Abstract
Gestational diabetes (GDM) burden has been increasing progressively over the past years. Knowing that intrauterine exposure to maternal diabetes confers high risk for macrosomia as well as for future type 2 diabetes and obesity of the offspring, health care organizations try to provide effective control in spite of the limited resources. Artificial-intelligence-augmented telemedicine has been proposed as a helpful tool to facilitate an efficient widespread medical assistance to GDM. The aim of the study we present was to test the feasibility and acceptance of a mobile decision-support system for GDM, developed in the seventh framework program MobiGuide Project, which includes computer-interpretable clinical practice guidelines, access to data from the electronic health record as well as from glucose, blood pressure, and activity sensors. The results of this pilot study with 20 patients showed that the system is feasible. Compliance of patients with blood glucose monitoring was higher than that observed in a historical group of 247 patients, similar in clinical characteristics, who had been followed up for the 3 years prior to the pilot study. A questionnaire on the use of the telemedicine system showed a high degree of acceptance.
Gestational diabetes (GDM) is classically defined as carbohydrate intolerance that begins or is first recognized during pregnancy but in recent years the preferred definition has been modified trying to distinguish women with preexisting diabetes. 1 GDM occurs when beta cell function is not sufficient to overcome the insulin resistance produced by changes in diabetogenic hormones during pregnancy. Several adverse outcomes have been associated with GDM, including fetal macrosomia, birth trauma and perinatal complications. Importantly, the risk of these outcomes increases continuously as maternal fasting plasma glucose levels increases. 2 In 2010, the International Association of Diabetes and Pregnancy Study Group (IADPSG), proposed criteria for GDM based on the results of the 75-g glucose tolerance test. 3 The thresholds represent the glucose values at which the odds of infant birth weight, cord C-peptide, and percentage body fat >90 percentile were 1.75 times the estimated odds of these outcomes. The implementation of IADPSG diagnostic criteria in Spanish population would increase GDM prevalence from 11% to 36%, 4 an unaffordable burden which needs rethinking the current GDM clinical protocol.
Women with GDM should measure their blood glucose (BG) concentration at least four times daily (fasting and one hour after each meal) to detect hyperglycemia. Results are usually recorded in a glucose logbook, along with dietary information, allowing recognition of women who should begin insulin therapy to decrease the risk of macrosomia. 5 There is conflicting evidence as to whether ketonuria is associated with an adverse impact on fetal cognitive development but it is usually recommended to monitor fasting urinary ketones. 6
Telemedicine has been applied to pregnancies complicated by diabetes with the main impact being a reduction in the number of outpatient clinical encounters. 7 However, the lack of smart decision-support tools seems to be an obstacle for the general adoption of telemedicine systems in diabetes. 8 Face-to-face visits usually take a well-predefined time slot. However, using more flexible telemedicine applications without automated decision support, data transmission confirmation, analyses, and feedback to patients may be even more time consuming. Mobile applications, which are well accepted by patients with diabetes, allow using automatic processing tools to provide real-time advice based on BG monitoring. An example could be the automated confirmation that metabolic control parameters are maintained within normal levels, saving time to doctors and nurses.
There is limited experience in artificial intelligence methodologies applied to decision support for diabetes management. Nevertheless, the huge amount of available data that potentially influence glucose behavior has caught the attention of computer engineers on diabetes. MobiGuide is a mobile telemedicine system that can be run in Android devices, based on computer interpretable guidelines (CIG) for giving personalized decision support to GDM patients, which can be delivered anytime everywhere. The MobiGuide Project is an EU-F7 funded project that has developed a patient guidance system integrating hospital and monitoring data into a personal health record (PHR) accessible by patients and care providers. In addition, the system supply personalized secure clinical-guideline-based guidance also outside clinical environments. Its ubiquity has been achieved by having a decision support system (DSS) at the back end, and on the front end by utilizing body area network (BAN) technology and developing a coordinated light-weight DSS. Methodology to extract patient-oriented knowledge from clinical practice guidelines is specified elsewhere. 9 In brief, clinicians developed a consensus guideline which was formalized and then customization of the CIG was added to take into account patient context (eg, reduced number of BG measurements during periods of semiroutine meal schedules). The consequent plan activation or notification affected patients and, when necessary, doctors. For example, when ketonuria value was introduced by the patient as positive, a question was asked of her: “Have you been eating your prescribed amount of carbohydrates?” If the answer was “yes” and no previous notification had been sent before, then the notification to the patient was, “Ketonuria has been positive, please increase your carbohydrate bedtime by 10 grams.” The designed pilot study was aimed at assessing the feasibility of the system and the advantages of monitoring blood glucose, ketonuria, diet, blood pressure, and physical activity in GDM patients.
