Abstract
Background:
Current insulin pumps now feature advanced functions for calculating insulin dosages, delivering insulin and analyzing data, however, the perceived usefulness of these functions in clinical settings has not been well studied. We assessed the use and patient perceptions of an insulin delivery system (Accu-Chek® Combo, Roche Diagnostics, Mannheim, Germany) that combines an insulin pump and a handheld multifunctional blood glucose meter with integrated remote control functions.
Methods:
This prospective, observational, multicenter study enrolled 74 type 1 diabetes patients within 13 weeks after starting use of the pump system. At 4 to 24 weeks, investigators collected usage data from the latest 14-day period.
Results:
Seventy-two patients completed the evaluation, aged 39 ± 15 years, diabetes duration 16 ± 13 years, HbA1c 8.3 ± 1.6%. At follow-up, 62 (86.1%) patients used the remote control for ≥50% of all boluses, 20 (27.8%) used the bolus advisor for ≥50% of all boluses, and 42 (58.3%) viewed at least 1 of the e-logbook reports. More than 95% of users appraised the functions as easy-to-use and useful; median scores from VAS (0 = useless to 100 = indispensable) ranged from 72 to 85.
Conclusion:
A high percentage of study patients used the system’s advanced features, especially the remote control feature for bolusing. Overall, patients assessed the functions as useful and easy to use. Results support the implementation of these smart capabilities in further insulin pump developments.
Keywords
Treatment with intensive insulin regimens, using insulin pumps, has been shown to be an effective method for improving glycemic control and preventing long-term diabetic complications 1 and is the recommended therapy for most individuals with type 1 diabetes. 2
Although current insulin pump designs now feature advanced functions, such as automated bolus advisors and remotely controlled bolus and basal insulin administration, the functionality and perceived usefulness of these functions in clinical settings have not been well studied. The present study evaluated a current insulin pump system to determine whether and/or to what degree its advanced functions are utilized, practical, and useful in individuals.
Methods
The primary objective of this prospective, observational, multicenter study was to assess and document the utilization of the advanced functions of the Accu-Chek® Combo insulin pump system (Roche Diagnostics GmbH, Mannheim, Germany) as used in clinical practice, with secondary focuses on frequency of function utilization, patient perceptions of function ease of use, and utility and change in HbA1c values from baseline.
Subjects
Patients were recruited from 22 centers in France, including 4 regional university hospitals, 12 local public hospitals, and 6 private practices. In accordance with the directives of article L-1121-1 of the French Code for Public Health Concerning Biomedical Research, the study design did not require approval from local Ethics Committees. All subjects provided informed consent for study participation, and the local law on individual data protection was applied for the study data management. Inclusion criteria were type 1 diabetes, aged >15 years, and treatment with the insulin pump system during the past 3 months. Exclusion criteria were disease worsening, or severe disease, which, according to the investigator, could hinder satisfactory progress during the patient observation period.
Insulin Pump System
This insulin pump system includes a remotely controlled insulin pump (Accu-Chek Spirit Combo, Roche Diagnostics GmbH, Mannheim, Germany) and a handheld wireless communicating multifunctional blood glucose meter (Accu-Chek Performa Combo or Accu-Chek Aviva Combo, Roche Diagnostics GmbH, Mannheim, Germany). The blood glucose meter allows patients to remotely control the insulin pump functionalities, displays the current basal rate of the pump, and provides integrated applications to assist patients in daily diabetes management: bolus advisor and data management (digital e-logbook) (Figure 1). The safety and efficacy of the bolus advisor are documented in a number of clinical studies.3-7

Handheld remote control device with integrated blood glucose monitoring and automated bolus advisor functions. A 24-hour glucose trend graph report is shown in the display.
Procedures
All patients initiated use of the insulin pump system within 3 months of enrolling in the study. Use of the specific advanced functions at system initiation was decided by patients and their physicians. Training in insulin pump handling and management, flexible insulin therapy (FIT), and carbohydrate counting was determined by physicians, according to patient needs and local clinical practice procedures.
