Abstract
Background:
Diabetes disproportionately affects the US Latino population, due to socioeconomic pressures, genetics, reduced access to care and cultural practices. While efforts to improve self-care through interventions incorporating family are highly rated by Latinos, family can be both supportive and obstructive. To develop effective interventions, this role needs clarification.
Methods:
We conducted group interviews in Spanish and English with 24 participants with diabetes from a mobile health diabetes self-care intervention. We imported transcripts into Dedoose, a qualitative computer analysis program and analyzed them with a modified grounded theory technique. Utilizing an iterative process, we reexamined transcripts with new codes derived in each round of analysis until saturation was reached. We employed techniques to improve trustworthiness (co-coding, member checking). Broad categorical themes arose from the initial codes and were developed into a conceptual model of barriers to and strategies for diabetes management.
Results:
Family and family responsibilities emerged as both a supportive and obstructive force for diabetes self-care. While the desire to care for family motivated patients, food at family gatherings and pressure from managing multiple family responsibilities contributed to poor diet choices. Yet, some patients believed their diabetes caused their immediate family to make healthier choices.
Conclusions:
Among these predominantly Latino patients, family and family responsibilities were key motivators as well as obstacles to self-care, particularly regarding nutrition. Finding the ideal design for social support mHealth-based interventions will require careful study and creation of culturally based programs to match the needs of specific populations, and may require educating family members to provide effective social support.
Although diabetes is a nationwide epidemic, US Latinos are a particularly vulnerable population with higher prevalence of disease and rates of complications than non-Hispanic Whites.1,2 Many factors contribute to this inequality, includinggenetic, socio-economic pressures, reduced access to care and cultural practices.3-6 Diabetes self-management and education interventions have successfully improved diabetes self-care and glycemic control, and may be more effective when tailored for vulnerable populations.7-9 SMS (short message service)text-message-based mobile health interventions are a potential solution to combat this health disparity, given the low cost of implementation and scalability. National estimates indicate most low income Latinos have mobile phones capable of receiving basic SMS messages, 10 but app use among Latinos with diabetes is low. 11 mHealth (mobile health) interventions have great heterogeneity in outcomes, and most have had modest treatment effect.12-16 One avenue to increase the effectiveness of mHealth behavioral interventions for Latinos with diabetes is to harness the power of familismo or the cultural practice of giving family priority, which is common among US Latinos. 17 By incorporating family members into diabetes education and behavior change interventions, diabetes self-management and education interventions may become more powerful.
Higher baseline levels of social support are associated with better diabetes self-care and glycemic control; 18 in addition, larger support networks are associated with improved diabetes self-care.19,20 Social support interventions for diabetes often focus on improving knowledge of diabetes among family members and engaging these key social support providers into a patient’s health care and self-care. 18 Interventions that increase social support for patients with diabetes by educating and activating family members may subsequently improve a patient’s diabetes self-care. Latinos in particular have rated these types of interventions highly,21-23 but the literature is not consistent in demonstrating the benefits of support from family and friends. Some social support interventions have shown improved outcomes, while others have shown no effect. 24 Furthermore, social support can be both supportive and obstructive to good self-care choices, particularly in vulnerable populations.25,26
Explanations for these inconsistent results from predominantly quantitative studies include heterogeneous interventions, dissimilar patient populations, and the mixed effect of family for different individuals. If social support strategies will be incorporated into mHealth interventions, these differences in changes in self-care behaviors and glycemic control must be explored.
Through an inductive, qualitative analysis of focus group interview transcripts, we gained a deeper understanding of the relationship between social support and diabetes management among low income, predominantly Latino patients. We conducted group interviews with patients who had participated in a patient-focused diabetes intervention, originally planned to explore general barriers to diabetes management and strategies to overcome obstacles. During our qualitative analysis, the role of family and close friends in patients’ self-care decisions arose repeatedly in all group interviews. Family (including close friends) emerged as an important component of patients’ self-care, emphasizing the importance of understanding the role of family in this population. Consistent with recent literature,26,27 some of the themes regarding family were supportive and others were obstructive to good self-care.
