Abstract
The pentose xylose is enriched in edible algae, and is increasingly used as a slowly metabolized carbohydrate in functional food. It is known to interfere with glucose-dehydrogenase-based (GDH) blood glucose measurement systems for patients self-testing. The aim of our study was to investigate the extent of xylose interference in commercially available blood glucose meters. A heparinized whole blood sample was manipulated to contain 3 different glucose concentrations (50-80 mg/dL, 130-160 mg/dL, and 250-300 mg/dL) and 4 different xylose concentrations (0 mg/dL, 25 mg/dL, 50 mg/dL, and 100 mg/dL). Each sample was measured 3 times with 2 different strip lots per test meter (AccuChek Aviva, AccuChek Connect, Contour Next, FreeStyle Freedom Lite, FreeStyle Insulinx, MyStar Extra, OneTouch Verio IQ, and Wellion Calla, reference: YSI GlucoStat analyzer). For analysis, we calculated the xylose capture rate, that is, the xylose amount wrongly displayed as glucose. No xylose interference was seen with 4 meters: AccuChek Aviva (mean capture rate 0%), AccuChek Connect (–2%), MyStar Extra (10%), and Wellion Calla (8%). In contrast, substantial interference was observed with Contour Next (100%), FreeStyle Freedom Lite (104%), FreeStyle Insulinx (120%), and OneTouch Verio IQ (162%). We observed xylose interference in several GDH-based meters. This may become important with increased use of xylose in dietary and functional food products, in particular in products designed for weight loss. Our findings may affect the meter selection for patients who are consuming such food products as part of their lifestyle treatment regimen.
The pentose carbohydrate xylose is the main component for the hemicellulose xylan, which comprises about 9-30% of plants. Xylose is found in the seeds and embryos of most edible plants (eg, guava, pears, blackberries, loganberries, raspberries, aloe vera gel, kelp, echinacea, boswellia, psyllium, broccoli, spinach, eggplant, peas, green beans, okra, cabbage, and corn). It was first isolated from wood by Finnish scientist Koch in 1881. 1 Xylose is metabolized by the human organism, but it is not a major human nutrient, and consequently largely excreted by the kidneys. It is incompletely absorbed by passive diffusion in human duodenum and jejunum, by a mechanism that is different to the transporter for glucose. Although absorption seems to be slower in children when given orally, it does not appear to be age-dependent in adults since other factors are more likely to be involved in absorption rates. Once Xylose enters the bloodstream, it is quickly distributed to the liver where it is partly metabolized. Xylose also travels to other tissues where it is required, including the kidneys, fat, and muscles. 2 The unabsorbed remainder is delivered to the ileum and the colon, where bacterial fermentation occurs, producing hydrogen that can be detected in the breath. 3 The most interesting property of xylose for the current food industry is its caloric value: xylose metabolically contains 0 calories per gram, which as investigated almost 80 years ago. 4 Oral consumption of D-xylose stimulates GLP-1 secretion to a greater and more sustained degree than glucose in healthy older subjects, 5 because the comparably larger length and region of small intestine exposure to xylose determine a larger GLP-1 release. 6 D-Xylose also slows gastric emptying compared with water. 7 In animal experiments, D-xylose exerted antidiabetic effects by regulating blood glucose levels via regeneration of damaged pancreas and liver tissues, and regulation of PEPCK, a key rate-limiting enzyme in the process of gluconeogenesis. 8 It has, for example, been shown that coconut-derived D-xylose favorably affected postprandial glucose and insulin response when used as a supplement with sucrose in men. 9
As repeated glucose and insulin excursions are closely related to metabolic stress, the proper application of xylose in different sucrose-based products may lessen the undesired effects of sucrose. Subsequently, xylose has become a carbohydrate of interest for use in functional food products, in particular for products recommended for dietary use in diabetes and obesity. It can, therefore be expected that regular xylose uptake will become a more frequent condition in the daily life of diabetes patients. In addition, the number of people living on a vegetarian or vegan diet is currently increasing and their food represents a source of substantial xylose uptake. This situation has to be considered, when selecting blood glucose meters for patient self-testing of blood glucose, as blood glucose strip technologies based on glucoside-hydrogenase have been identified to be subject to interference by xylose.10,11
No or only very limited data exist about the real potential risk for wrong readings for patients consuming xylose containing food. It may be useful to quantify the influence of xylose on glucose meter readings to be able to define educational needs, if applicable. The aim of our study was to quantify the potential interference of xylose with the readings of several commercially available blood glucose meters for patient self-testing.