Baseline (visit 1)
The baseline visit was defined as the first routine clinic visit that occurred within 3 months of starting the insulin pump system. Investigators confirmed patient eligibility, obtained informed consent and documented patient history, physical status, lifestyle, presence/severity of diabetic complications, current insulin regimen, and current HbA1c. Investigators used structured interviews in conjunction with self-reported data (eg, patient logbooks) to assess patient baseline knowledge of the insulin pump system’s advanced functions, including blood glucose testing function, remote control functions for delivering insulin boluses, programming/using temporary basal rate and suspending pump activity, integrated data management tool (digital e-logbook), and bolus advisor function.
Follow-up (visit 2)
This visit was defined as the next routine clinic visit occurring 4-24 weeks subsequent to visit 1. At visit 2, investigators again used structured interviews and patient-reported data to assess frequency of advanced function use during the prior 14 days, which can be considered as reliable and clinically relevant period 8 and obtain patient feedback regarding the ease of use and utility of those functions.
Measures
The following measures were used to assess patient utilization and perceptions of the insulin pump system functions during the 14 days prior to visit 2: blood glucose meter; use of temporary basal rate; use of pump suspension; use of the handheld remote control device to deliver insulin boluses and program/utilize temporary basal rate and pump suspension; bolus advisor option; use of data management reports (view data, trend, standard day, standard week, target); consult ongoing basal rate; and blood glucose test reminder option. All data regarding frequency of use of the various functions were self-report by patients. In addition, patients provided qualitative data at the end of visit 2, using Likert scales (ease of use of functions) and visual analog scales (VASs) for the usability of functions.
Statistical Analysis
Clinical and laboratory data are presented as mean (SD) or median (interquartile ranges), apart from incidence data. Comparative analyses were done using Wilcoxon’s test and chi-square or Fisher’s tests. Statistical analyses were performed with SAS version 9.3 (SAS Institute, Cary, NC), and P values < .05 were considered statistically significant.