Subjects, Materials, and Methods
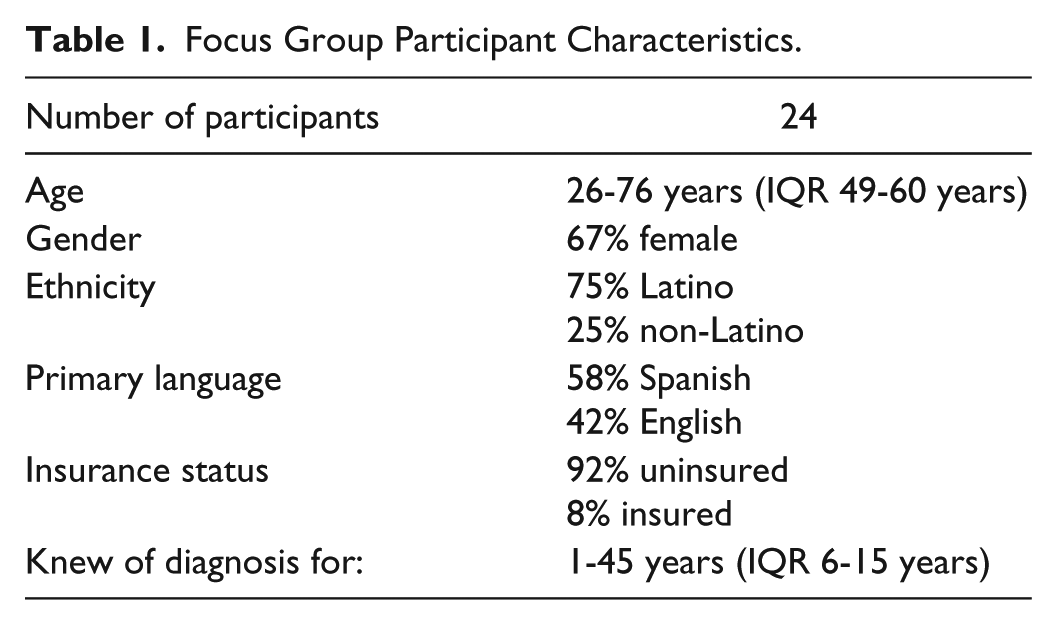
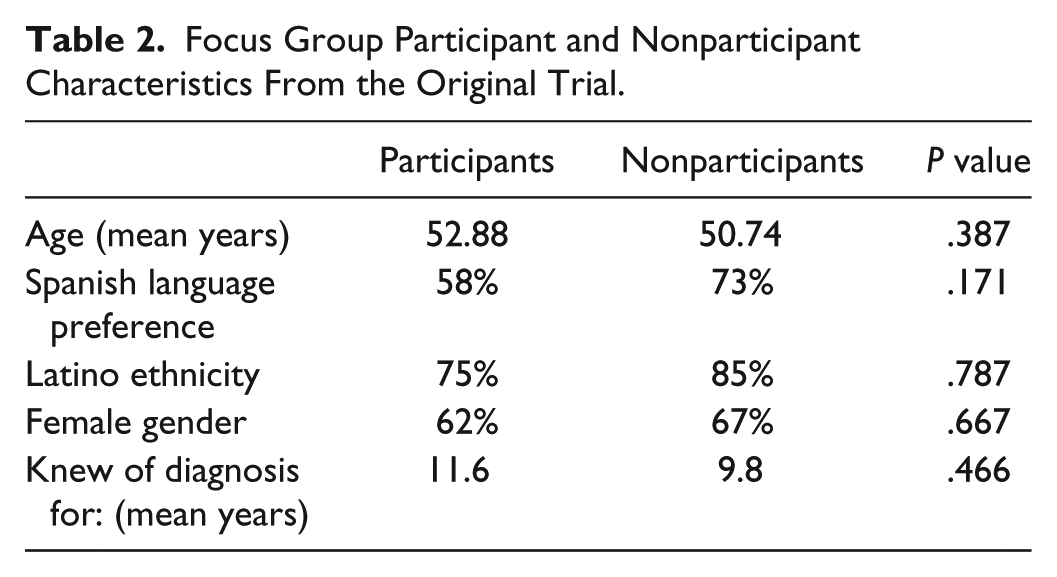
In this qualitative study, we conducted a series of focus group interviews with patients with diabetes within two months of completing a text-message-based educational and motivational program. 28 The purpose of the interviews was to examine the barriers to self-care activities and the strategies patients use to overcome them, and if patients perceived that the intervention had changed these strategies. All 47 patients who completed the intervention were invited to participate in the focus groups through a series of phone calls and text messages. All 24 patients who agreed to participate in the focus groups were compensated for their time and travel. Participants were predominately, Latino, female and uninsured (see Table 1). Those patients who participated in the interviews did not differ from those who did not age, ethnicity, gender, language preference, or years with diabetes (see Table 2.) The focus groups were stratified by language and by gender to improve the comfort of participants with sharing life experiences. 29 At the start of each focus group, an internally developed anchor survey was administered (Appendix A) and healthy dinners were provided to ease the start of conversation. 29 Groups lasted between 90 to 120 minutes, and a semistructured question guide was used (Appendix B). The interview guide was developed through three rounds of piloting and revision with two experienced qualitative researchers. Experienced, bilingual team members facilitated the focus groups. Focus groups were audio-recorded, transcribed, and translated in the case of Spanish-language groups. These transcripts were uploaded to Dedoose®, a web-based qualitative analysis program. 30 This study was approved by the local Institutional Review Board prior to initiation.