Patients and Methods
The blood for this laboratory experiment was drawn from a healthy donor based on an IRB-approved blood sampling study in compliance with local and international legal requirements. .
The devices and strips tested (2 meters/product, 2 strip lots) were obtained through regular distribution channels. We purchased the following devices: AccuChek Aviva and AccuChek Connect (both Roche Diagnostics, Mannheim, Germany, glucose dehydrogenase method), Contour Next (Ascensia, Leverkusen, Germany, glucose dehydrogenase method), FreeStyle Freedom Lite and FreeStyle Insulinx (both Abbott, Wiesbaden-Erbenheim, Germany, glucose dehydrogenase method), MyStar Extra (Sanofi, Berlin, Germany, glucose oxidase method), OneTouch Verio IQ (Lifescan, Neuss, Germany, glucose dehydrogenase method), and Wellion Calla (MedTrust, Dresden, Germany, glucose oxidase method). The YSI 2300 STAT PLUS Glucose Analyzer (Life Sciences/Kreienbaum, Langenfeld, Germany, glucose oxidase method) was utilized as in-house reference method. The samples were tested with the reference method before and after completion of the experiments for each manipulated sample.
All experiments were performed under similar environmental conditions. Samples with different blood glucose concentrations (50-80 mg/dL, 130-160 mg/dL, and 250-300 mg/dL) were artificially prepared by spiking with highly concentrated glucose solutions or by artificial glycolysis outside of the body. Four different levels of xylose were artificially prepared for each glucose concentration: 0 mg/dL, 25 mg/dL, 50 mg/dL, 100 mg/dL. After xylose adjustment, the final glucose concentration was determined with the reference method, and the degree of oxygenation (85-100%) and the hematocrit (35-45%) of the prepared samples were confirmed to be in the target ranges.
For each device type, 2 meters were tested in parallel with 3-fold measurements with 2 strip lots each for all 12 prepared blood samples, resulting in 144 measurements per device. Before and after sample measurements, quality control measurements were performed using the respective control solutions provided by the manufacturers.
Statistical Analysis
Basis for the analysis were the methods as set forth by ISO15197 and National Laboratory Guideline for Chemical Interference Testing. The mean values and standard deviations for each meter type/sample combination were calculated and the coefficient of variation was determined. In addition, the mean absolute deviation from the reference methods was calculated for all 3 glucose ranges. The mean glucose values of the xylose-free samples were set to 100% to determine the percentage deviation at the 3 other different xylose levels. For each meter, the mean xylose interference at all xylose concentrations was calculated to determine the amount of xylose present in the sample that is wrongly displayed as glucose (xylose capture rate). The data were graphically displayed for each meter type and glucose concentration and for the entire sample set.
