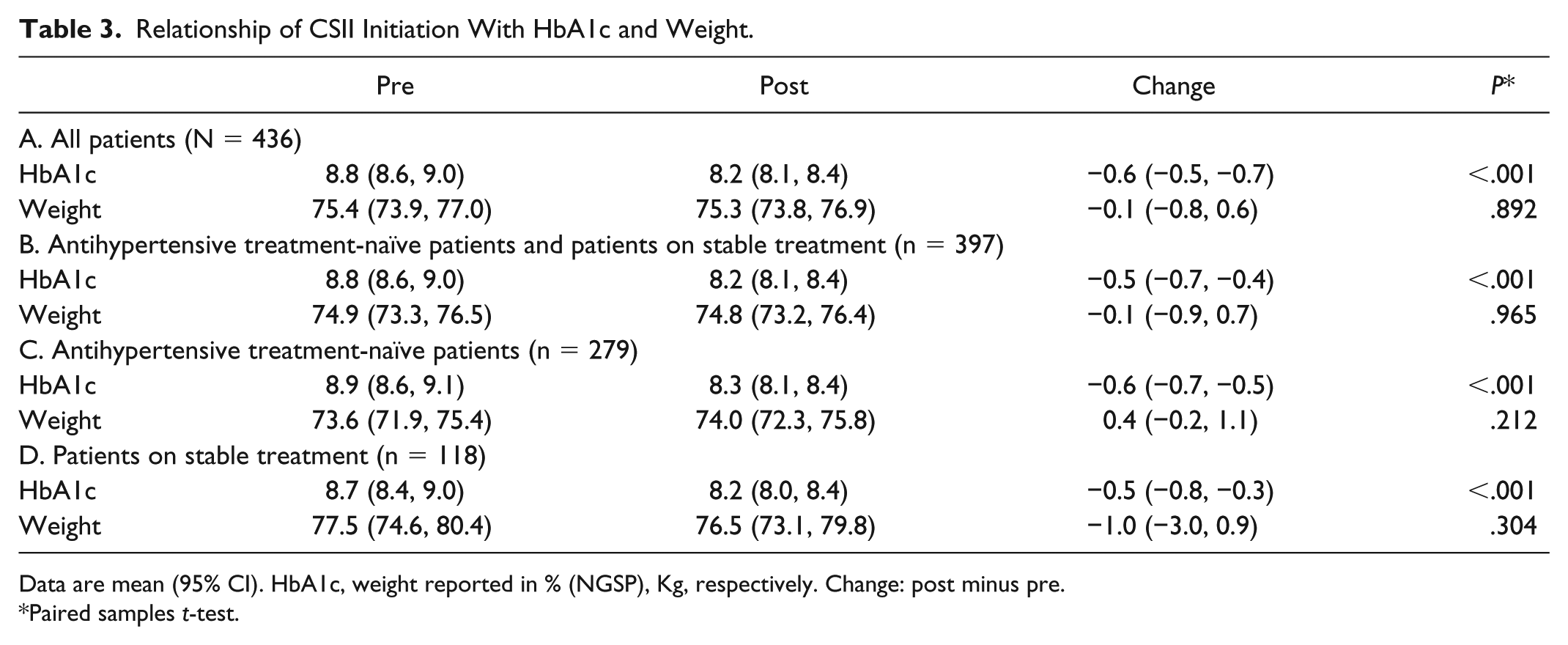
Data are mean (95% CI). HbA1c, weight reported in % (NGSP), Kg, respectively. Change: post minus pre.
Paired samples
Multivariate linear regression was used to assess whether age, sex, duration of diabetes, pre-CSII BP values, or changes in HbA1c or weight predicted changes in BP (Table 4). Only pre-CSII BP was found to have a strong independent association with BP reduction in all of the patients’ groups. In the whole cohort, higher pre-SBP was strongly associated with greater SBP lowering (β coefficient [standard error]: −0.438 [0.040],
Predictors of Blood Pressure Changes in Multivariable Linear Regression.
