Abstract
Background:
The physiologic delay in glucose diffusion from the blood to the interstitial fluid and instrumental factors contribute to the delay between changes in plasma glucose (PG) and measurements made by continuous glucose monitors (CGMs). This study compared the duration of this delay for three CGMs.
Methods:
A total of 24 healthy adolescent and adult subjects with type 1 diabetes wore three CGM devices simultaneously for 48 hours: Dexcom G4 Platinum, Abbott Navigator, and Medtronic Enlite. The time delay between PG and CGM-estimated plasma glucose (CGMG) was estimated by comparing time-shifted CGMG with reference PG taken every 15 minutes.
Results:
The delay estimated by our approach was larger for the Navigator than for the G4 Platinum in adolescents (7.7 ± 1.1 versus 5.6 ± 0.9 min,
Conclusions:
There are differences in the estimated PG to CGMG time delays between CGM devices in the same subjects. The delay between PG and CGMG is smaller for adolescents than for adults. The PG–to–CGMG time delay is influenced by both instrument and host factors.
Most continuous glucose monitors (CGM) devices on the market use an electrochemical sensor inserted subcutaneously so that it accesses the interstitial fluid (ISF) space to measure glucose concentrations. 1 The CGM-estimated plasma glucose (CGMG) is an estimate of the plasma glucose (PG) made using measurements in the interstitial fluid that are calibrated using PG. Physiologic time lag or equilibration time has been defined as the time required for the movement of glucose from the vascular to the ISF space, and has been estimated to be 5-6 minutes in healthy adults and 7-8 minutes in fasting patients with type 1 diabetes (T1D) by direct measurement using a microdialysis catheter in the abdominal subcutaneous space.2,3 As a result of this delay, and the corresponding PG to CGMG time delay, calibrations performed during periods when glucose is changing rapidly can lead to persistent inaccuracy in CGMG measurements. Instrumental factors can also create delay that is added to the physiologic lag. It has been argued that delay caused by the physiologic component is minimal compared to the instrumental component. 4 Both physiologic lag and sensor lag have the potential to adversely affect CGM accuracy, particularly during periods of rapidly changing glucose levels. 5 It has been suggested that the effect of these delays on sensor accuracy is not substantial when compared to other sources of error, 6 but this may depend on the CGM device used. In devices with a large component of random error it might be difficult to detect the effect of lag on accuracy separate from these random errors.
We have estimated and compared the PG to CGMG time delays corresponding to three different CGM devices, the Dexcom G4 Platinum, Medtronic Enlite, and Abbott Navigator, worn simultaneously by adult and adolescent subjects with type 1 diabetes.
Methods
Experimental Protocol
The clinical protocol was approved by the Massachusetts General Hospital and Boston University Human Research Committees, and was previously described in detail.7,8 During a study of automated glycemic regulation with a bionic pancreas, twenty-four healthy volunteers (12 adolescents, 12 adults) with type 1 diabetes each wore three CGM devices simultaneously for 48 hours. The CGM devices used in this study were the Abbot Navigator (Navigator), Dexcom G4 Platinum (G4 Platinum), and Medtronic Enlite (Enlite) (the sensor used in the 530G with Enlite sensor-augmented pump). Approximately 24 hours before the start of the study, the CGM sensors were inserted into the skin of the subject’s abdomen and linked to their respective receivers. At the start of the experiment an intravenous (IV) catheter was placed to draw blood for reference PG sampling every 15 minutes with the GlucoScout (International Biomedical), a point of care device with laboratory-grade accuracy. 9 Approximately 24 hours after insertion the first calibrations were performed according to the manufacturers’ instructions, except that venous PG measurements were used instead of point-of-care fingerstick measurements.
Additional calibrations were timed according to the manufacturers’ instructions. If the CGMG did not meet the International Organization for Standardization (ISO) 5197: 2003 numerical criteria for accuracy (>95% within 15 mg/dl of reference <75 mg/dl or within 20% of reference ≥75 mg/dl) compared to PG values obtained before breakfast and dinner, then an extra forced calibration was performed.