Results
Of the 74 consecutive patients enrolled, 72 completed the study and were included in the analysis. Two patients were lost to follow-up. Baseline patient characteristics are summarized in Table 1. Most patients had no training in FIT and carbohydrate counting, and approximately 83% of patients were naïve to insulin pump therapy prior to starting the insulin pump system.
Patient Characteristics.
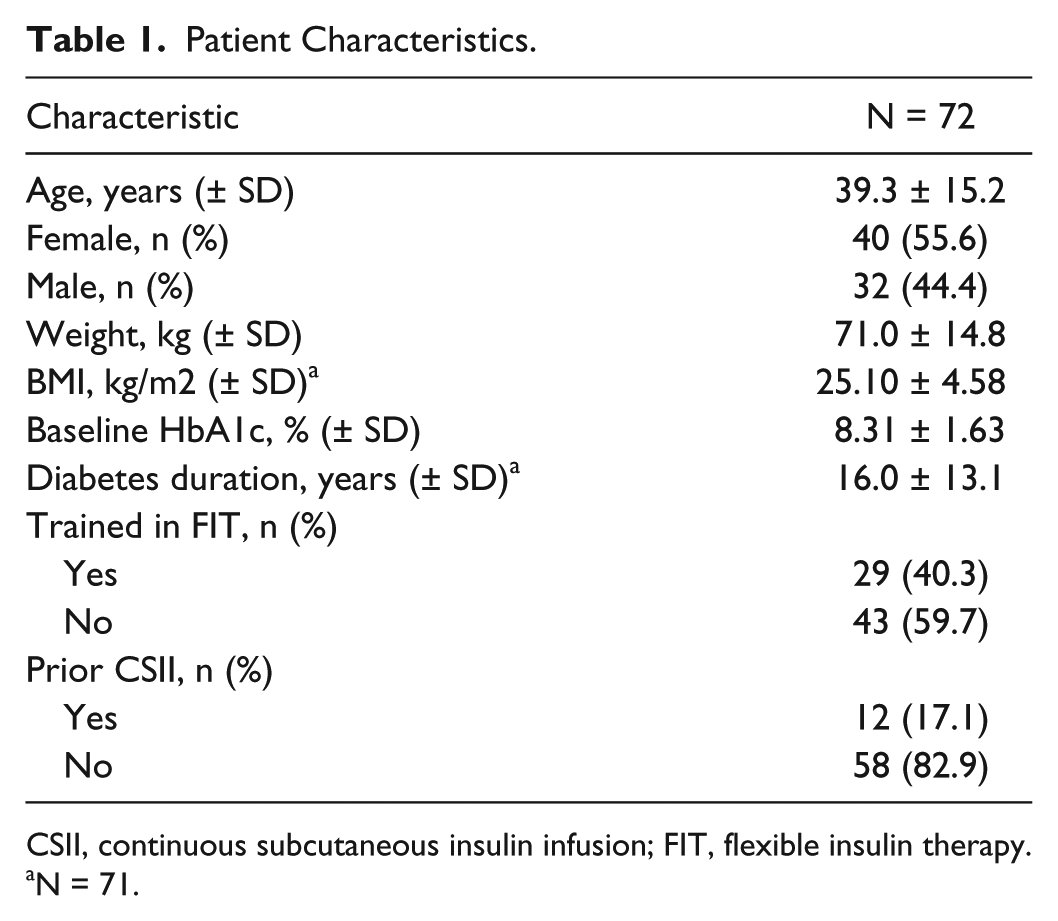
CSII, continuous subcutaneous insulin infusion; FIT, flexible insulin therapy.
N = 71.
Baseline Assessment (Visit 1)
The majority of patients had baseline knowledge of the system functions: 70 (98.6%) knew the insulin pump system provided blood glucose monitoring capability and 63 (88.7%) used this function to test their glucose. Most patients also knew that the remote control mode allowed to administrate an insulin bolus (n = 70, 98.6%), to program the temporary basal rate (n = 61, 85.9%), to suspend the pump (n = 68, 95.8%), contained an integrated e-logbook function (n = 62, 87.3%) and a bolus advice function (n = 60, 84.5%). All above data are from 71 patients.
Follow-Up Assessment (Visit 2)
The average time between visit 1 and visit 2 was 83 days (IQR: 39-71-108). No significant changes were observed in physical activity or the mean doses of insulin versus baseline, and no device-related adverse events were reported. However, mean HbA1c decreased 0.45 ± 1.13%, P = .0004 (Wilcoxon test) from baseline at study end.
Blood glucose monitoring function
Sixty-eight (94.4%) patients used the blood glucose monitoring function. Twenty-three (31.9%) patients were using this function in conjunction with another meter.
Bolus delivery function
All 72 patients used the bolus delivery feature, whereas 66 (91.7%) used the remote control feature to deliver boluses and 62 (86.1%) used the remote control feature for ≥50% of all boluses, including 46 (63.9%) who used it always. Median (IQR) VAS for the utility of the remote control bolus delivery function was 85 (74-93) (Figure 2). Among all patients who used the bolus delivery feature (remote or pump), most agreed (strongly agreed [64.8%], somewhat agreed [32.4%]) that the function was easy to use.
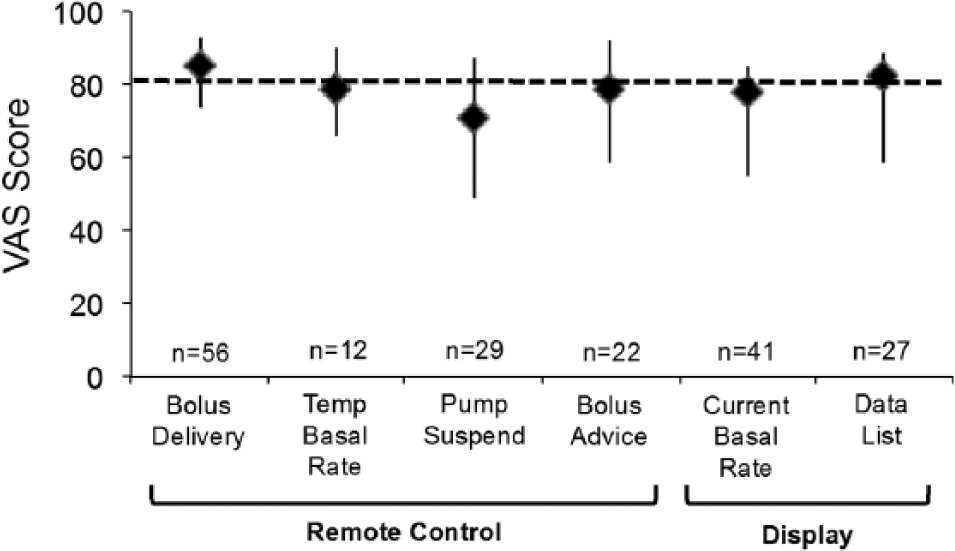
Assessment advanced function utility (VAS). VAS is scored as 0 = useless to 100 = essential. Bars indicate the 25th and 75th percentiles. Overall, VAS were available from 80% of users.
