Abstract

Type 1 diabetes is immune-mediated diabetes requiring lifelong insulin therapy for optimal glycemic control. Insulin degludec is a newer ultra-long-acting insulin. 1 The insulin degludec has been shown improve fasting glucose control and associated with reduced nocturnal and overall hypoglycemia compared to insulin glargine.2-4 Flexibility in taking insulin and a lower rate of hypoglycemia makes insulin degludec an ideal choice of insulin for patients with type 1 diabetes.
We report four patients with type 1 diabetes without any previous history of severe hypoglycemia had severe hypoglycemia that required hospitalization after switching to insulin degludec based on FDA approved label dose conversion recommendations. The patients’ characteristics, insulin glargine dose, conversion dose to insulin degludec and A1c are shown in Table 1. All four patients were nonadherent to insulin regimen. They had a history of missing either insulin glargine and/or mealtime insulin at least once a week or more frequently and testing their blood glucose once or twice a day; less than recommended glucose testing frequency. Due to the history of missing basal insulins and poor glycemic control, all four patients were switched to insulin degludec (about 1:1 dose conversion as per approved label) to improve flexibility in taking basal insulin any time of the day. All four patients had a severe hypoglycemia within 2-10 weeks of starting insulin degludec that required hospitalization (Table 1). Two reported seizure and two had loss of consciousness. One patient had three episodes of seizures due to severe hypoglycemia. In all four patients, severe hypoglycemia occurred at night or in the early morning.
Characteristics of the Patients Who Had Severe Hypoglycemia After Insulin Degludec Initiation.
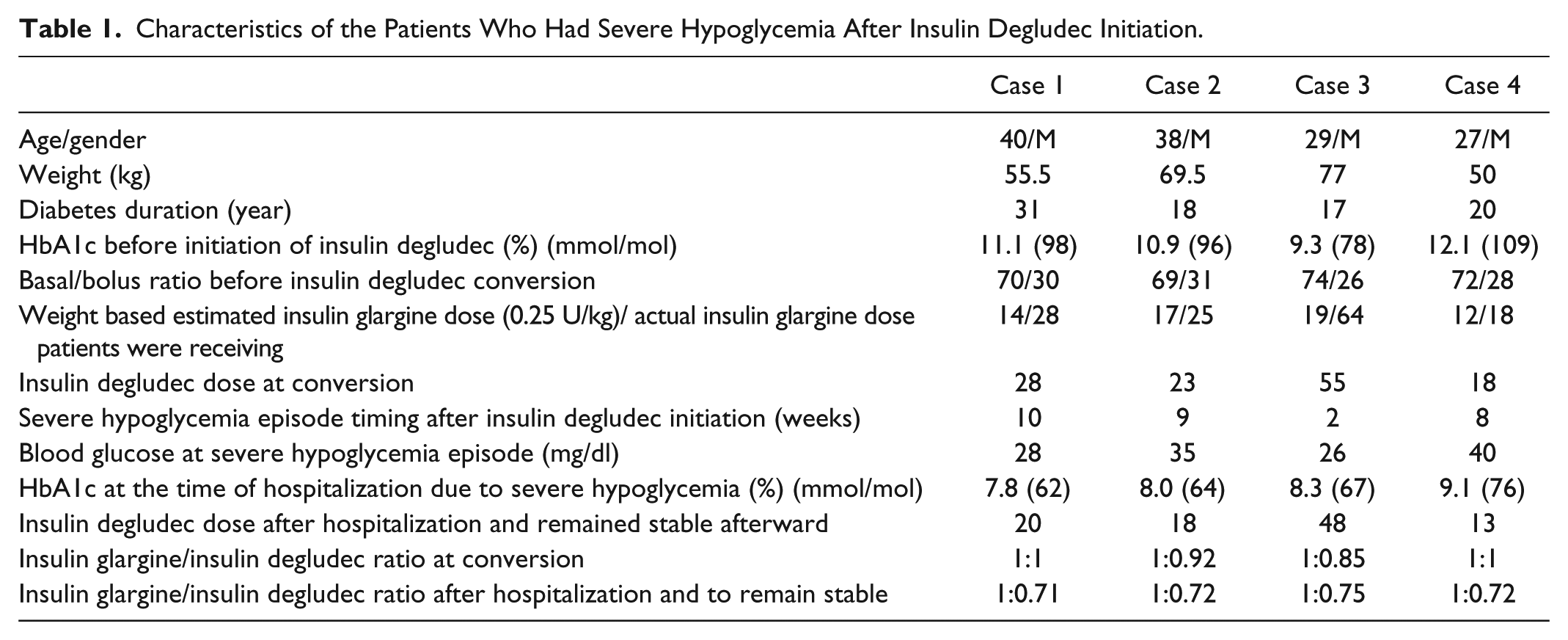
Missing insulin injections is an underrecognized problem among patients with type 1 diabetes. According to Type 1 Diabetes Exchange Registry, nearly 15% of patients with type 1 diabetes are missing insulin at least once a week or more frequently and missing insulin injections was associated with a higher A1c. 5 Many providers would increase the dose of insulin among these patients, and thus the total amount of long-acting insulin will be higher than the actual dose needed to have an optimal control. In all four patients, weight-based basal insulin dose calculations were 50% lower than the actual basal insulin dose they were receiving (Table 1). Insulin degludec has been approved for flexible dosing, 1 changing 1:1 from insulin glargine among these patients with type 1 diabetes results in higher insulin dose than required. This resulted in severe hypoglycemia among these patients. Randomized controlled trials with insulin degludec excluded type 1 diabetes patients who were not compliant with insulin regimen and had A1c greater than 10% 2-4. Therefore, the approved label 1:1 conversion dose may not be appropriate for type 1 diabetes patients with A1c greater than 10%. We recommend using up to 30% less insulin degludec or calculating the dose based on weight (0.4-0.5 units/kg of body weight and 50% of total insulin dose should be starting basal insulin dose) for patients with type 1 diabetes who are not compliant with insulin injections and have a high A1c.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VNS served as Advisory Board member for Sanofi and as a consultant for Dexcom. VNS’ employer had received research fundings from Type 1 Diabetes Exchange, Sanofi US and Dexcom Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
