Abstract
Background:
A 2017 umbrella review defined the technology-enabled self-management (TES) feedback loop associated with a significant reduction in A1C. The purpose of this 2021 review was to develop a taxonomy of intervention attributes in technology-enabled interventions; review recent, high-quality systematic reviews and meta-analyses to determine if the TES framework was described and if elements contribute to improved diabetes outcomes; and to identify gaps in the literature.
Methods:
We identified key technology attributes needed to describe the active ingredients of TES interventions. We searched multiple databases for English language reviews published between April 2017 and April 2020, focused on PwD (population) receiving diabetes care and education (intervention) using technology-enabled self-management (comparator) in a randomized controlled trial, that impact glycemic, behavioral/psychosocial, and other diabetes self-management outcomes. AMSTAR-2 guidelines were used to assess 50 studies for methodological quality including risk of bias.
Results:
The TES Taxonomy was developed to standardize the description of technology-enabled interventions; and ensure research uses the taxonomy for replication and evaluation. Of the 26 included reviews, most evaluated smartphones, mobile applications, texting, internet, and telehealth. Twenty-one meta-analyses with the TES feedback loop significantly lowered A1C.
Conclusions:
Technology-enabled diabetes self-management interventions continue to be associated with improved clinical outcomes. The ongoing rapid adoption and engagement of technology makes it important to focus on uniform measures for behavioral/psychosocial outcomes to highlight healthy coping. Using the TES Taxonomy as a standard approach to describe technology-enabled interventions will support understanding of the impact technology has on diabetes outcomes.
Keywords
Introduction
Multiple umbrella reviews identify significant improvement in clinical outcomes, including hemoglobin A1C (A1C) when technology is part of the model of care.1-4 These technologies include diabetes devices (eg, connected pens, glucose monitors and continuous glucose monitors), mobile devices (eg, mobile applications, wearables, fitness trackers) and technology-enabled communications (eg, text messaging, 2-way chat). In a 2017 review of high-quality systematic reviews, a framework evolved identifying 4 key elements of technology-enabled interventions associated with significant change in A1C. 1 The technology-enabled self-management (TES) feedback loop includes communication between the care team and people with diabetes (PwD); transmission and analysis of patient generated health data (PGHD); general or tailored education based on ADCES7 Self-Care Behaviors™, 5 informed by PGHD; and individualized PGHD feedback delivered in real-time or asynchronously by technology or care team. 1
As technology has evolved, digital/virtual programs focused on chronic condition management and supported by employers or health systems emerged (eg, Onduo, Livongo®, BlueStar®) that implement the TES feedback loop elements to provide care, education, and support. Additionally, the Association of Diabetes Care & Education Specialists (ADCES) has continued to define the role of the diabetes care and education specialist in the integration and implementation of technology-enabled self-management solutions. 6 While achieving target A1C is an essential piece of the puzzle, it is important to understand the type of technology, design of the interventions, user experience, and workflow, which influence glycemic, other physiological, and behavioral/psychosocial diabetes self-management outcomes.7,8 Recent systematic reviews have evaluated the impact of technology in diabetes care and education. While numerous authors cite the 2017 review, most focus on the association of technology with lowering A1C. Since 2017, the National Standards for Diabetes Self-Management Education and Support 9 and the American Diabetes Association (ADA) 10 Standards of Care (SOC) have recognized the TES framework for effective technology-enabled interventions. However, it is unclear if the TES framework is being used as a guide to develop and evaluate technology-enabled interventions.
The purpose of this umbrella review was to (1) develop a taxonomy to describe intervention attributes in technology-enabled interventions; (2) review recent, high-quality systematic reviews and meta-analyses to determine if the elements of the TES framework were described and whether the elements continue to contribute to improved diabetes outcomes; and (3) to identify gaps in the literature regarding technology-enabled diabetes self-management.
Methods
Phase 1 Taxonomy Development
We reviewed the TES framework from the 2017 review 1 and identified additional key technology attributes needed to describe the active ingredients of technology-enabled interventions that lower A1C levels. As an exemplar, we applied the medication full prescribing information (FPI) 11 to define the detailed attributes of technology-enabled interventions, including indications, class, mechanism of action, active ingredients, dose, route, frequency, duration, and adverse effects.
The attribute definitions were evaluated for appropriateness and completeness in a 2-step process by applying the definitions to 3 representative technology studies.12-14 First, 2 authors applied the definitions to each study, then a third author reviewed for discordance. The definitions were then revised and applied to the same studies in a second review. Figure 1 shows the application of the definitions to the studies.
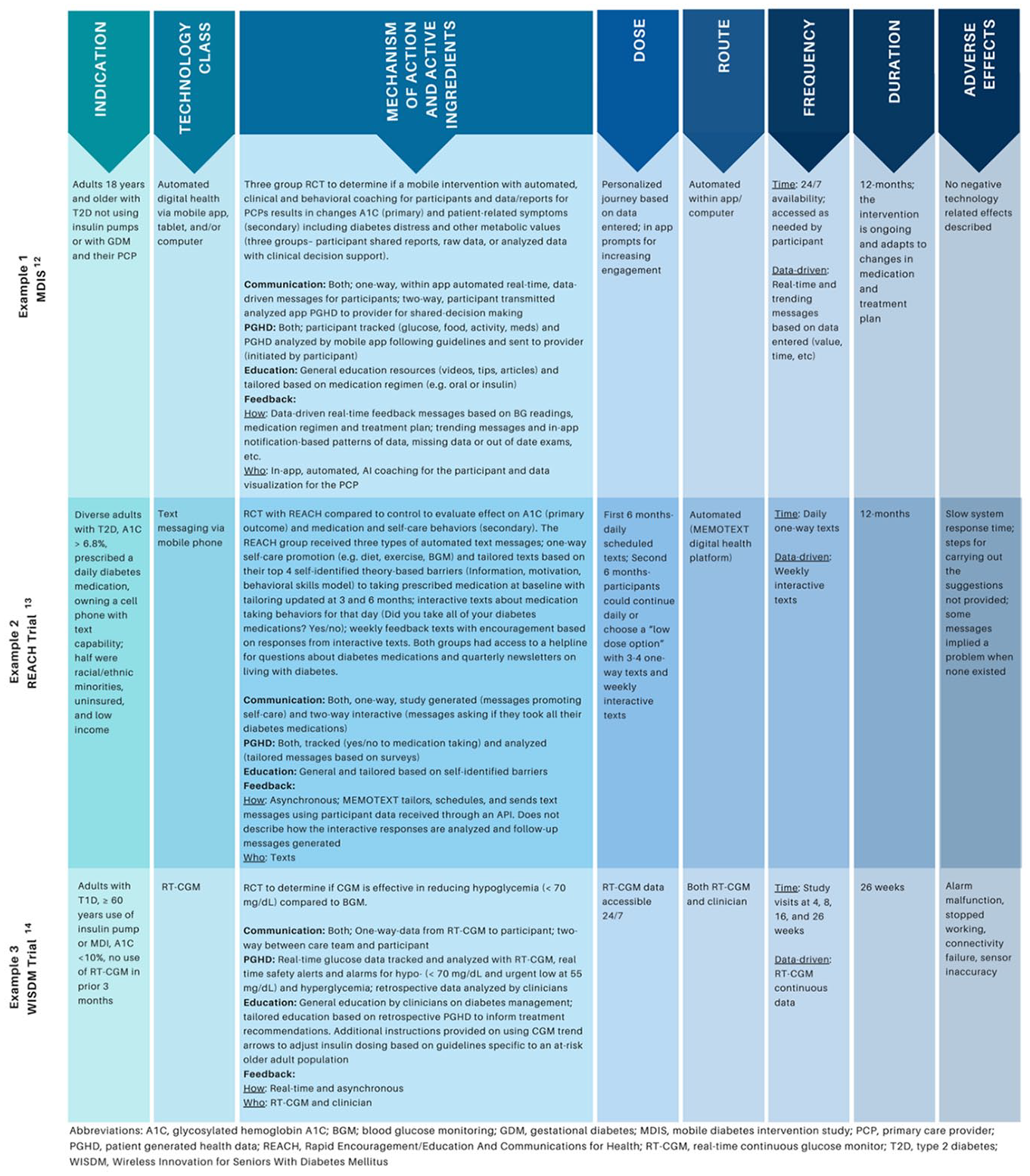
Taxonomy definitions applied to exemplar studies.