Temporary basal rate function
Eighteen (25.0%) patients used the temporary basal rate function, 14 (19.4%) used the remote control for this function, and 12 (16.6%) patients used the remote control for ≥50% of all rate adjustments, including 9 (12.5%) who used it always. Median (IQR) VAS for the utility of the remote control temporary basal rate function was 71 (49-87) (Figure 2). Among those patients who used the temporary basal rate function (remote or pump), most agreed (strongly agreed [58.8%], somewhat agreed [41.2%]) that the function was easy to use.
Suspend pump function
Thirty-seven (51.4%) patients used the manual suspend pump function; 20 (27.8%) used the remote control device for this function, including 15 (20.8%) who used the remote control for ≥50% of all pump suspensions. Median (IQR) VAS for the utility of the remote control temporary basal rate function was 71 (49-87) (Figure 2). Among the 37 patients who used the pump suspend function (remote or pump), most agreed (strongly agreed [51.5%], somewhat agreed [48.5%]) that the function was easy to use.
E-logbook function
Forty-two (58.3%) patients consulted at least 1 of the integrated e-logbook reports. The “data list” was the most commonly used report by patients (n = 35 [48.6%]); 21 (29.1%) patients stated that they viewed the report several times per week (Figure 3). Median (IQR) VAS for the utility of the “data list” report was 82 (59-89) (Figure 2), and most patients agreed (strongly agreed [48.8%], somewhat agreed [46.3%]) that the feature was easy to use. Among these patients 20 to 26 (47.6 to 61.9%) viewed the other reports (Figure 3).
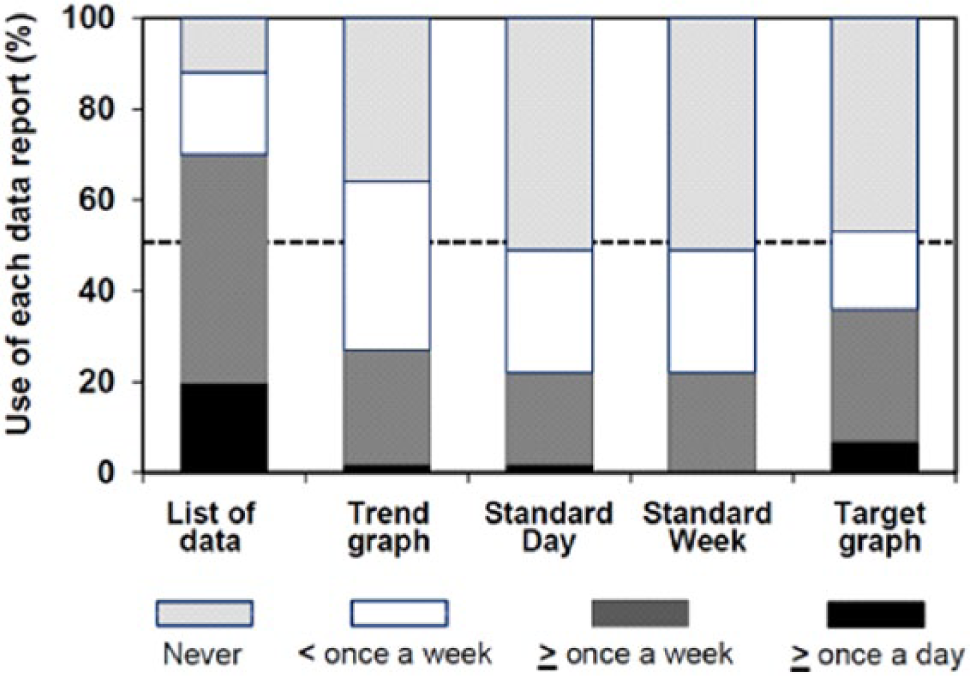
Use of integrated e-logbook reports.