PG to CGMG Delay Estimation
CGMG values were reported by the devices at 5-minute intervals and were linearly interpolated between the reported measurements to reflect the estimated values at 1-minute intervals. These interpolated arrays (one for each subject) were then offset in 1-minute increments relative to the reference PG values, the offset ranging from 0 to 30 minutes. Each PG value was compared with the most closely temporally associated value from the interpolated CGM value array. The point accuracy of each CGM in each subject for each offset was measured in terms of the mean absolute relative difference (MARD), |
For each subject and CGM device the mean differences between the absolute relative differences (ARDs) at each time offset versus the reference ARD at zero offset was calculated [mean ΔARD = (ARD offsetx) – (ARD offset0)]. The mean ΔARDs for each subject were then averaged in the adults and adolescent groups separately. The offset time at which the mean ΔARD in these populations of subjects reached a minimum provided an estimate of the time delay for each CGM in that population.
The relationship between the PG and the CGMG is more complicated than can be fully understood by time-shifting the CGMG forward with respect to PG and identifying the location of minimum differences between them. Indeed, CGMG is a distorted version of PG and is not expected to coincide with PG by a mere temportal shift. 10 However, the simple methodology used here does allow us to estimate the effective delay between PG to CGMG, and to note differences in the apparent delay between different CGM devices systems in the same subject population and between different subject groups using those same CGM devices.
The Wilcoxon signed rank test was used to compare the estimated delay for the three CGM devices in both the adolescent and adult populations, and the Wilcoxon rank sum test was used to compare differences between estimated delays for each CGM device in adolescents versus adults.