Patients and Methods
Twenty patients diagnosed with GDM were included in this pilot study. Inclusion criteria were GDM diagnosed according to National Diabetes Data Group criteria 10 before 34th gestational week, age older than 18 years, familiarity with smartphones and computer technology in general, and willingness to participate. We excluded the following categories of patients: diabetes diagnosed previously to the pregnancy (pregestational diabetes); hemoglobin A1c>6.5%; active treatment with drugs that could increase glycemia; blindness; severe mental disturbances; inability to speak/understand at least one of the following languages: Spanish, Catalan, or English; and frequent need to reside in remote areas without cellular network data coverage. The enrolled patients were given a monitoring system made by three components: (1) A smartphone device with 3G/4G data network capability and a specifically designed software to collect the clinical data and a messaging system (Figure 1), which included an accelerometer to detect the level of physical activity and an application that allowed one to manually introduce the results of fasting daily ketonuria determination (-/+/++); (2) a glycometer device with Bluetooth connectivity (Accu-Chek Aviva Connect, Roche Diagnostics GmbH, Mannheim, Germany); and 3) a blood pressure (BP) monitor with Bluetooth connectivity (Bluetooth Blood Pressure Monitor 708-BT, Omron Healthcare Co, Ltd, Kyoto, Japan). At the moment of the enrollment the patients were trained on the app as well as the different devices. Personalization was achieved by considering patient preferences (eg, sending reminders at patient-specific meal times) and context. The patient data needed for the DSS was copied from the hospital electronic medical record (EMR) (Hewlett Packard Healthcare Information System, Hewlett-Packard Development Company, L.P., Madrid, Spain ) into the PHR at the moment of enrollment and periodically refreshed to keep it up to date. HL7’s virtual medical record standard was used as the data model of the PHR. 11
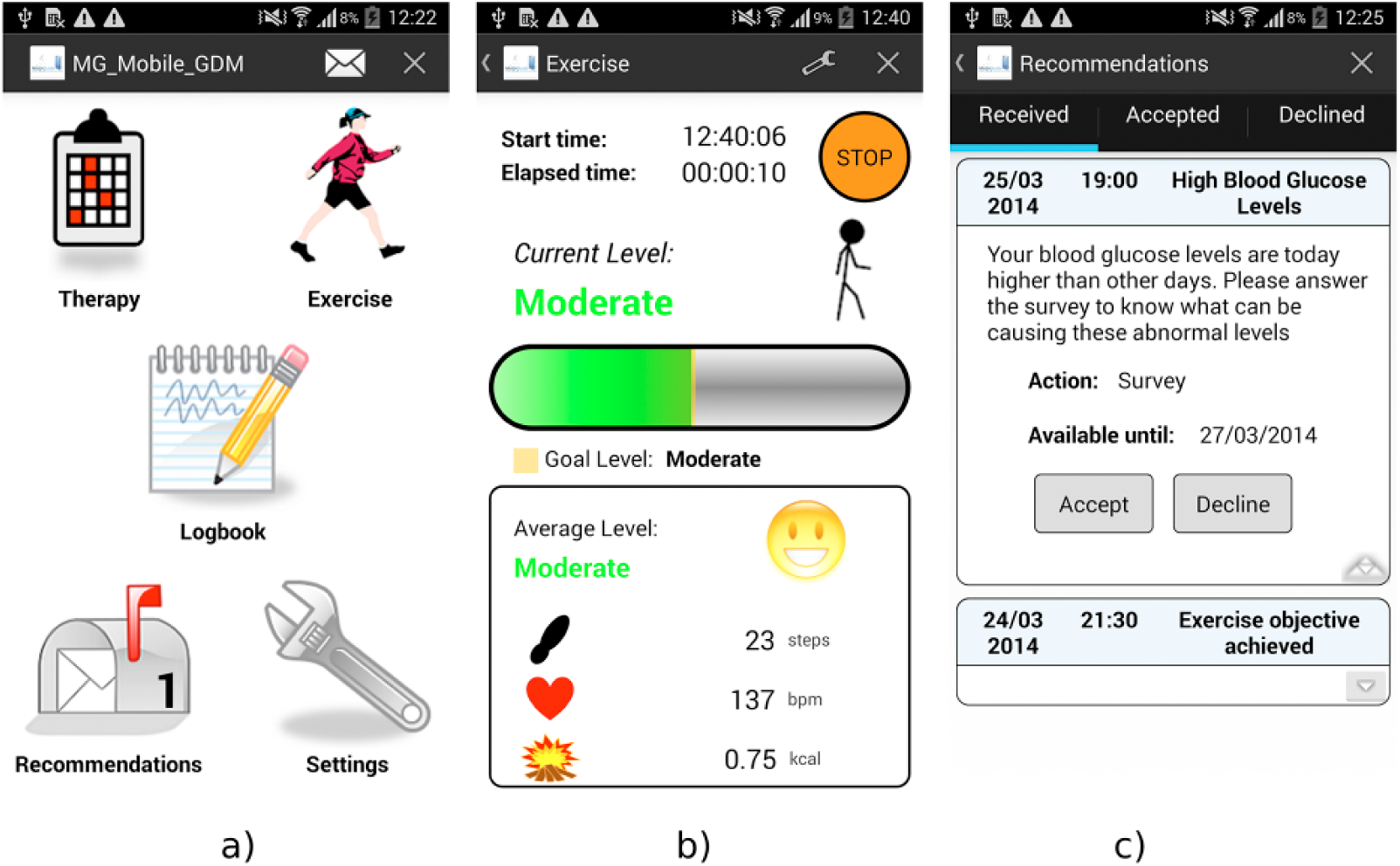
MobiGuide user interface examples. (a) Home screen. (b) Physical activity. (c) Advice delivered to the patient: survey for diet compliance because of hyperglycemia.