Consult ongoing basal rate display
Fifty-three (73.6%) patients used the ongoing basal rate function; 22 (30.6%) consulted the display every day. Median (IQR) VAS for the utility of the ongoing basal rate function was 78 (55-85) (Figure 2). Among the patients who used the ongoing basal rate display, most agreed (strongly agreed [51.0%], somewhat agreed [49.0%]) that the function was easy to use.
Blood glucose test reminder function
Twenty-four (33.3%) patients received a reminder to perform a glucose test when they attempted to administer a bolus prior to testing; 14 (19.4%) patients received a reminder several times per week. Median (IQR) VAS for the utility of the test reminder function was 79 (60-88) (Figure 2).
Bolus advisor option
Thirty (41.7%) patients used the bolus advisor function and 20 (27.8%) used the function for ≥50% of all bolus insulin delivery, including 14 (19.4%) for all boluses. Median (IQR) VAS for the utility of the bolus advisor function was 79 (59-95) (Figure 2). Among the patients who used the bolus advisor function, most agreed (strongly agreed [56.7%], somewhat agreed [40.0%]) that the function was easy to use.
In a subgroup analysis, users of the bolus advisor function (n = 30) were compared to nonusers (n = 42). The male/female ratios were different (1:2.3 vs 1:0.8, respectively, P = .037), as was the number of users with prior insulin pump experience (9 [30%] vs 3 [7.5%], P = .013) at baseline. More bolus advisor users than nonusers also used the temporary basal rate function during the 14-day period prior to visit 2: 13 (43%) versus 5 (12%), P = .024. It is noteworthy that more bolus advisor users than nonusers received structured education on FIT, which included training in carbohydrate counting: 18 (60.0%) versus 11 (26.2%), P = .009. Among bolus advisor users who received FIT training, 61% applied sustained FIT (all meals).
Discussion
The results from this real-life study indicate a high utilization of many of the advanced functions provided by the Accu-Chek Combo insulin pump system, specifically the blood glucose monitoring option, remote control function for delivering bolus insulin, and ongoing basal rate display. Patients rated these functions as being very useful (VAS rating) and easy to use. Although use of other functions, such as the bolus advisor, pump suspension, and e-logbook, was less common, those patients who used these functions also found them to be useful and easy to handle.
Interestingly, the percentages of patients in our study who used the temporary basal rate, pump suspension, and bolus advisor functions were similar to current clinical practices that are described in several retrospective cohort analyses that assessed percentages of insulin pump patients who used these options.3,9-12 In those studies, investigators found that use of temporary basal rates ranged from 13% to 76% and bolus advisor use ranged from 16% to 54%.3,9-12 It is noteworthy that none of these reported the frequency of use of the advanced functions by users, nevertheless in a study by Walsh and colleagues, 92.7% of 396 insulin pump wearers used the bolus advisor function of their insulin pumps, routinely (≥2 times/day) to calculate their carbohydrate boluses. 13
In a recent randomized, crossover study of patients treated with insulin pumps and continuous glucose monitoring (CGM), Battelino and colleagues reported new data on the use of temporary basal rate, pump suspension, and bolus advisor functions. 8 Data were collected as 15-day downloads from patients’ insulin pumps. Utilization of each advanced function was documented prior to the end of the 2 study periods and expressed as an overall number of applications per day resulting from the practice of all study patients. Although frequency of use patterns were not reported, one could infer (from standard deviation data) wide ranges of use—from high frequency of utilization to no use. Interestingly, the use of temporary basal rates, insulin pump suspension, and bolus advisor significantly increased during the CGM period of this study.