A bootstrapping technique was used to estimate the degree of uncertainty of the estimated time delay using the array containing each subject’s mean ΔARD at each offset for each CGM. We used 100% sampling with full replacement to generate 1000 bootstrap samples from the mean ΔARD arrays to produce 1000 estimates of the offset that was optimal in order to minimize the ΔARD in both populations. A mean optimal offset (an estimate of the delay) and standard deviation were calculated for each CGM device in each population. Linear regression analysis was performed to determine if there was a relationship between subject age and estimated delay in the combined population of both adults and adolescents.
Results
Subjects
The mean age of the adult subjects was 45 ± 14 (26-66) years with a mean duration of diabetes of 27 ± 15 (7-54) years and a mean body mass index of 26 ± 3 (22-32) kg/m2. 8 The mean age of the adolescents was 15 ± 2 (12-18) with a mean diabetes duration of 7 ± 4 (2-13) years and a mean body mass index of 22 ± 3 (18-28) kg/m2. 8
Estimated Delay
For each of the three CGM devices in both populations there was a minimum in mean ΔARD at some time shift relative to time 0, demonstrating that there was an apparent delay in CGMG relative to PG (Figure 1, Supplemental Figure S1). We defined the apparent delay as the time shift that led to the minimum ΔARD, and this apparent delay differed between the three CGMs and in the two age groups (Figure 1, Table 1). The apparent delay was larger for the Navigator than for the G4 Platinum in both the adolescent (7.7 ± 1.1 versus 5.6 ± 0.9,
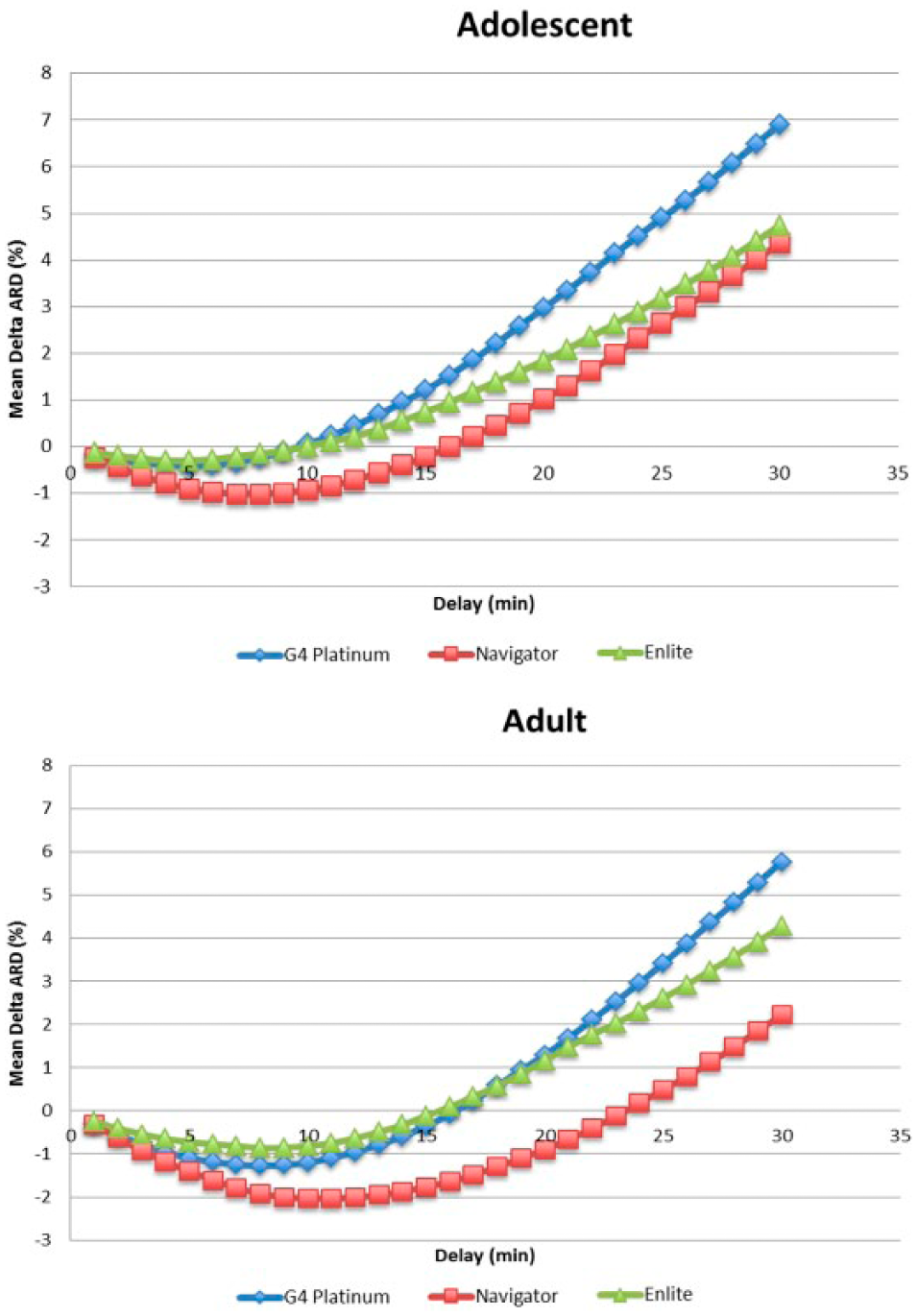
Effect of time shift on mean delta ARD (ΔARD). The degree of time shift that led to a minimum mean ΔARD differed between the three CGMs and in the two age groups. For the adolescent group, the maximum ΔARD was –0.6% at 6 minutes for the G4 Platinum, –1.0% at 8 minutes for the Navigator, and –0.2% at 4 minutes for the Enlite. For the adult group, the maximum ΔARD was –1.3% at 8 minutes for the G4 Platinum, –2% at 11 minutes for the Navigator, and –0.8% at 8 minutes for the Enlite.
Estimated delay by sensor for adolescents and adults.