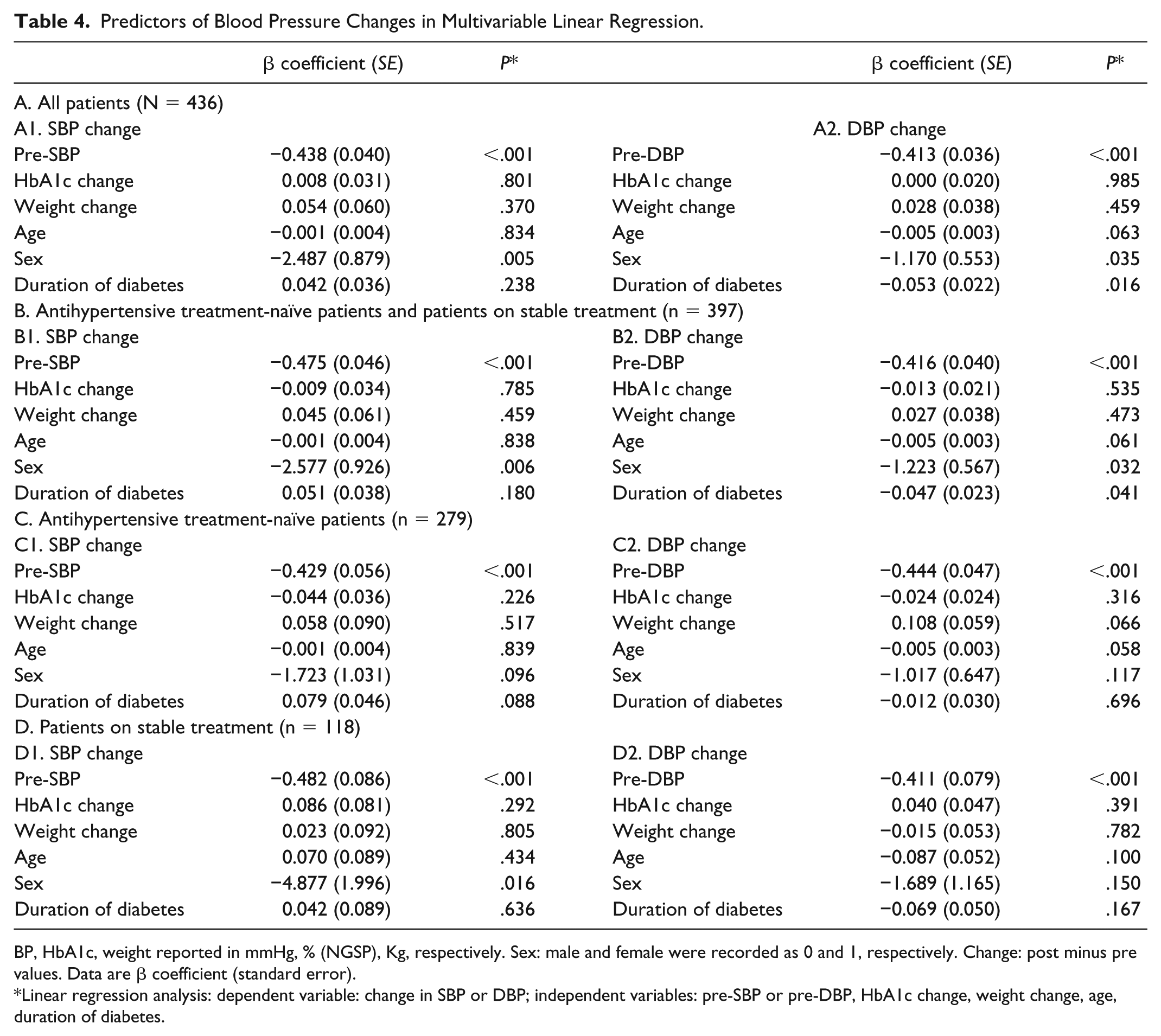
BP, HbA1c, weight reported in mmHg, % (NGSP), Kg, respectively. Sex: male and female were recorded as 0 and 1, respectively. Change: post minus pre values. Data are β coefficient (standard error).
Linear regression analysis: dependent variable: change in SBP or DBP; independent variables: pre-SBP or pre-DBP, HbA1c change, weight change, age, duration of diabetes.
Finally, we were interested to know whether BP lowering with CSII was greater in those taking stable antihypertensive therapy compared to those who were treatment-naïve. In a univariate analysis of variance that combined these group, the reductions of SBP and DBP in those taking antihypertensive medication were significantly greater than in those not taking antihypertensive medication (Table 5). However, after adjustment for pre-CSII BP values there was no significant difference in BP reductions between these two groups.
Association Between Use of Antihypertensive Medication and Blood Pressure Changes With CSII Treatment.