Focus Group Participant Characteristics.
Focus Group Participant and Nonparticipant Characteristics From the Original Trial.
We used a modified grounded theory analysis approach, an iterative process of coding and immersion in the data to recognize the patterns and links between the concepts voiced by the participants. A grounded theory approach focuses on the descriptions and perceptions of interview participants and other sources of qualitative data while trying to minimize outside theories and biases. 31 By choosing a qualitative method that allows for this more granular analysis of data, the subtle and complicated components of a phenomenon can be more clearly reconstructed. Three project team members (EREB, MR, KK) independently completed line-by-line open coding of two of the transcripts to maintain the detail of the data. An audit trail including memoing was maintained to document all analytic decisions to increase trustworthiness (the qualitative analogue of validity). 31 Through an iterative process of coding transcripts and discussing the coding schema, we developed a set of codes from the recurring ideas that emerged from the data. After the development of the preliminary set of codes, we conducted multiple rounds of co-coding until consensus on codes definitions was achieved. A test of coders’ agreement on application of these codes resulted in a Cohen’s pooled Kappa coefficient of greater than 0.7, showing good intercoder reliability. 32
After development of the codebook, we coded all transcripts through focused coding with the final codebook, while also checking to see if new codes needed to be created to capture new emerging themes. Saturation of codes was achieved by the fourth focus group of the five total focus groups, when no further codes were developed during analysis. We then used a technique known as axial coding, 31 comparing the situations where codes overlapped and the cases where these connections did not occur. Broad categorical themes arose from the initial codes and were developed into a conceptual model of barriers and strategies used to manage diabetes. This resulted in grounded descriptions of participants’ perceptions of their diabetes and how their life situation impacted it, and vice versa. Our final codebook contained 27 codes and 24 subcodes. We produced 327 pages of transcripts, which were read by three members of the research team. A total of 759 individual excerpts were coded with between 1 and 11 codes each. We conducted member checking by presenting the preliminary findings to a group of Promatoras, community volunteers who assist with health education and basic interventions, who reviewed our preliminary findings and compared the findings with their lived experiences with patients who had diabetes or their own experience with diabetes. The community health workers gave us additional insights and clarification into the role of social support in patients’ chronic disease management, which were incorporated into our understanding of family and diabetes self-care.
Results
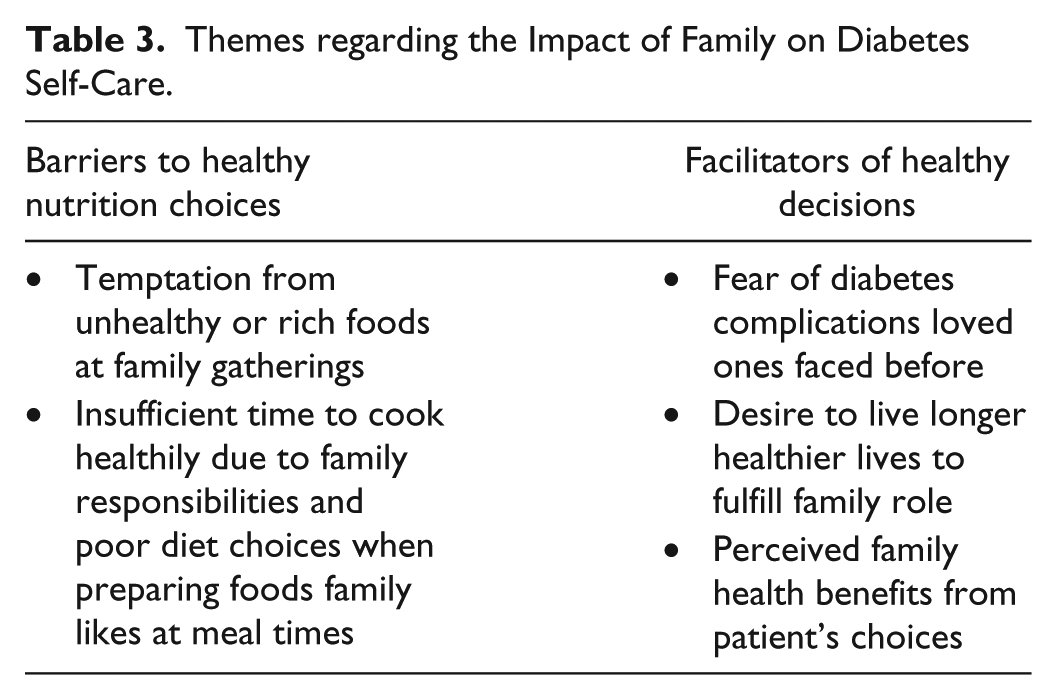
Family emerged as an important modifier in diabetes management, but with complex relationships affecting motivation and behavior choices. In some instances, family was viewed as a positive force, and in others situations family was viewed as obstructive to good self-care. Predominant themes (see Table 3) were (1) desire to care for family increases motivation (5/5 group interviews), (2) family as cautionary examples of poor diabetes management (5/5 group interviews), (3) nutrition challenges at family gatherings (3/5 group interviews), (4) balancing family responsibilities with healthy nutrition choices (2/5 group interviews; 2/3 female interviews), and (5) making healthier nutrition choices as a family (3/5 group interviews).
Themes regarding the Impact of Family on Diabetes Self-Care.
Desire to Care for Family Increases Motivation