Moreover, the higher percentage of bolus advisor users (75%) reported in a recent 6-month multicenter European observational study on the Accu-Chek Combo system suggests that both the clinic and patient expertise in diabetes and insulin pump management may be a major determinant of sustained FIT application and daily use of the bolus advisor function without CGM use. 6
Similarly, our results show that carbohydrate counting and FIT, as well as use of temporary basal rates, were applied by a significantly higher percentage of bolus advisor users than nonusers. This suggests that bolus advisor users may have greater expertise and a stronger commitment to their treatment regimens.14,15
In addition, a randomized study of a different insulin pump system reported that patients using wireless blood glucose meter-bolus advisor communication used the bolus advisor function twice as frequently than those assigned to devices without this capability. 16 This result strongly suggests that integrative device designs that reduce handling steps are an important patient need.
Some limitations apply to our study. The choice of an insulin pump device that offered a bolus advisor and remote control capabilities prior to the inclusion in the study may have created a bias toward patients who are interested in these functions. However, although this may have impacted the observed rate of use of the diverse remote control functions, it would not impact the qualitative assessment. In addition, standardized use of download software (eg, Accu-Chek Smart Pix) would have reduced reliance on patient self-reported data. The 20% missing function utility VAS might be those from patients who were less frequent users, nevertheless no exhaustive post hoc data analysis was done to test this hypothesis. Finally, this observational study was not designed to address changes in diabetes control related to the use of the insulin pump system. It should be noted that the decrease in HbA1c observed during the study period is a common change after initiation of insulin pump therapy, whereas 83% of the study patients were recently switched from MDI to insulin pump therapy prior to inclusion in the study. 17
Conclusions
In our assessment, patients with type 1 diabetes commonly used many of the multiple features of the Accu-Chek Combo insulin pump system, and patients rated the functions as useful and easy to use. The results of our study support further development and integration of advanced functions and capabilities into future insulin pump systems that will enable patients to practice more active self-adjustments of the insulin therapy and potentially improve their quality of life and metabolic control.
Footnotes
Acknowledgements
Results were presented at the 13th Diabetes Technology Meeting, San Francisco, October 30 to November 2, 2013. We express our gratitude to all patients and investigators who participated in the study. We would also like to thank Patrick Brunet-Lecomte from Eurofins Optimed Clinical Research (Gières, France) for the study data management and statistical analysis. Combo Study group: A. Brac de la Perriere (CHU Lyon), AH. Brinkane (CH Meulan), C. Chaussade (CH Perigueux), S. Clavel (CH le Creusot), P. Colin (CH Niort), E. Ghanassia (Sète), H. Grulet (CH Châlons-en-Champagne), F. Guillon-Metz (Caen), S. Hieronimus (CHU Nice), I. Hochner (Strasbourg), K. Lachgar (CH Eaubonne Montmorency), V. Le Galudec (Ilkirch-Graffenstaden), AS. Lerman (Metz), J.F. Martin (CH Le Mans), G. Matejka (Cabestany), P. Mattei (CH Saint Dié), L. Molines (CHU Marseille), F. Ouliac (La Seyne sur Mer), R. Piquemal (CH Blois), MF. Poncet (Albi), E. Sonnet (CHU Brest), and B. Trabulsi (CH Pontivy), France.
Abbreviations
CGM, continuous glucose monitoring; FIT, flexible insulin therapy; HbA1c, glycated hemoglobin; IQR, interquartile range; MDI, multiple daily injections; SD, standard deviation; VAS, visual analog scale.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RB is an employee of Roche Diagnostics, France. MP received fees from Roche Diagnostics France as study coordinating investigator, honoraria as a speaker or consultant from Eli-Lilly, Medtronic, Novo-Nordisk, Roche Diagnostics, Sanofi-Aventis, Ypsomed, and research funding from Novo-Nordisk and Sanofi-Aventis. KL has received honoraria as a speaker or consultant from Lilly, Medtronic, Novo-Nordisk, Roche Diagnostics, Sanofi-Aventis, BMS, MSD, Novartis, Lifescan, and Menarini. CGP has received consulting fees from Roche Diagnostics, CeQur, and Asante. The study investigators received remuneration from Roche Diagnostics France. JW is an employee of Roche Diagnostics Deutschland GmbH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Roche Diagnostics France, Meylan, France.