Patients were asked to download BG values (fasting and 1-hour postprandial) from the glycometer every 3 days as well as to inform on ketonuria status and diet compliance. Patients were also required to measure BP twice a week and advised to use the developed app to monitor physical activity episodes. Compliance was measured as the ratio “number of measurements downloaded”/“number of measurements expected.” During the study period patients were asked to follow their face-to-face visits with the nurse or doctor as usual. Health care providers involved in the study (doctors, nurses) used a web-based application to visualize all the patient data (EMR) including monitoring data, messages, and so on.
Patients satisfaction was evaluated using an ad-hoc questionnaire that patients filled in at the end of the study. Each question was answered with a 5 point semantic differential scale (the higher the better).
During the technical development of the MobiGuide system (2012-2014), we performed in parallel an observational prospective study to capture the main relevant clinical data for describing our usual population of GDM patients with respect to basal conditions, metabolic outcomes, delivery-related variables and neonatal complications.
Both studies (pilot study and the observational prospective GDM cohort study) were approved by the local ethics committee and patients signed the informed consent.
The statistical analysis was performed using SPSS software (IBM Software, version 23). Values are shown as mean ± standard deviation for normally distributed variables and median (interquartile range) for the ones nonnormally distributed. We used t-test or Mann-Whitney U test for mean comparison of numerical variables and chi-square for categorical data.
Results
All the patients completed the study except for one who moved to other region. The data set contains a total of 4561 BG measurements, 997 ketonuria values, 369 BP measurements, and 184 exercise bouts. Main clinical characteristics are shown in Table 1 and compared with those corresponding with the historical cohort. In brief, patients enrolled in the pilot study seem to be representative of the usual patients referred because of GDM diagnosis.
Comparison of Main Characteristics for the Intervention Group and the Historical Cohort.