Patients identified their relationship with their family members as critical to changing lifestyle habits to improve diabetes related behavior, because of the importance of family roles. This was the one of the most frequently identified theme, emerging from analysis from all 5 groups. Patients wanted to be healthy and physically present for their family members, particularly if they had young children and grandchildren. This desire helped many patients overcome the fatalism or denial that prevented changing diet and exercise patterns. Fear of leaving family members unsupported due to early death or disability was also a compelling motivator. However, patients did not frequently express that their family members provided direct support, advice or encouragement that helped them improve their health behaviors. Most patients spoke Spanish, so the excerpts are translated into English. Patients noted:
What motivates me is my children. To take care of them and myself, too. (Translated from Spanish) I have three grandchildren and I am 47 years old. I have more to do. (Translated from Spanish) Well at the beginning when I started with my illness, I would not take care or anything. . . . And now as time has gone by . . . I begin to think that if I don’t take my medicine. . . . I will not see my children grow. And that is what I asked God for. Just to see them grow. (Translated from Spanish)
Family as Cautionary Examples of Poor Diabetes Management
Family members and friends also provided indirect inspiration by serving as examples of poorly controlled diabetes. This was also a frequently identified theme, present in 5/5 group interviews. Early death of family members who had diabetes as well as complications such as neuropathy caused patients to assess their own health choices and to become more informed about their disease. This exposure to the risks of uncontrolled diabetes helped patients identify the link between their self-care, glycemic control, and eventual physical manifestations of poor diabetes management. They explained:
I thought of my grandmother who died of diabetes, and my mother of a heart attack, then I would say, okay, I don’t want to follow in the same circle that they were in, I have to find more information, I have to find other ways to help. (Translated from Spanish) What motivates me is that my mother died of the same thing. I say to myself, if I don’t take my medicine the same thing will happen to me. That is what motivates me. My mom couldn’t even walk no more. Her feet were just so swollen. Just started turning crooked, her nerves were just damaged, all the sugar just started drying up your nerves. That’s ugly.
Nutrition Challenges at Family Gatherings
Patients identified family as sources motivation, but also as presenting challenges. Patients experienced expectations at family gatherings as sources of dietary temptation. This perception of dietary challenge was identified in 3 out of 5 group interviews; all 3 were female participant group interviews; neither of the male participant group interviews identified this issue. The presence of food that was especially rich or had special emotional connotations created challenges to nutrition plans, even without explicit pressure to eat foods that did not fit into a patient’s nutrition plan. Some patients resorted to avoidance of social events or dietary bargaining to balance the nutrition challenges they faced with their desire to make healthy choices. This was more common in extended family gatherings, such as birthdays or large family dinners. As patients explained:
I have a big family. We have birthday parties, barbecues, . . . if I’m gonna have a piece of that cake, I know what it’s gonna do to me . . . I have to choose. What poison do I want? Do I want the barbecue sauce that’s full of seasonings and sugar and all that other—or do I want the cake? And I tell them all the time, I can’t eat like you. So if you’re gonna invite me over to dinner, you better have vegetables or a salad, baked chicken. They know, believe me, they can tell me better what I’m supposed to eat.