Differences in Delay Between Subjects
There was a large variation in the estimated delay between experiments for the same CGM device, with apparent delays varying from a minimum of 0 minutes for two Enlite sensors to a maximum of 16 minutes for one Navigator sensor (Supplementary Figures 2-7). There was no significant correlation between the delay for one sensor and that of another sensor in the same subject (Supplementary Figures 8-13,
Age Versus Delay
Analysis of all 24 subjects’ ages versus their estimated delay showed a significant correlation between longer apparent delay and increasing age for the G4 Platinum and Navigator (
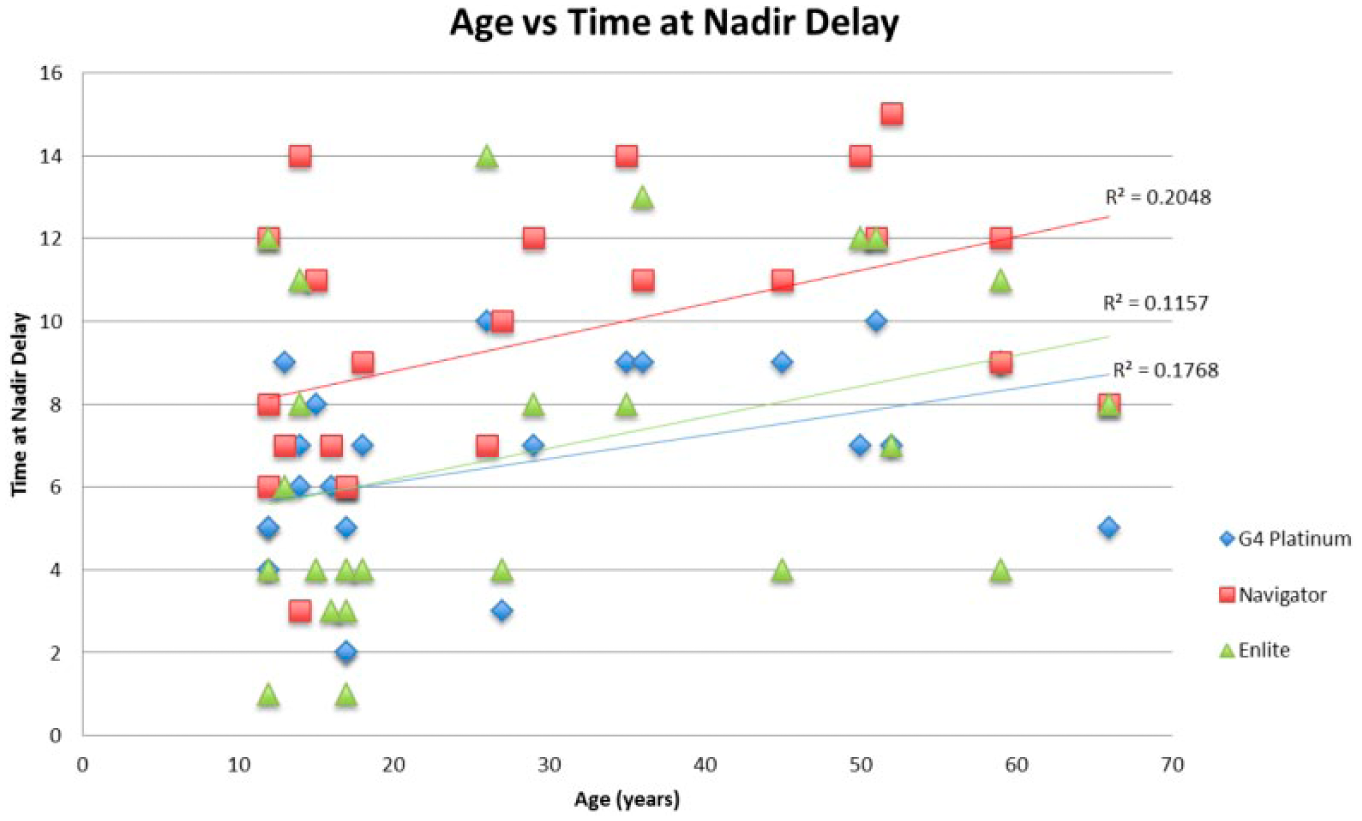
Age vs time shift at nadir delay. Delay correction was most beneficial with increasing age for the G4 Platinum and Navigator. The
Discussion
This study adds to the previous literature on the delay between blood glucose and CGM-estimated plasma glucose by evaluating the performance of three CGM devices when simultaneously worn by the same subjects. Although we used a very simple method of estimating the delay between changes in PG and the CGMG, the design of our trial in which each subject wore all three CGM devices allowed us to separate out the component of the apparent delays that are due to differences in the CGM systems. Our study included two currently available CGM devices for which delay data have not previously been reported, namely the G4 Platinum and the Enlite. An additional novel feature of our study is the analysis of adolescent versus adult populations.