Phase 2 Umbrella Review
We identified parameters a priori and searched for English language reviews published between April 2017 and April 2020, focused on PwD (population) receiving diabetes care and education (intervention) and using technology enabled self-management in randomized controlled trials (comparator) that impact, glycemic, other physiological, and behavioral/psychosocial outcomes. A medical librarian searched multiple databases using subject headings and text words related to technology, diabetes mellitus, self-management, self-care or patient education, and systematic reviews or meta-analysis following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations. 15 See Figure 2 for the PRISMA diagram.
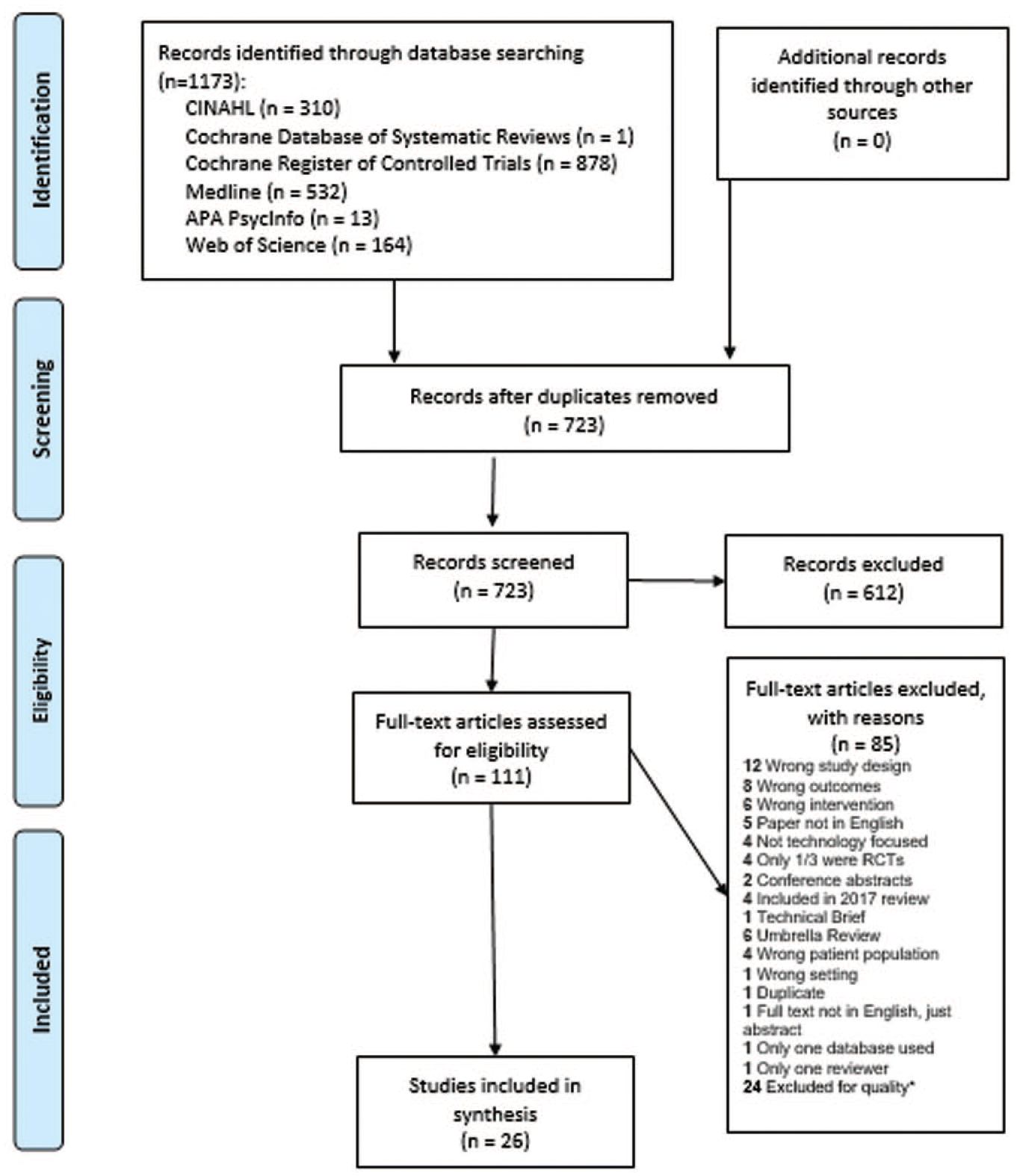
PRISMA flow diagram 2009.
Sources, Searching, and Review
Cumulative Index to Nursing and Allied Health Literature (CINAHL), Cochrane Database of Systematic Reviews, Cochrane Register of Controlled Trials, MEDLINE Complete, APA PsycInfo, and Web of Science were searched in April 2020 and repeated in June 2020 to retrieve current literature. (See Supplemental Appendix A). After identifying duplicates, reviewers screened abstracts from 723 articles specific to technology-enabled diabetes self-management or education interventions. Two independent reviewers screened each abstract (DI, ML, JB, JD, VC). A third reviewer (DG or MP) resolved disagreements. A total of 111 full text articles (US and international) met inclusion criteria. Reviews were included if 2 authors reviewed for inclusion criteria and identified 2 or more databases for their search strategy. Reviews were excluded if they did not measure outcomes, if studies were protocols of reviews or incomplete, if technology was not the focus of the study, or if studies were included in the 2017 review. Studies of school site/classroom interventions, diabetes devices only, or solely feasibility, costs, and mortality were excluded. Full text review was completed on 111 studies. Each study was reviewed by 2 independent reviewers (DG, DI, JB, JD, ML, MP, VC) and conflicts were discussed to consensus. Fifty studies were retained.
