Abstract

In a 2014 article published in this journal, Waki et al 1 demonstrated that a smartphone-based self-management support system for patients with type 2 non-insulin-dependent diabetes, called DialBetics, decreased HbA1c by 0.4% compared with standard diabetes care. The system provides a real-time, partially automated, interactive system, which delivers interpretation of the patients’ data. These include biological parameters and information on exercise and dietary content, the latter being calculated from data imputed by patients. Appropriate actionable information is then forwarded by the system to individual patients. The goal is to help patients in achieving diabetes self-management.
Following the Japanese experience, we conducted an observational, prospective, nonblinded, longitudinal clinical trial, designed to test this system in 35 Italian patients with type 2 diabetes. The study was conducted in collaboration with the University of Tokyo at Istituti Clinici Scientifici Maugeri in Pavia, Italy.
Compared with the Japanese cohort, Italian patients had a similar age and male/female ratio, but a shorter duration of diabetes and a greater BMI. Similar to the Japanese cohort, Italian patients showed an HbA1c above 7%.
The study was designed with a 3-month intervention period, followed by a 3-month standard care period. After the intervention period, mean HbA1c, body weight, waist circumference, and systolic blood pressure significantly decreased by 0.47%, 1.8 kg, 3.5 cm, and 8.2 mmHg, respectively. A not significant lowering trend of serum LDL-cholesterol was evident. The 10-year coronary heart disease risk was significantly reduced (Table 1). As assessed by the Summary of Diabetes Self-Care Activities questionnaire, during the intervention period the patients’ compliance to diet and physical activity increased in 34% and 54% of cases, respectively. No change was perceived in quality of life. Because readings were telemetrically transferred to the team, the dose of antidiabetes medication was immediately adjusted: none of our patients experienced hypoglycemia. In two patients the dose of antidiabetes drugs was decreased, and in one sitagliptin was stopped. The system was perceived as useful and easy to use; the dropout rate was 8%.
Changes in Clinical and Laboratory Outcomes from Baseline (T0) to 3 Months (T1 = Intervention) and 6 Months (T2 = Follow-Up Observation).
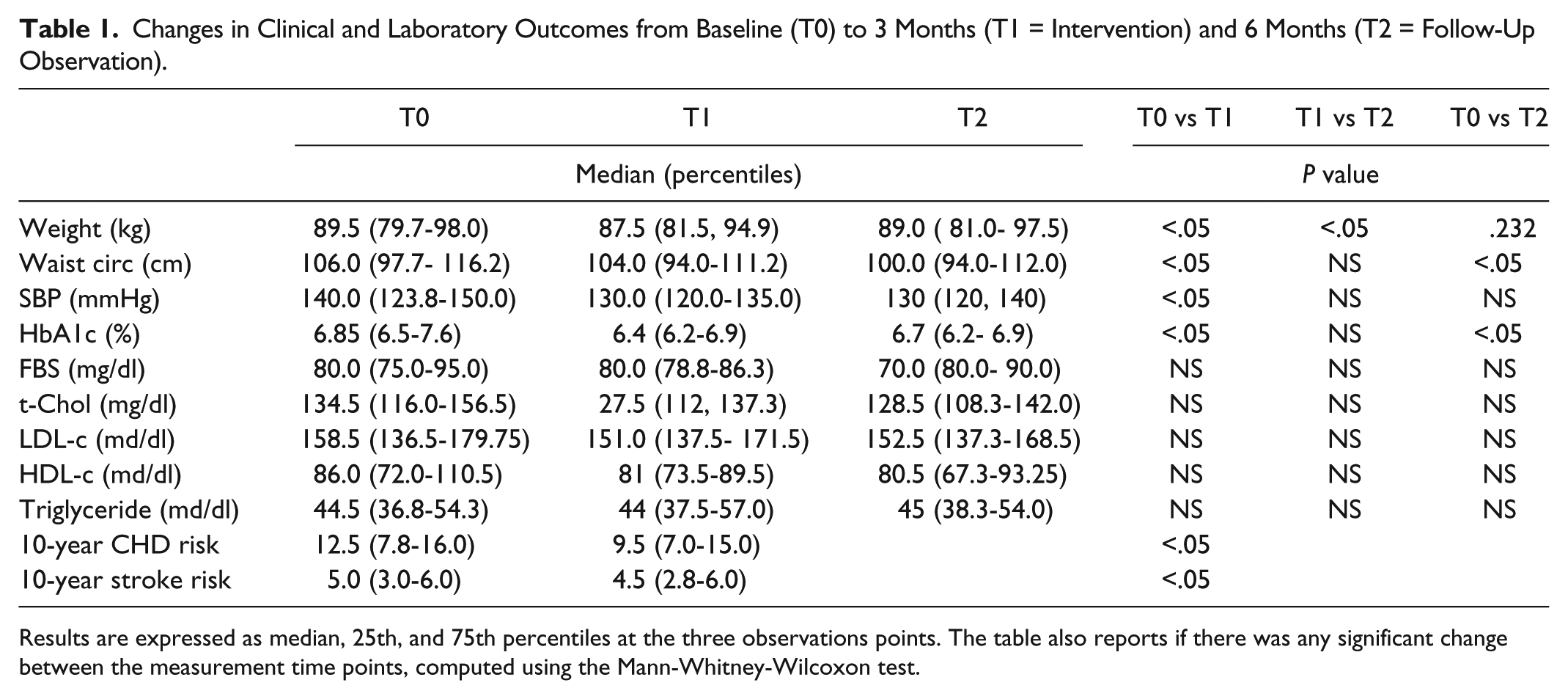
Results are expressed as median, 25th, and 75th percentiles at the three observations points. The table also reports if there was any significant change between the measurement time points, computed using the Mann-Whitney-Wilcoxon test.
This study has several limitations: the sample size was small, the design was not randomized or controlled, and the follow-up period was short. Moreover, our patients maintained during the observation period the HbA1c reduction, obtained during the intervention period, but regained the lost body weight (37% of them regained > 1 kg in 3 months). This suggests a short effect for the utilization of the system and reflects a low efficacy of this system in maintaining its benefits in the long term. Thus, only a continuous utilization of the support system is effective.
In conclusion, we showed that our system, originally devised for a Japanese population, could be a safe, useful, and interactive tool to optimize the metabolic control also in an Italian population of patients with non-insulin-treated type 2 diabetes. This system would be useful for reaching ambitious, stricter glycemic targets and for avoiding the intensification of medical treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