We found that the estimated delay between PG and CGMG was larger for the Navigator than for the G4 Platinum. There was a similar nominal difference between the Navigator and the Enlite that did not reach statistical significance. This is consistent with instrumental factors, in addition to physiological factors, playing a significant role in producing the measured delay. 4 We have reported previously that the G4 Platinum and the Navigator were more accurate than the Enlite. 9 This is true despite the significantly longer delay for the Navigator relative to the G4 Platinum and a nominally longer delay for the Navigator relative to the Enlite. The maximal reduction in MARD that could be achieved by time-shifting the Enlite data relative to PG was 0.2% in the adolescent population and 0.8% in the adult population, which is much less than the difference in MARD between the Enlite and the more accurate G4 Platinum and Navigator CGM devices, 9 demonstrating that factors other than delay have a larger effect on accuracy, consistent with previous reports. 6
A surprising, and to our knowledge novel, finding in this study was that the adolescent subjects had shorter estimated delays as a group relative to the adults, for all three CGM devices evaluated. The delay correlated positively with increasing age when all subjects were included in the analysis. We previously found that accuracy was better in adolescents than in the adults for both the Navigator and the G4 Platinum, although not for the Enlite. 9 This suggests that aging of the skin may have a negative impact on equilibration of glucose between the blood and the interstitial fluid and/or the ability of the CGM sensor to accurately measure glucose in the interstitial fluid. However, our sample is not large enough to perform multivariable analysis to isolate the contribution of age relative to other factors that may influence delay or CGM accuracy. Finally, we found that different CGM sensors did not behave more similarly in individual subjects than they did in the age groups as a whole. This suggests that the observed delays were more closely related to general characteristics of the sensors themselves, differences between individual sensors of the same type, local effects at the site of insertion, and/or differences in the physiology shared within the age groups, rather than differences between individual subjects within the age groups that affected the delay of all CGM devices similarly.
Although the delay has been shown to have an effect on CGM accuracy, previous studies have disagreed regarding the magnitude of the delay present and its effect on accuracy. Zisser et al performed a post hoc analysis of the Dexcom Seven System (a previous generation of the Dexcom CGM) versus venous blood glucose. 11 They found the time delay (lag) of the CGM to be 5.7 minutes and concluded that the effect of time delay on sensor accuracy was not substantial. Garg et al compared two CGM systems concurrently, the Dexcom Seven System and the Abbot Navigator, and found a time delay (lag) of 4.5 ± 5 minutes for the Dexcom Seven and 15 ± 7 minutes for the Navigator, 4 consistent with the longer delay for the Navigator versus the newer G4 Platinum in this study. Leelarathna et al found the time delay of the Navigator to be 12 minutes, and Kovatchev et al found the Navigator delay to be 12.4 and 12.6 minutes in the two simultaneously placed sensors.12,13 There have been no previous studies of delay for the G4 Platinum and the Enlite CGM devices.
Possible sources of differences between reference PG values and CGM values include (1) errors in calibration of the CGM; (2) errors in CGM measurement of glucose in the interstitial fluid independent of calibration error; (3) errors in measurement of reference PG; and (4) delay between changes in glucose level in the blood and interstitial fluid environments. Differences in sensor chemistry between CGM devices, and in the algorithms that convert the voltage signal from the CGM sensor to an estimate of blood glucose, may contribute to the delay between changes in PG and CGMG . This functional delay will be in addition to any true physiologic delay of glucose equilibration between these two compartments. Our finding that the sensor with the longest delay (Navigator) was more accurate than a sensor with shorter delay (Enlite) suggests that factors other than instrumental delay have more quantitatively important effects on CGM accuracy. Furthermore, it appears that the Navigator and the G4 Platinum are achieving comparable accuracies with different strategies given the significantly longer delay of the Navigator than the G4 Platinum.