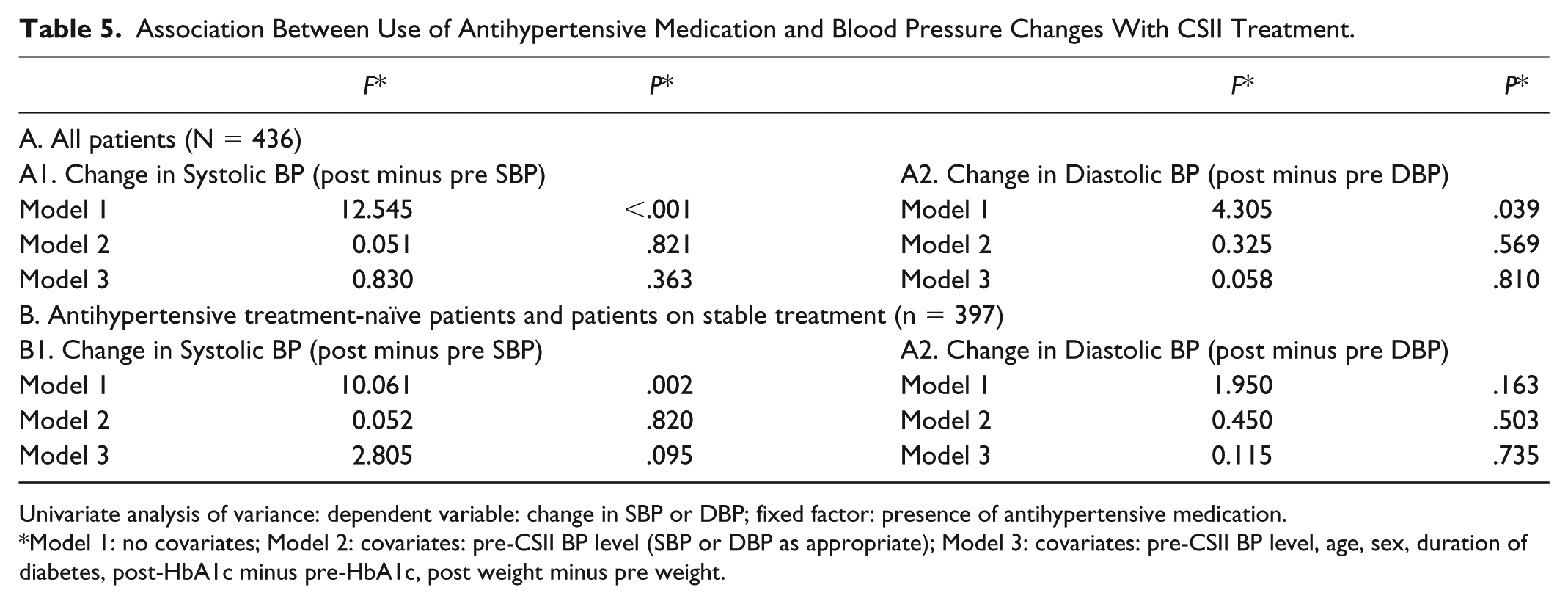
Univariate analysis of variance: dependent variable: change in SBP or DBP; fixed factor: presence of antihypertensive medication.
Model 1: no covariates; Model 2: covariates: pre-CSII BP level (SBP or DBP as appropriate); Model 3: covariates: pre-CSII BP level, age, sex, duration of diabetes, post-HbA1c minus pre-HbA1c, post weight minus pre weight.
Discussion
Main Findings
In this retrospective analysis, we found a reduction in SBP and DBP after initiation of CSII in a large cohort of adults with T1D. The changes in BP, especially regarding SBP, were mostly driven by the changes noted in the group of patients receiving stable antihypertensive treatment. In this group mean SBP and DBP reductions were −4 and −2 mmHg and the proportion with SBP >130 mmHg and/or DBP >80 mmHg dropped from 59% to 39% post-CSII.
Prior Studies
As far as we are aware, no dedicated study has focused on assessing blood pressure changes occurring in association with CSII therapy.
In previous studies CSII or sensor augmented pump (SAP) therapy have been shown to independently reduce albumin excretion rate when compared to MDI after adjustment for HbA1c and BP.4,5,7 In these studies no significant changes in mean/medial values of BP on CSII are reported. However no paired data analysis was performed and analysis has not taken into account changes in the status of hypertensive treatment during follow-up. In a small pediatric cohort study assessing changes in vascular function with CSII therapy a reduction of BP at 3 weeks was reported but this reduction was not maintained after one year. 8 The small sample size of the study certainly limits its ability to demonstrate significant BP changes. In a recent large observational study from the Danish Adult Diabetes Registry, CSII users found to have lower SBP compared to MDI users but the difference was no longer statistical significant after adjustment for sex, age, duration of diabetes, smoking, body mass index, and use of lipid-lowering and blood-pressure-lowering medication. 9 The lack of paired data before and after CSII is obviously a major limitation to draw conclusions regarding the effect of CSII on BP.