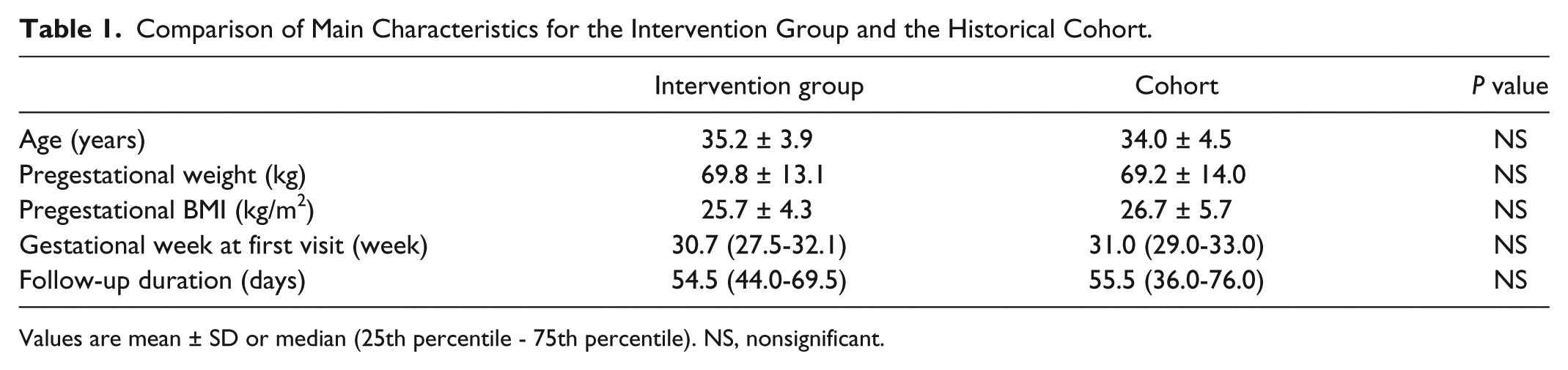
Values are mean ± SD or median (25th percentile - 75th percentile). NS, nonsignificant.
Tables 2 and 3 show the comparison of the metabolic and perinatal outcomes between telemedicine users and the historical cohort. The only differences observed between groups were systolic and diastolic BP, which were lower in the intervention group.
Metabolic Outcomes for Patients in the Intervention Group Compared With Those Observed in the GDM Historical Cohort.
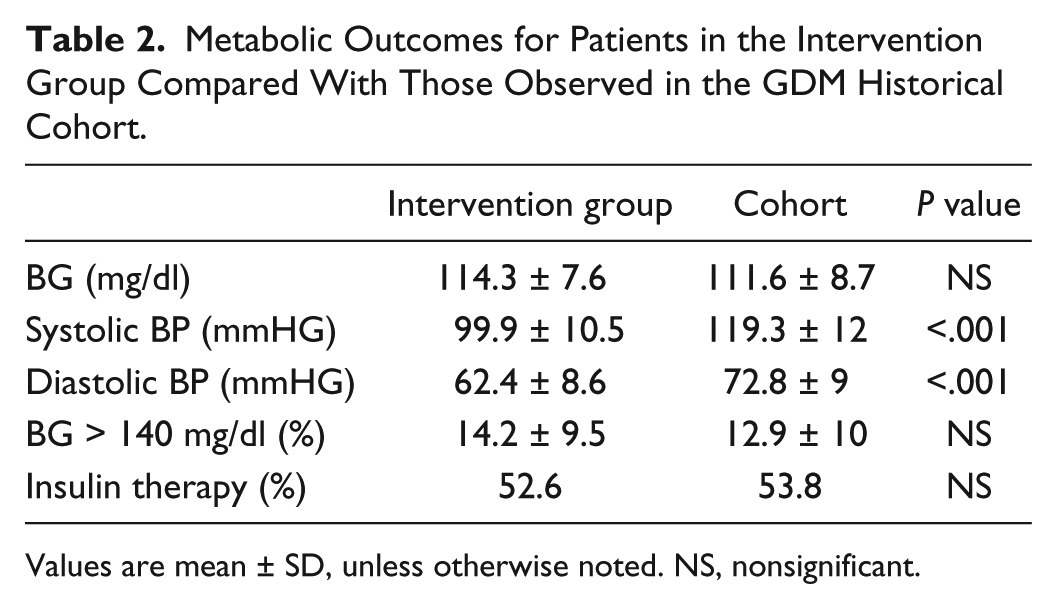
Values are mean ± SD, unless otherwise noted. NS, nonsignificant.