Our observations that delays were significantly less for all sensors in the adolescents relative to the adult population, and that the delays correlated positively with subject age are intriguing. We previously showed that the G4 Platinum and the Navigator had a higher point accuracy (lower MARD) in adolescents than in adults. 9 More studies are needed to validate this observation, but this data could be consistent with reduced microcirculation in the subcutaneous tissue with age, leading to slower equilibration of glucose between blood and interstitial fluid. However, the differences in accuracy between adults and adolescents are larger than can be explained solely by differences in the delay. 9
The clinical relevance of time delay between PG and CGMG is not limited to effects on point accuracy. The impact of the delay on decision making can be particularly important at times when the trajectory of PG changes quickly and when the PG is near the hypoglycemic threshold. As an example, consider the use of CGM as the input to an automated glucose control system (artificial or bionic pancreas). Such systems are capable of more tightly controlling PG than is typical for patients with type 1 diabetes. As a consequence, the PG is much more often stable in the target range, and is therefore near the hypoglycemic threshold more often. If something causes the PG to begin to fall rapidly, as can occur during even mild exercise such as walking, the proximity to the hypoglycemic threshold provides very limited time to respond. Any delay between the change in the trajectory of PG and that change being reflected in the CGMG trajectory will reduce the time available for the automated system to take an action, such as delivery of glucagon, to prevent a hypoglycemic event.
A limitation in this study is that it did not use the most recent CGM from Dexcom, the G5, and the most recent CGM from Abbott, the Freestyle Libre. In both cases the underlying sensor technology is very similar to the sensors we studied, with the modifications being limited to the calibration algorithm (or lack of calibration in the case of the Libre), the signal processing, and the design of the transmitter. It is unclear to what extent these factors would have affected the measured delay. Another limitation is that the youngest and oldest subjects were respectively 12 and 66 years of age; thus, we do not know if the correlation between age and delay is present in subjects younger and older than the participants in this study. In addition, the subjects had limited activity in an inpatient setting during the study, being sedentary except for a period of structured exercise lasting approximately 30 minutes 8 and the ambient temperature was controlled. More activity or variability in ambient temperature, as is found in the outpatient setting, could have affected the delay observed. Finally, we used the simple method of time-shifting to estimate the delay between changes in PG and changes in CGMG; thus, the method provided an estimate for the combined differences in the timing of changes in PG and the interstitial fluid as well as instrumental delays in responding to these changes. Finally, we did not use more sophisticated modeling techniques to estimate the time constants for equilibration of glucose from the plasma to the interstitial fluid compartments.10,14,15
Conclusion
There are differences in the delay between PG and CGMG among different CGM devices worn by the same subjects. The delay between PG and CGMG appears to be smaller for adolescents than for adults and appears to increase with age. The time delay between PG and CGMG is influenced by both instrument and host factors. Factors other than instrumental time delay have more quantitatively important effects on CGM accuracy.
Footnotes
Acknowledgements
We thank all our volunteers for their time and dedication, the study staff at the Massachusetts General Diabetes Research Center and Boston University, and the members of the Partners Human Research Committee and Boston University Medical Campus Institutional Review Board for their oversight of the study.
Abbreviations
ARD, absolute relative difference; BG, blood glucose; CGM, continuous glucose monitoring; CGMG, CGM-estimated plasma glucose; GS, GlucoScout; ISO, International Organization for Standardization; IV, intravenous; MARD, mean ARD; PG, plasma glucose; YSI, Yellow Springs Instrument.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJR has received loaned equipment and technical assistance from International Biomedical, Abbott Diabetes Care, Medtronic, Dexcom, Sweet Spot Diabetes, Insulet, and Hospira. FHEK holds a patent related to a fully automated control system for type 1 diabetes (US 7,806,854) and pending patent applications related to a blood glucose control system (PCT/US 11/058,688 and PCT/US 13/870,634), all assigned to Boston University. SJR received consulting fees from Medtronic (through Diabetes Technology Management), lecture fees from Tandem Diabetes, Sanofi Aventis, Eli Lilly, Abbott Diabetes Care, and Biodel, serves on the scientific advisory boards for Tandem Diabetes and Companion Medical, and holds a pending patent application for a blood glucose control system (PCT/US 13/870,634), assigned to Partners HealthCare and Massachusetts General Hospital. The other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Grant 2012PG-T1D020 from the Helmsley Charitable Trust (to SJR).
Supplementary Material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