Screening for Methodological Quality
Four reviewers in teams of 2 (DG and DI; JB and JD) assessed the 50 studies for methodological quality, including risk of bias, following
the AMSTAR-2 quality assessment guidelines. 16 This 16-question guideline was designed to identify critical flaws pertaining to study design and allow researchers to assign overall confidence. The research team identified 9 critical questions (# 1,2,3,4,5,6,8,9,16). Studies with one or more critical flaws were excluded. Studies with non-critical flaws but no critical flaws were included in the review and final analysis. Twenty-six reviews were retained. (See Supplemental Appendix A for critical questions and critical flaws)
Data Extraction and Analysis
Eight reviewers in teams of 2 (JB and JY; JD and AH; DG and DI; VC and ML) independently entered data into a data extraction table then met to confirm assessments. Tables included the following information: research question and components of PICO (population, intervention, comparator group, outcome); description of included studies (number, year, location, duration, participant characteristics, types of clinicians involved in interventions, technology and devices used); outcomes (technology usability, behavioral/psychosocial, ADCES7 Self-Care Behaviors™, physiological and glycemic including A1C); and TES features. Supplemental data were used where available to gather complete information.
Results
Phase 1
Building upon the 2017 TES feedback loop 1 and definitions applied in this review, we developed a preliminary taxonomy to be used when describing technology-enabled interventions. Similar to other taxonomies, 17 the Taxonomy of Technology-Enabled Self-Management Interventions (TES Taxonomy) was developed for 2 purposes: (1) to standardize the description of technology-enabled self-management interventions and (2) to ensure that future research uses the taxonomy to encourage replication, comparison, and evaluation. 18 Table 1 describes the attributes and the definitions of the TES Taxonomy.
Taxonomy of Technology-Enabled Self-Management Interventions (TES Taxonomy).
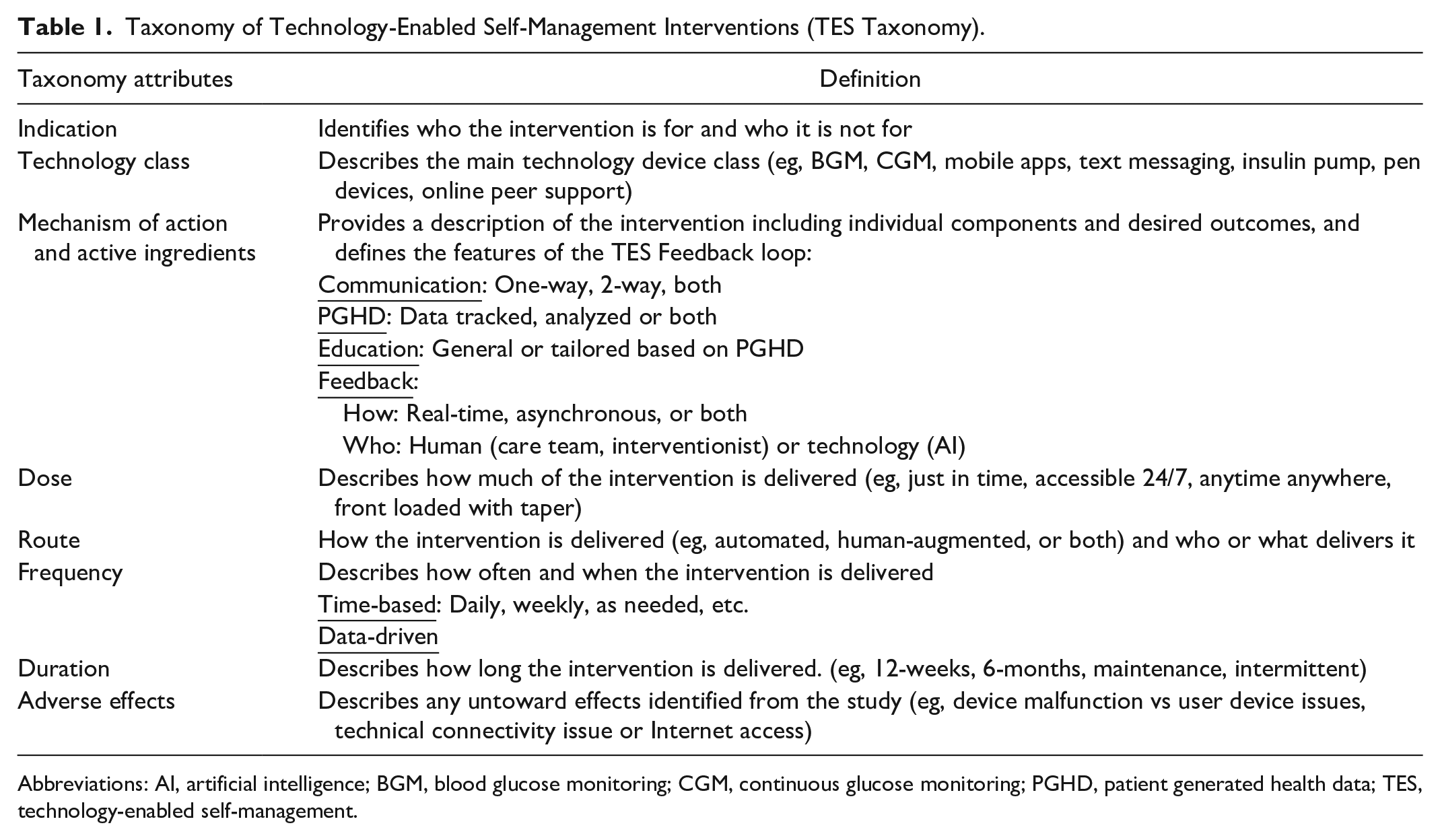
Abbreviations: AI, artificial intelligence; BGM, blood glucose monitoring; CGM, continuous glucose monitoring; PGHD, patient generated health data; TES, technology-enabled self-management.