Comparison of Perinatal Outcomes Between the Group of Women Who Used Smart Telemedicine System and the Historical Cohort.
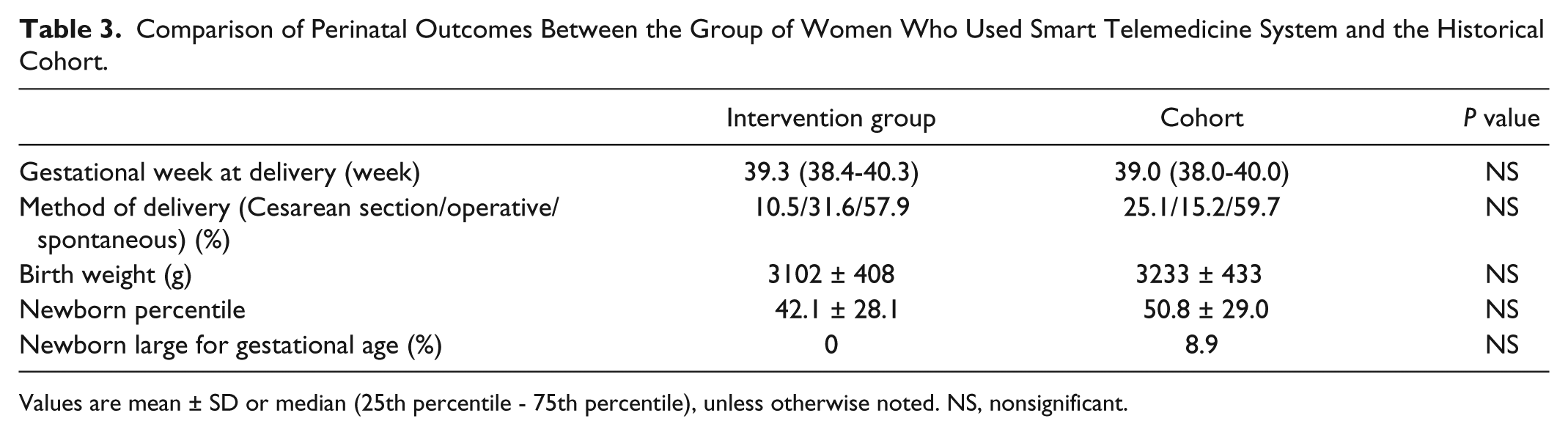
Values are mean ± SD or median (25th percentile - 75th percentile), unless otherwise noted. NS, nonsignificant.
Patients in the study downloaded BG data every 2.97 ± 1.6 days and showed a higher degree of compliance (1.01 ± 0.1 vs 0.87 ± 0.3, P < .05) than that observed in the historical cohort. BP data was downloaded every 7.6 ± 5.9 days and mean compliance with respect to BP measurements was 0.81 ± 0.2. Ketonuria status was informed every 3.2 ± 1.7 days, and mean patients compliance with ketonuria determination was 0.98 ± 0.04. Patients attended the diabetes and pregnancy clinic a median of 6 (5-7) times.
All the patients received at least one message asking about diet compliance because of hyperglycemia and 6 patients were also asked because of ketonuria. Seven patients were advised to contact the doctor because of persistence of the metabolic problem despite of prescribed diet change when appropriate. In 16 patients the system automatically reduced the recommended frequency of BP/ketonuria monitoring because of persistent normal values and high compliance.
Figure 2 show the results of the participants end-of-study questionnaire, mirroring a high degree of patients satisfaction with the system.