Mechanistic Insights
a. Several studies have shown that when compared to MDI treatment, the total daily insulin dose is ~10-20% lower with CSII.10-12 Lowering the insulin dose could lower BP because insulin is known to have direct effects on the kidney resulting in sodium retention and it can also increase circulating levels of norepinephrine.13-15 These BP lowering effects may be particularly relevant in hypertensive salt sensitive individuals. A decrease in the sodium pool could also lead to decreased intraglomerular pressure which could bridge the results of our study to those showing a beneficial effect of CSII on albumin excretion rate.4,5,7,16
b. The effect of glycemic variability on the vascular bed has been a field of growing interest. Blood glucose variability has been shown to be less on CSII than on MDI. 17 CSII has been found to be associated with lower levels of arterial stiffness when compared to MDI and this effect was independent to HbA1c level. 18 Thus the effect of a more stable glycemic control on the vascular system might be another factor underlying the modulation of BP by CSII. A study with acquisition of continuous glucose monitoring data pre- and post-CSII treatment would be helpful to investigate this further.
c. Another potential explanation is the possibility of an improved adherence to treatment following initiation of CSII. CSII initiation is accompanied by an intense medical/nursing input and patients are commonly motivated on trying a new start in their diabetes management. These factors may result in a better adherence to their medication. We can only speculate that this factor could help to explain the higher drop in BP observed in the group under antihypertensive treatment compared to the antihypertensive treatment-naïve group. However we should note that after adjustment for pre-CSII BP values there was no significant difference in BP reductions between those on antihypertensive treatment and antihypertensive treatment-naïve patients.
Clinical and Research Implications
Our study highlights potentially important clinical benefits of CSII therapy on BP levels especially in those with preexisting hypertension (with BP lowering ~4/2 mmHg). Our data are supported by plausible mechanistic explanations. Future clinical trials involving CSII/SAP should be designed to include an assessment of BP ideally assessed using ambulatory devices and for there to be embedded research on potential mechanisms of benefit.
Strengths and Limitations
Our study has several strengths: First, this is the first study dedicated to this research question. Second, we studied a large cohort of CSII-treated T1D patients with detailed information on BP therapy. The latter factor is particularly important because alterations in antihypertensive therapy could be a major confounder in such an analysis. Third, we stratified patients based on their antihypertensive therapy status. Fourth, we chose to study within-person BP changes so that patients acted as their own control. This study design improves statistical power and may reduce bias compared to a two-cohort parallel design observational study since patients identified for CSII therapy may be intrinsically different to those not CSII-treated. Fifth, changes in the status of antihypertensive treatment took place only in a limited number of patients (39 out of 436). Moreover, the number of cases that antihypertensive treatment was initiated or intensified was balanced to the number of cases that medication was discontinued or deescalated. Thus the effect of this group in the outcomes of the total cohort is expected to be a limited one.
We acknowledge some limitations. First, this was a retrospective study. However, our case finding and data collection processes may have protected us from bias. Case ascertainment was based on computer records of consecutive patients and we included all those satisfying prespecified selection criteria. Data used are part of our routine clinical record and therefore there are limited opportunities for reporting bias. We did however rely on clinicians to accurately record changes to a treatment in our records. Second, this was a single-center study, which could potentially limit the generalizability of our findings. However, we have one of the largest cohorts of CSII-treated patients in the United Kingdom and our practice is in line with national guidance. Finally, we have no data regarding total insulin dose, glycemic variability, and albumin excretion rate.
Conclusion
We showed BP reduction in association with CSII therapy particularly in those with preexisting hypertension (higher BP levels). These original findings may partly explain the beneficial effects of CSII therapy on CV morbidity/mortality and microvascular complications in other studies. Our findings should be replicated in clinical trials that include mechanistic evaluations specially regarding the role of insulin dose and glycemic variability alterations.
Footnotes
Acknowledgements
Mrs Juliet Ward (nee Morris) contributed to data collection and patient care but passed away before this publication. Dr Andrew Hindle contributed to data collection. We acknowledge all staff at Manchester Diabetes Center who provided patient care. Part of the data contained in this article was presented as an abstract at the EASD meeting in 2016.
Abbreviations
BP, blood pressure; CSII, continuous subcutaneous insulin infusion; DBP, diastolic blood pressure; MDI, multiple daily injections; SAP, sensor augmented pump; SBP, systolic diastolic pressure; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MKR has acted as a consultant for GSK, Roche, and MSD, and participated in advisory board meetings on their behalf. MKR has received lecture fees from MSD and grant support from Novo Nordisk, MSD, and GSK. LL reports having received speaker honoraria from Minimed Medtronic, Animas, Roche, Sanofi, Insulet and Novo Nordisk, serving on advisory panel for Abbott Diabetes Care, Roche, Sanofi, Minimed Medtronic, Animas, and Novo Nordisk, grants to attend educational meetings from Sanofi, Dexcom, Novo Nordisk, and Takeda, Research support from Novo Nordisk and Dexcom.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