Balancing Family Responsibilities with Healthy Nutrition Choices
Outside of food temptations at social events, family and family responsibilities presented other obstacles to making healthy food choices. Patients noted that a rushed lifestyle and managing the needs of multiple family members created time pressure that made undermined nutrition choices. Food preparation responsibilities and expectations of family meal times also created difficulty in making healthy nutrition choices. This theme was identified in 2 of 3 group interviews with female participants and in none of the interviews with male participants. Patients noted:
I’m hungry, my kids are hungry. Oh, we gotta hurry. Go watch a movie. Because it’s the first day. So, oh, it’s gonna take me a long time to cook right? Okay, you know what McDonald’s, Jack-in-the-Box. Fast food. My habit was that I would eat a huge plate of food at four, five, six in the afternoon. That was a time when my husband got home, and that was a time that I would have my meal. So I had to change that—and I had to make my husband change because he would say, you no longer sit to eat with me. Okay, I will sit here with you but I cannot eat because I already ate at my given time. So then I could not eat at that hour anymore. (Translated from Spanish)
Making Healthier Nutrition Choices as a Family
While the predominant way families interacted with nutrition decisions was detrimental for patients, some patients believed their diagnosis of diabetes helped their immediate family members make healthier nutrition choices. This was identified in 3 of the 5 group interviews, which were all Spanish-language interviews; this theme was not identified in any of the English-language interviews. They noted family members leading healthier lives when a diabetes diagnosis forced everyone to make better nutrition choices. In addition, patients noted that they took better care of themselves when caring for family members who also had diabetes, especially when meals were shared, as the following statements illustrate:
Logically when one has diabetes, well everybody has to know. It is like the whole family has diabetes because your eating habits change, your habits, and in that respect thanks be to God, because at least our children begin to eat healthier. But when one gets diabetes, is not just you. In reality, the whole family comes into it. (Translated from Spanish) You know he [patient’s husband who also has diabetes] eats the things that he not supposed to. What I learned is that dealing with him helped me deal with mine because that last [time] I went to the doctor, he told me that whatever I was doing, to keep doing it because my thyroid was controlled, my diabetes was controlled, and I had my liver swollen for too many years and they said it was okay. (Translated from Spanish)
Discussion
Social support has largely been considered a positive force on diabetes self-management. However, recent research has suggested that not all support is equal, and that some types of support can be detrimental. This inductive, qualitative study of urban, low-income patients who completed a mHealth intervention for diabetes allows for a deeper exploration of the complicated role of family as both support and obstacle. While living up to family responsibilities motivated patients to make healthy behavior changes, food at larger family gatherings and the pressures of managing family responsibilities contributed to poor choices for patients. However, some patients believed their diabetes caused their immediate family to make healthier choices. Activating family members may augment mobile health interventions, but only if supportive behaviors are emphasized.
Many diabetes experts and clinical diabetes educators encourage support from family members to improve self-care behaviors, acknowledging the powerful influence of loved ones over patients.17,33,34 Traditionally, social support has been viewed as positive, as most literature shows positive associations between increased social support and improved self-care behaviors and glycemic control.35-37 In addition to encouraging good self-care, strong relationships with family have been found to improve psychological and emotional well-being of patients, which independently are associated with improved self-care and glycemic control.38,39 While family offers important support and drive internal motivation for patients, recent literature has shown that the quality of support is important when trying to improve patient outcomes. Recent work by Mayberry and colleagues has elucidated nonsupportive behaviors that were detrimental to patients’ self-care.25,26,40 Patients in these studies noted difficulties with obstructive behaviors regarding dietary choices. While the intent of family members may be supportive, some behaviors resulted in making patients healthy choices more difficult. In particular, patients with low health literacy were prone to poorer glycemic control when faced with obstructive family behaviors, such as sabotaging nutrition choices. 40 Nutrition choices are key to better lifestyle of good self-care, and must be emphasized to both patients and family members.