Phase 2
Study Characteristics
Twenty-six reviews, published between 2017 and 2020, conducted in multiple countries, were included in this umbrella review,19-44 See Table 2. The number of studies in the selected reviews ranged from 7 to 111, and the number of participants ranged from 30 to 23,648. Participants included children through older adults with mean age range from 8-80 years. Three (11.5%) reviews did not report age ranges26,34,42, 2 (9.5%) reported only “adults.”27,29 Four (15.4%) included studies with only T1D,22,37,42,44 14 (53.8%) included studies with only T2D,19,20,23,24,27-29,31-33,38,39,42,44 and 12 (46.2%) reviews included both T1D and T2D.21,24-26,29,34-36,39,41-43 Studies utilized a variety of clinicians; 3 (11.5%) studies did not report type of clinician20,23,37, 5 (19.2%) studies stated only “healthcare professionals.”19,32,39,41,42
Study Characteristics (
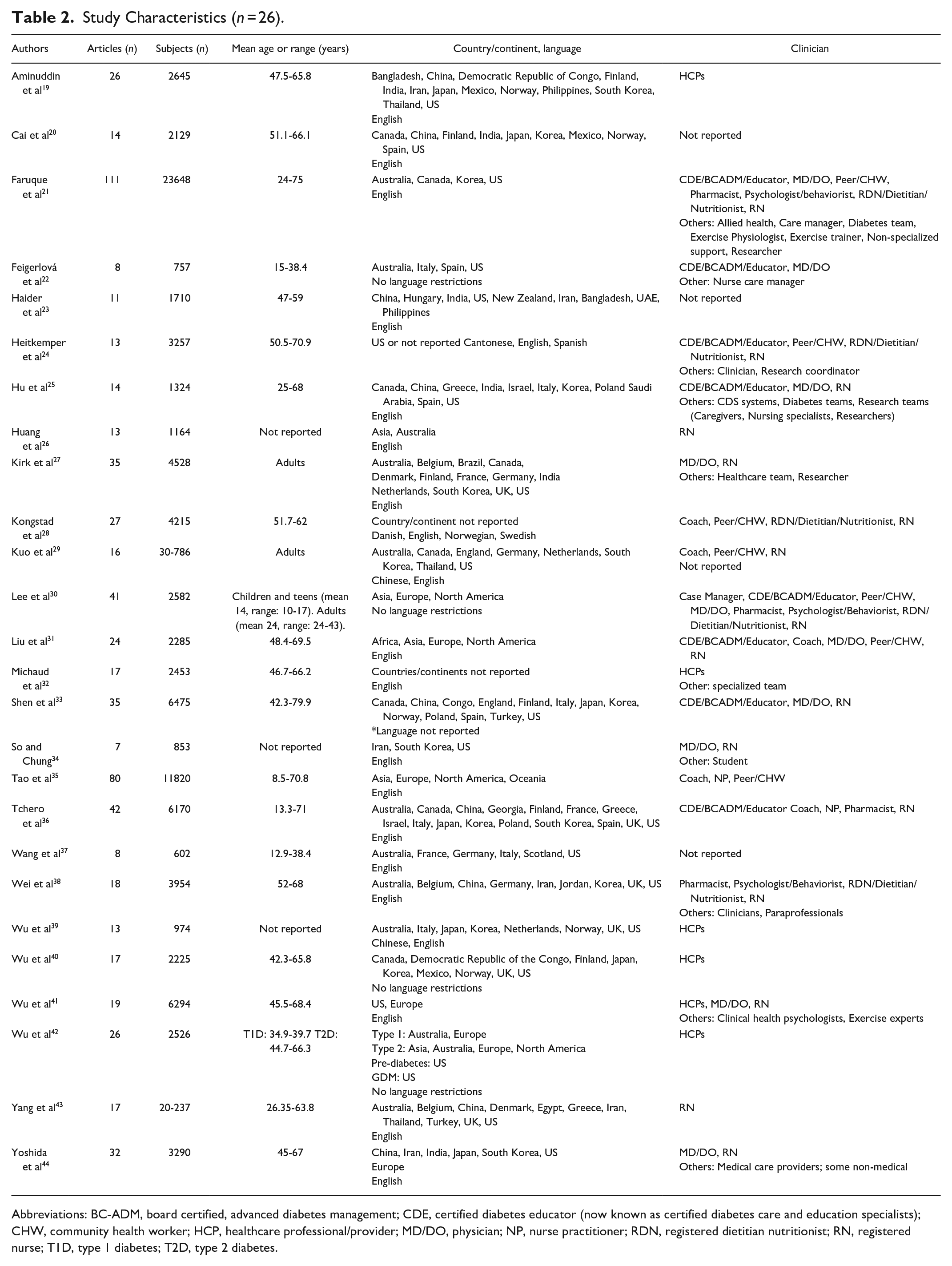
Abbreviations: BC-ADM, board certified, advanced diabetes management; CDE, certified diabetes educator (now known as certified diabetes care and education specialists); CHW, community health worker; HCP, healthcare professional/provider; MD/DO, physician; NP, nurse practitioner; RDN, registered dietitian nutritionist; RN, registered nurse; T1D, type 1 diabetes; T2D, type 2 diabetes.
Technology and Outcomes
Studies (
Types of Technology Included in the Interventions and Outcomes Measured (
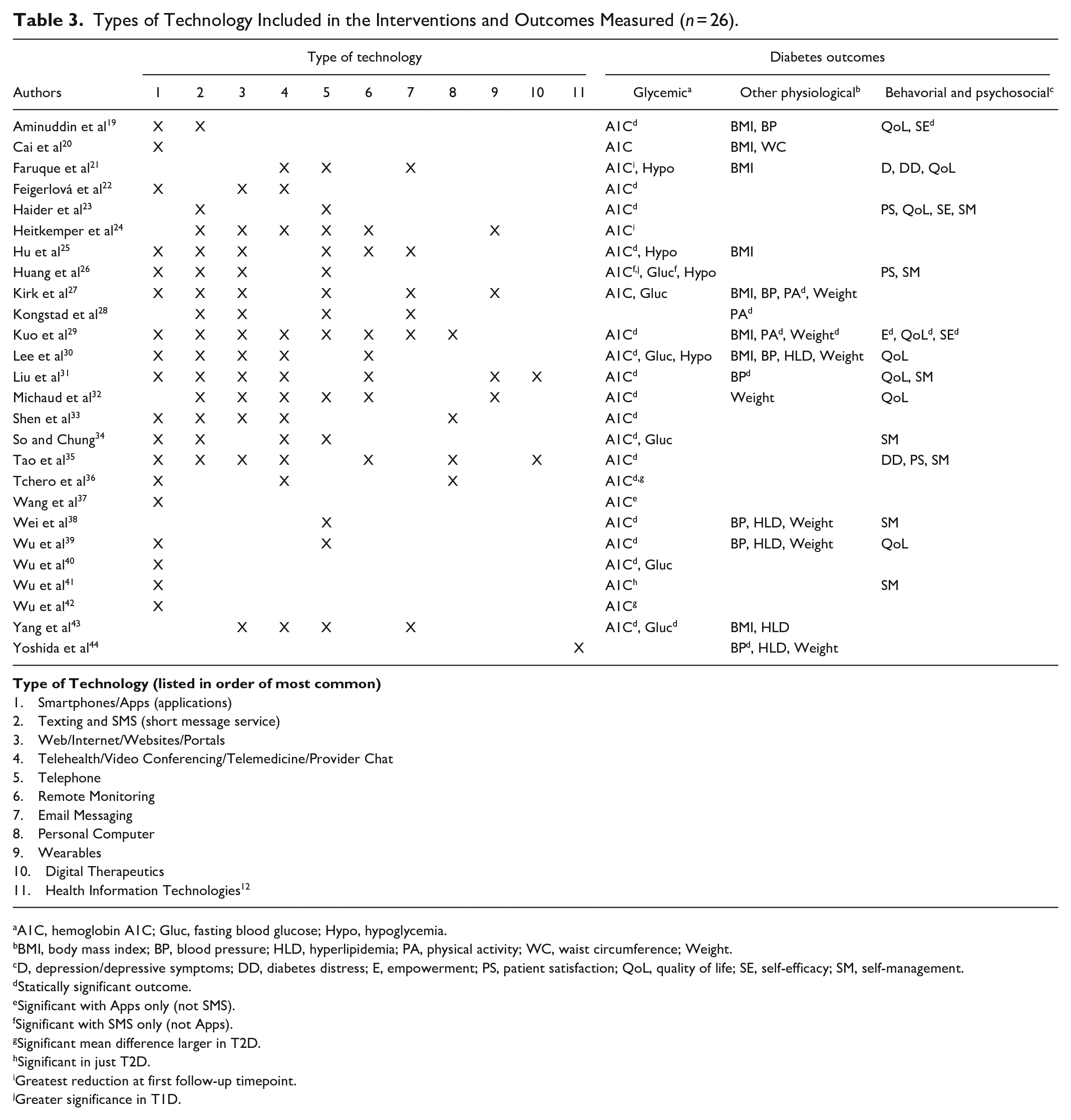
A1C, hemoglobin A1C; Gluc, fasting blood glucose; Hypo, hypoglycemia.