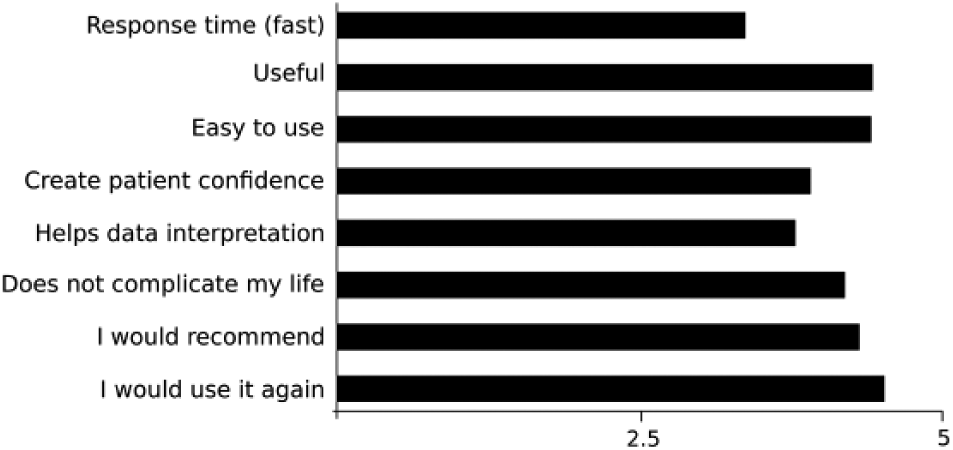
Results of the participants end-of-study questionnaire (5 = strongly agree).
Discussion
Our study results indicate that the developed smart telemedicine system is a feasible and well-accepted mobile DSS for guiding GDM. Furthermore, compliance with BG monitoring performance was clearly higher than that observed with usual care. Our interpretation is that immediate data analysis and feedback engage patients on performing BG measurements. In addition, compliance with BP regular measurement was very high considering our group of normotensive pregnant women. Another important contribution of the system was the automated plan of diet adjustments to correct either hyperglycemia or ketonuria. CIGs are usually applied to help professionals to make better decisions. Our smart telemedicine patient guidance system, helps patients through advice in accordance with the specific clinical guideline.
Metabolic and perinatal outcomes were similar to those results showed in our cohort of GDM patients except for BP, which was lower in the intervention group. The most plausible explanation is that BP in patients using telemedicine was measured at home, and probably for this reason the mean values were lower than those observed in the cohort of GDM patients.
A few randomized control trials have addressed the effect of telemedicine interventions on GDM management.12-16 Very recently, a metanalysis found a mild but significant effect of telemedicine solutions on HbA1c in comparison with usual practice (5.22 ± 0.7 % vs 5.37 ± 0.6; mean difference –0.14% [95% CI −0.25, –0.04%]). 17 Another previous metanalysis found fewer unscheduled visits while maintaining quality of life, BG levels and rate of cesarean section. 7 Telemedicine interventions applied to GDM were based on data transmission with or without reminders but in all cases required expert analysis for giving asynchronous feedback to patients. The developed smart mobile telemedicine system is a new and unique system: incorporates CIG, integrates sensors, retrieves key data from the EMR, takes into account personal context variables, and proposes diet changes without medical supervision to correct hyperglycemia and/or ketonuria.
Our study has some limitations. The study group was small, and no control group was included. However, a comparison with a wide cohort group was made to evaluate the telemedicine system in terms of safety and efficacy. Hardware was available to all the patients, and no limitations were imposed. However, a few patients were excluded because of limited technical skills. In consequence, even though the telemedicine system tested a very user-friendly application, in real life there may be limitations for this reason.
Conclusions
GDM is the most prevalent metabolic alteration diagnosed during pregnancy. As BG levels tend to increase week after week, weekly face-to-face visits are the current recommended protocol, producing a huge burden for health care providers. Artificial-intelligence-augmented telemedicine can offer a good alternative, saving resources while maintaining the standards of care proposed in the clinical guidelines.
The data presented in this pilot study show the feasibility and acceptance of the developed smart telemedicine system DSS, a mobile application that includes CIG, access to data from the EMR, as well as access to data downloaded from the glycometer, BP monitor, and activity sensors. Patients’ compliance with BG tests, the key parameter for GDM monitoring, seems to benefit from the immediate analysis and feedback. The next step would be a comparison of the usual care with the use of this new telemedicine system in a randomized controlled trial.
Footnotes
Acknowledgements
The authors wish to thank the patients for their enthusiastic participation in this study.
Abbreviations
BAN, body area network; BG, blood glucose; BP, blood pressure; CIG, computer interpretable guidelines; DSS, decision support system; EMR, electronic medical record; GDM, gestational diabetes; IADPSG, International Association of Diabetes and Pregnancy Study Group; PHR, personal health record.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by the European Commission through the FP7 MobiGuide Project.