In this study, the role of family in improving patients’ self-care was passive; we must activate family members and friends in addition to patients. Patients’ desire to care for their family and wish not to develop the same complications as ill family members motivated improving self-care. While family members were not specifically interviewed in this study, previous studies indicate that family members feel they lack the expertise to provide good care and this can be an obstacle for family member to give advice or support lifestyle changes. 41 Future mHealth interventions could overcome this barrier with a diabetes educational curriculum for family members to correct misperceptions about proper diabetes self-care. There may also be a role for teaching family members to provide positive coaching rather than nagging their loved ones. 40 In addition, family members could be encouraged to work with the identified patient to rid the home of unhealthy food options, to exercise with patients or to provide tangible social support such as picking up medication refills, or scheduling doctors’ appointments. mHealth is a promising strategy to engage family members, as it removes the barrier of travel and time required of family members to be trained.
Prior work has shown patients to be highly receptive to incorporating family members into mHealth and telehealth interventions with promising results in self-care.37,42 However these investigations have been limited by poor recruitment of supporters and these kinds of interventions will require researchers, program planners and designers to find ways to engage a family member in a disease that may not directly impact them. We found gender and language differences in the role of family in diabetes self-care, consistent with prior findings that social support interacts with disease management support differently between genders,5,43 In addition, there is emerging evidence that women and men respond differentially to mobile interventions to increase social support. 44 Intervention designers must consider that activating the family of a female patient may require different strategies than the family of a male patient.
Our study has several limitations. The study from which these patients were selected had participated in a 6-month educational intervention study that was delivered via mobile text messages to encourage and challenge patients to make positive behavior changes, and we did not interview the “usual care” controls from that trial. Those who participated in the focus groups may represent a biased sample of the initial study population. Patients who came to the focus group interviews were likely those who were most engaged in the intervention. Also, these focus groups were conducted with an inductive aim and purposive sampling; the findings are not generalizable in isolation, and require an understanding of context. This approach, however, allowed us to explore the complicated relationship between social support and diabetes self-care. Moreover, our findings triangulate with prior literature and add to our understanding of social support and diabetes self-care among Latinos, particularly in regard to possible mHealth interventions.
For this population of predominantly Latino patients, family roles and responsibilities were key motivators as well as obstacles to diabetes self-care, particularly regarding nutrition choices. Our findings highlight that while family and loved ones can be powerful motivators for change, the patterns of diet choices and behaviors perpetuated by family members can be harmful when patients are trying to make self-care modifications. Finding the ideal mechanisms to augment social support for patients that already have a strong network and how to create a positive support system for patients that lack one will require careful study and the creation of culturally synchronous interventions to match the needs of specific populations. Reaching these key supporters is also critical; incorporating low-cost communication via mobile devices may increase the reach of interventions to people who do not have the time or transportation to accompany their loved one to a visit to a medical visit. Harnessing the power of these existing social networks through new communication technologies such as text messaging and online social media websites may be the key to improving diabetes self-care among the most vulnerable.
Footnotes
Appendix A
Anchor Survey
Name: _______________________________________
Age: _________________________________________
Race: □ White □ Black □ Asian/Pacific Islander □ American Indian
□ Other □ Decline to State
Ethnicity: □ Latino □ Non-Latino □ Decline to State
Cellphone number: _____________________________
How long have you had diabetes? ________________
How long have you had a cell-phone? ________________
Do you have a regular telephone at home as well? ? □ Yes □ No
In your family, do people share a cellphone? □ Yes □ No □ Sometimes
How many people share the cellphone you use?_________
Do you keep your cellphone with you all of the time? □ Yes □ No
Where else do you leave it?
□ In a purse
□ The car
□ On a table,
□ Other ________
How often do you receive text messages?
□ Less than once a week
□ Once a week
□ Once a day
□ Up to 5 times a day
□ More than 5 times a day
How often do you send text messages to other people?
□ Less than once a week
□ Once a week
□ Once a day
□ Up to 5 times a day
□ More than 5 times a day
Appendix B
Acknowledgements
We are grateful to the patients who participated so willingly in the study. We acknowledge Adriana Berumen for assistance with data collection and organization of focus groups.
Abbreviations
mHealth, mobile health; SMS, short message service.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a mobilizing for health grant from the McKesson Foundation and the National Institutes of Health (SC CTSI NIH/NCRR/NCATS UL1TR000130, NIDDK 1F32DK094547, NIDDK 1K23DK106538, SC CTSI NIH/NCRR/NCATS KL2TR000131).