BMI, body mass index; BP, blood pressure; HLD, hyperlipidemia; PA, physical activity; WC, waist circumference; Weight.
D, depression/depressive symptoms; DD, diabetes distress; E, empowerment; PS, patient satisfaction; QoL, quality of life; SE, self-efficacy; SM, self-management.
Statically significant outcome.
Significant with Apps only (not SMS).
Significant with SMS only (not Apps).
Significant mean difference larger in T2D.
Significant in just T2D.
Greatest reduction at first follow-up timepoint.
Greater significance in T1D.
The studies explored diabetes self-management (glycemic, other physiological and behavioral/psychological) related outcomes (Table 3). For glycemic outcomes, most (
Interventions incorporating multiple modalities (eg, texting or telehealth in combination with web-based tools) more frequently had significant outcomes compared to single interventions. There is also strong evidence supporting A1C reduction with a variety of technologies (Table 3). Three studies identified that technology-enabled interventions were more effective in people with T2D,36,39,40 one study determined interventions were more effective in T1D; 26 however, no pattern emerged to identify technologies with the greatest reduction in A1C for specific populations. Two studies reported greater short-term A1C reductions at the first follow-up timepoint.21,24
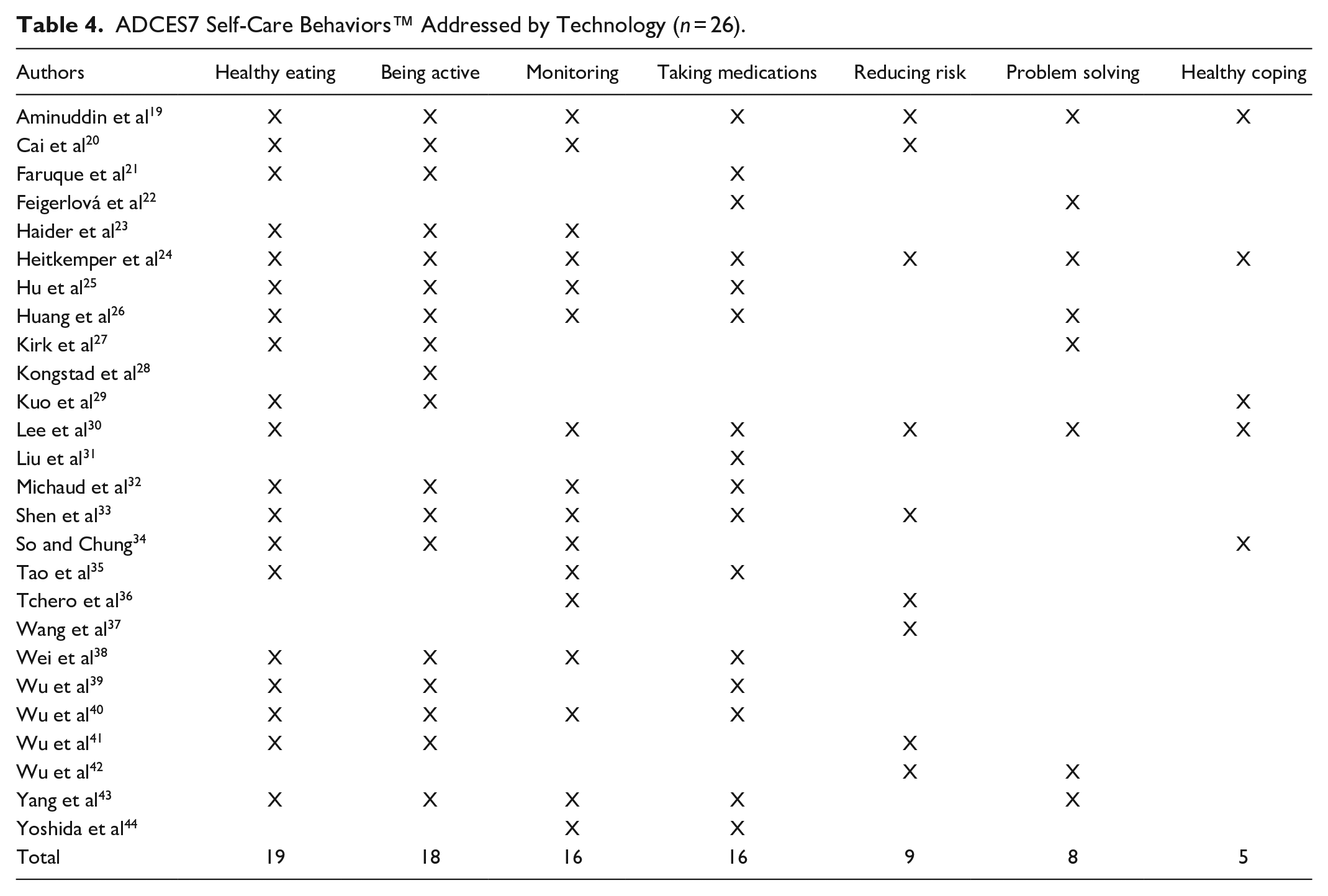
Table 4 identifies the ADCES7 self-care behaviors™ 5 addressed by the studies included in the reviews. Healthy eating was the most common self-care behavior addressed in the studies and being active ranked second highest. Only eight (38%) studies included problem–solving,19,22,24,26,27,30,42,43 which ranked second lowest, and 5 (23.8%) studies focused on healthy coping.19,24,29,30,34
ADCES7 Self-Care Behaviors™ Addressed by Technology (
Intervention Features
In this review we evaluated the 4 essential intervention features of the TES feedback loop: communication, patient generated health data (PGHD) analysis, education, and feedback. We used the TES feature definitions from Greenwood et al, 1 and expanded the definition of feedback into 2 sub-categories. To compare the results of this study with the 2017 research, the 21 studies that included a meta-analysis with significant mean difference in A1C between the intervention and control groups are displayed in Table 5. In the table, the authors are ordered by the mean difference in A1C from greatest to least.
Technology-Enabled Self-Management (TES) Feedback Loop Intervention Features of Studies With Meta-Analysis and Significant A1C Mean Difference Between Intervention and Control Group (
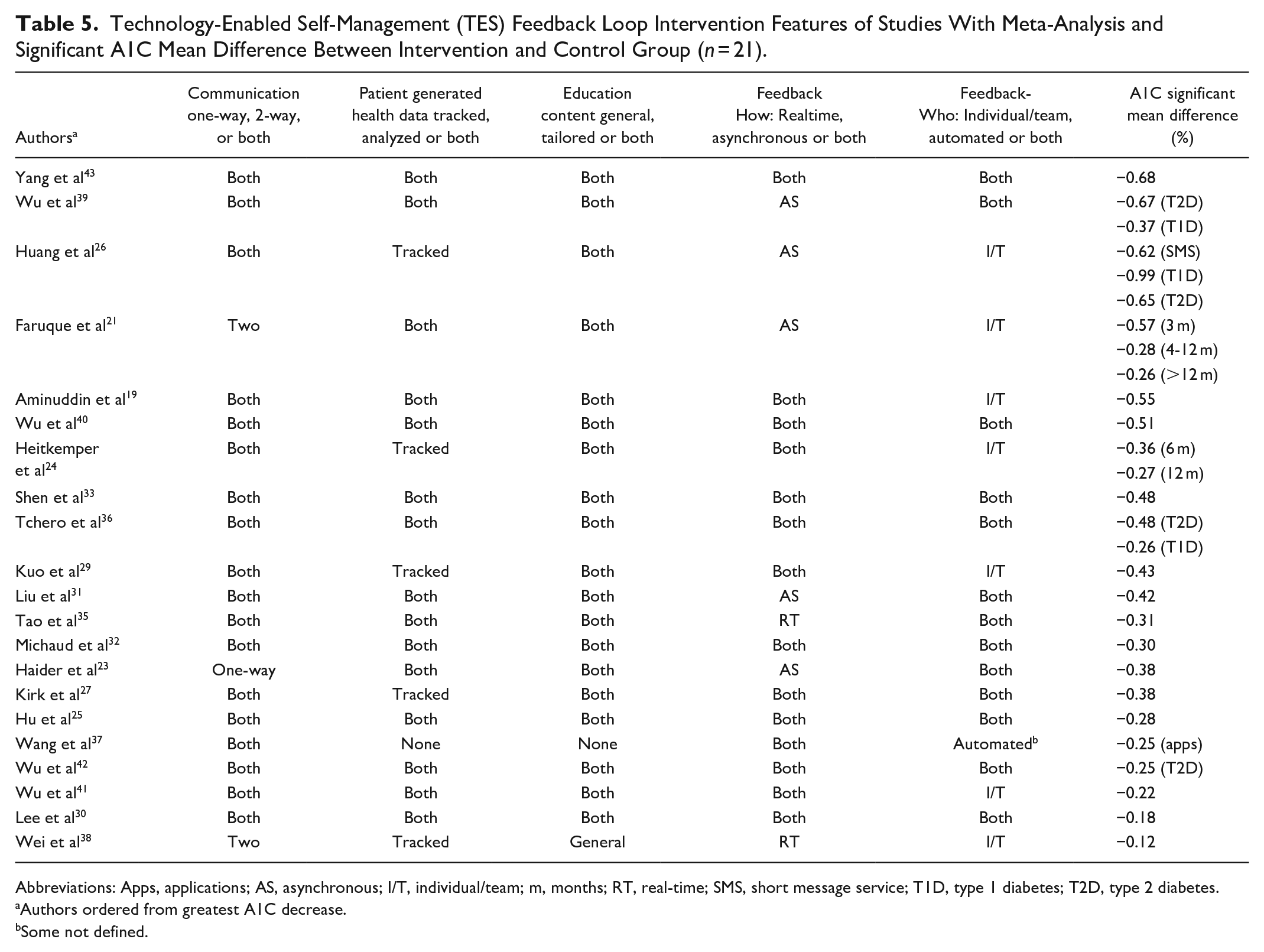
Abbreviations: Apps, applications; AS, asynchronous; I/T, individual/team; m, months; RT, real-time; SMS, short message service; T1D, type 1 diabetes; T2D, type 2 diabetes.
Authors ordered from greatest A1C decrease.
Some not defined.
Communication was defined as a one-way or 2-way exchange of information between the participant and care team. Twenty reviews described studies with 2-way communication, with one study including only one-directional texts. 23 while 18 (85.7%)19,20,22,24-37,39-44 of the studies included both one-way and 2-way communication. PGHD was defined as either tracked or tracked and analyzed (both). Fifteen (71.4%) of the reviews19,21,23,25,30-33,35,36,39-43 described PGHD as both and 5 (23.8%) reviews24,26,27,29,38 described as tracked only. One did not clarify how PGHD were used, but detailed that interventions included either texts or a diabetes management app. 37 Education content was defined as either general self-management education or tailored based on self-care behaviors and PGHD, with 19 of the reviews identifying both types of education (90.4%).19,21,23-27,29-33,35,36,39-43 General education was described in one 38 (4.7%) and not described in one 37 (4.7%). In this review, we expanded feedback into 2 sub-categories: how the feedback was provided, either real time or asynchronously, and who provided the feedback, an individual/diabetes team member or via an automated response. In this review, feedback to participants was reported as asynchronous by 5 (23.8%) reviews,21,23,26,31,39 both asynchronous and real-time by 1419,24,25,27,29,30,32,33,36,37,40-43 (66.6%) and only real-time by 235,38 (9.5%) reviews. The feedback was reported as delivered by an individual/diabetes care team in 7 (33%) reviews.19,21,24,26,29,38,41 One (4.7%) described only automated feedback 37 and 13 (61.9%) reviews23,25,27,30-33,35,36,39,40,42,43 described both individual/team and automated feedback.
Gaps for Technology-Enabled Self-Management
A key finding is that the systematic reviews and meta-analyses lacked overall intervention descriptions making it difficult to identify the mechanism of action of the intervention and which features impacted health outcomes. Supplemental Appendix B applies the TES Taxonomy to the 21 meta-analyses in Table 5. Five additional gaps were identified (Table 6). There was lack of evaluation of the impact of continuous glucose monitoring (CGM) or mobile health interventions, and minimal focus on healthy coping or evaluation of pyschosocial impact. In addition there was limited description of theoretical frameworks used to develop interventions. Finally, studies need to report on ethnicity/race and health disparities.45-50
Research Gaps Identified and Future Opportunities.
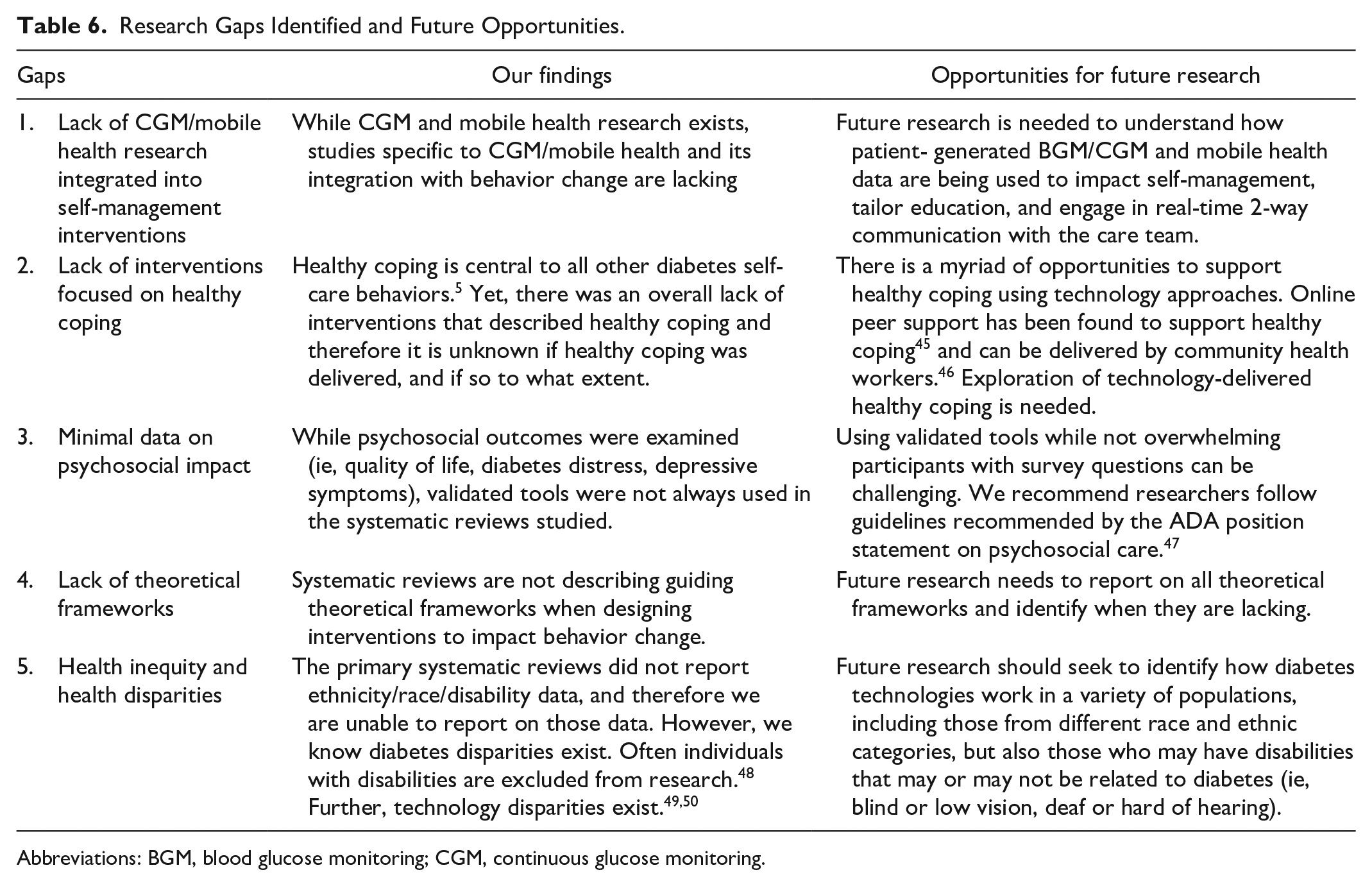
Abbreviations: BGM, blood glucose monitoring; CGM, continuous glucose monitoring.
Discussion
The intent of this umbrella review was to develop a taxonomy to describe intervention attributes in technology-enabled interventions and review high-quality systematic reviews and meta-analyses, published since the original 2017 review, to determine if the elements of the TES framework are being described in the interventions, and if the elements continue to result in achieving diabetes outcomes. Additionally, this umbrella review identified significant gaps in the literature regarding technology-enabled diabetes self-management. Our findings confirm technology-enabled self-management interventions with a TES feedback loop continue to be associated with a reduction in A1C.
The Taxonomy of Technology-Enabled Self-Management Interventions (TES Taxonomy)
Given the evolving models of care and the rapid influx of technology-enabled interventions, more emphasis needs to be placed on clear description of the design and process of implementing interventions. The lack of description of intervention components and how the TES features are operationalized makes it difficult to pinpoint types or features of technology-enabled interventions that impact glycemic, other physiological and behavioral/psychosocial diabetes self-management outcomes. Research, including systematic reviews, should fully describe intervention details, including participant characteristics; who the interventionist(s) were; what type of technology was used, including details on automation or tailored delivery; how often participants interacted with technology and/or the interventionist, including the duration of the intervention; while following a systematic process. A 2019 paper referenced key characteristics of digital interventions that need to be identified prior to recommending the intervention, specifically the “mechanism of action” that will impact outcomes. 51 Incorporating the TES Taxonomy can support recommendations of technology-enabled interventions by clinicians. Recent research describes similar challenges with digital health interventions, indicating a lack of consistency in reporting original research, which makes conducting systematic reviews more challenging.8,52 This lack of overall description could happen for several reasons, including publication word limitations or proprietary concerns. Scientists should be clear when reporting methodology so appropriate conclusions can be drawn.
Update to the 2017 Review
In this update, mobile and smartphone interventions were successful in lowering A1C with 2 studies reporting greater change in A1C in T2D compared to T1D.36,42 Similar to our original review, there was heterogeneity in the interventions, technologies, and methodologies. In addition to A1C being the most common clinical outcome reported, there is a lack of assessment and evaluation of psychosocial and self-management outcomes. When assessed, self-report and inadequately validated instruments were used, limiting the ability to draw comparisons and statistical conclusions.
Most reviews were meta-analyses focusing on A1C outcomes, not narratively summarizing interventions in detail. Compared to 2017, this update found that more reviews described “both” basic and advanced TES framework features, possibly reflecting the evolving technology available for interventions. Several studies in our update identified that interventions combining multiple technologies lowered A1C significantly which corresponds to the ADA 2021 SOC recommendation: “systems that combine technology and online coaching can be beneficial in treating prediabetes and diabetes for some individuals.” 10
Different from the 2017 review, we extracted the clinician’s specialty (eg, RN, MD, coach) to understand their role in implementing the technology intervention. However, clinicians were not always reported, and it was not clear which clinicians were conducting the intervention tasks. Overall, intervention details were minimal, making it challenging to understand the active ingredient of the technology. When we applied the TES Taxonomy to the 21 meta-analyses (Supplemental Appendix B), we were able to define a more complete picture of the interventions that significantly lowered A1C; however, there were missing data for several attributes. If original studies and reviews incorporated a taxonomy to describe interventions, data synthesis would be streamlined, and more clarity would exist.
Research Gaps Identified and Future Opportunities
Based on gaps identified, opportunities for future research are suggested (Table 6). Despite the rise in adoption of CGM and integrated apps, and a focus on time-in-range as a metric of diabetes management, current reviews do not include these elements. Mobile health and digital health interventions may be evaluated more often as real-world evidence and not in randomized controlled trials, thus not included in systematic review data.
Coping is the cornerstone for diabetes self-management;
5
however, healthy coping was not evaluated in these studies. In this update, 15% (
Technology intervention development and research should be guided by theoretical frameworks, yet they were rarely described. Frameworks typically address behavioral or therapeutic approaches, and they can also guide technology implementation. While the TES framework describes the necessary intervention features to achieve diabetes outcomes, it does not describe the process of implementing technology into a clinic, health system, or community setting. The ADA SOC 2021 indicate that “diabetes technology, when coupled with education and follow-up, can improve the lives and health of PwD; however, the complexity and rapid change of the diabetes technology landscape can also be a barrier to patient and provider implementation.” 10 People with diabetes need HCPs who are knowledgeable about diabetes technologies, especially as diabetes technology use is shifting from specialty to primary care.
Two recent publications provide guidance regarding technology optimization and integration.53,54 The Identify, Configure, Collaborate (ICC) Framework describes a 3-step, simplified, systematic approach to optimize technology-enabled diabetes care and education. This framework can streamline the process of technology implementation and use of PGHD. Additionally, the DATAA Model, presents a simple approach for HCPs and PwD to collaborate and evaluate ambulatory glucose profile data. 54 These publications can support researchers and clinicians in systematically describing their processes to facilitate evaluation and understanding of what works.
Health disparities exist with diabetes technology. Recent data from the T1D Exchange demonstrated that while use of technology has increased over the past 10 years, individuals with low socio-economic status (SES) and non-white racial/ethnic groups have lower technology use and higher A1C levels. 50 Smartphones can increase access to diabetes technologies, and yet diverse populations continue to be underrepresented.49,50 Research has shown that implicit provider bias limits the prescription of technology in lower SES and non-white populations. 49 Lack of trust in research teams, difficulty recruiting, and overt exclusion from studies is problematic. Future studies, when possible, could consider subgroup analysis of underrepresented groups.
Designing technology interventions to address disparities, disabilities, and non-English speaking individuals (inclusive design) could increase inclusion and equity. Developing options for one population (eg, flashing light alerts for Deaf populations) may provide derivative benefits for other populations.48-50 Diabetes technologies should be accessible, affordable, and available to all populations.
Conclusion
Technology-enabled diabetes self-management interventions continue to be associated with improved clinical outcomes. The rapid adoption and engagement with technology requires a focus on uniform measures for psychosocial outcomes, including healthy coping. Using the TES Taxonomy as a standard approach to describe technology-enabled interventions will support further understanding of the impact that technology has on diabetes outcomes and provide a systematic framework for ADCES to frame strategic conversations for technology and practice.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968211036430 – Supplemental material for A New Taxonomy for Technology-Enabled Diabetes Self-Management Interventions: Results of an Umbrella Review
Supplemental material, sj-pdf-1-dst-10.1177_19322968211036430 for A New Taxonomy for Technology-Enabled Diabetes Self-Management Interventions: Results of an Umbrella Review by Deborah A. Greenwood, Michelle L. Litchman, Diana Isaacs, Julia E. Blanchette, Jane K. Dickinson, Allyson Hughes, Vanessa D. Colicchio, Jiancheng Ye, Kirsten Yehl, Andrew Todd and Malinda M. Peeples in Journal of Diabetes Science and Technology
Supplemental Material
sj-pdf-2-dst-10.1177_19322968211036430 – Supplemental material for A New Taxonomy for Technology-Enabled Diabetes Self-Management Interventions: Results of an Umbrella Review
Supplemental material, sj-pdf-2-dst-10.1177_19322968211036430 for A New Taxonomy for Technology-Enabled Diabetes Self-Management Interventions: Results of an Umbrella Review by Deborah A. Greenwood, Michelle L. Litchman, Diana Isaacs, Julia E. Blanchette, Jane K. Dickinson, Allyson Hughes, Vanessa D. Colicchio, Jiancheng Ye, Kirsten Yehl, Andrew Todd and Malinda M. Peeples in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to acknowledge the Association of Diabetes Care & Education Specialists for their support in completing this publication.
Abbreviations
A1c, hemoglobin A1c; AI, artificial intelligence; API, application programing interface; AS, asynchronous; BGM, blood glucose monitoring; BMI, body mass index; BP, blood pressure; CGM, continuous glucose monitoring; GCM, gestational diabetes; D, depression/depressive symptoms; DD, diabetes distress; E, empowerment; Gluc, fasting blood glucose; HCP, healthcare professional; HLD, hyperlipidemia; Hypo, hypoglycemia; I/T, individual/team; M, months; MDI, multiple daily injections; MDIS, mobile diabetes intervention study; PA, physical activity; PGHD, patient generated health data; PS, patient satisfaction; QoL, quality of life; REACH, rapid education/encouragement and communications for health; RCT, randomized clinical trial; RT, real time; SE, self-efficacy; SM, self-management; TES, technology-enabled self-management; T1D, type 1 diabetes; T2D, type 2 diabetes; WC, waist circumference
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Deborah Greenwood is an employee of Dexcom Corporation and faculty for Lifescan Diabetes Institute, consultant for Lifescan, and Silverfern; Digital health advisory board, Novo Nordisk.
Michelle L. Litchman was the PI of an investigator-initiated trial funded by Abbott Diabetes Care unrelated to this study.
Julia E. Blanchette is a consultant for WellDoc, Inc; a consultant/independent contractor for Insulet Corporation and Tandem Diabetes; Advisory Board for Cardinal Health and Provention Bio; Research Support from the Association of Diabetes Care and Education Specialists and the Certification Board for Diabetes Care and Education unrelated to this study.
Jane K. Dickinson has no relevant disclosures.
Allyson S. Hughes has no relevant disclosures.
Jiancheng Ye has no relevant disclosures.
Kirsten Yehl is on staff at the Association of Diabetes Care & Education Specialists.
Andrew Todd has no relevant disclosures
Malinda Peeples is an employee of Welldoc, Inc.
Diana Isaacs serves on the speaker’s bureau for Dexcom, Abbott, Medtronic, Novo Nordisk and a consultant for Lifescan. She has served on Advisory Boards for Medtronic, Lilly, and Prevention Bio.
Vanessa D. Colicchio is a full-time PhD candidate at the University of Utah College of Nursing and has no relevant disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Association of Diabetes Care & Education Specialists
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