Results
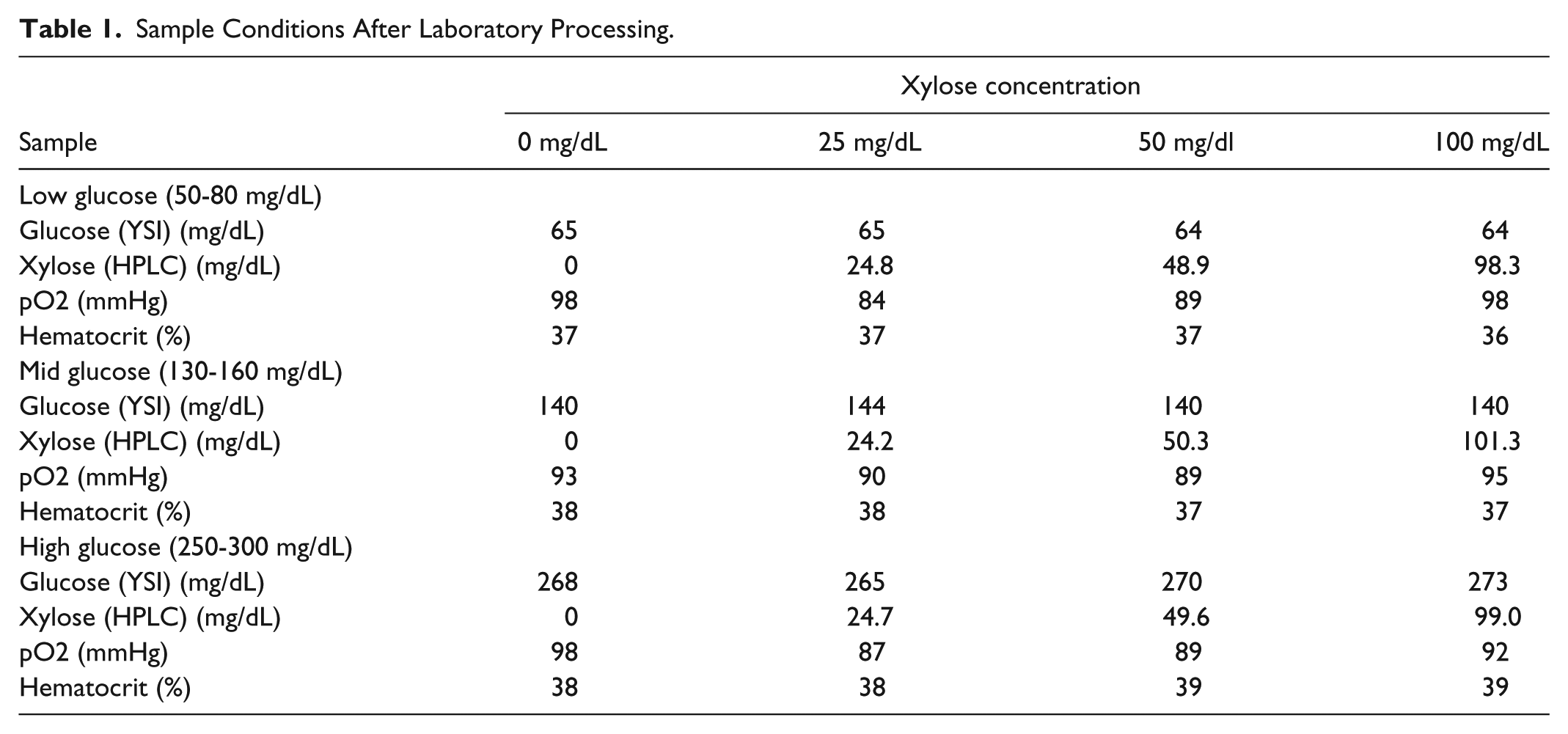
The entire laboratory experiment was performed during 1 day and under stable environmental conditions. Blood was drawn from a healthy female voluntary healthy donor (age 30 yrs.) and immediately processed in the laboratory. All samples were prepared in accordance to the laboratory protocol. Oxygen saturation as well as hematocrit were within physiological levels in all cases prior to the performance of the experiments. All tested devices delivered acceptable results when tested with the manufacturer’s control solutions before and after the experiment. The obtained glucose and xylose concentrations were within the anticipated ranges. The sample characteristics after manipulation are listed in Table 1.
Sample Conditions After Laboratory Processing.
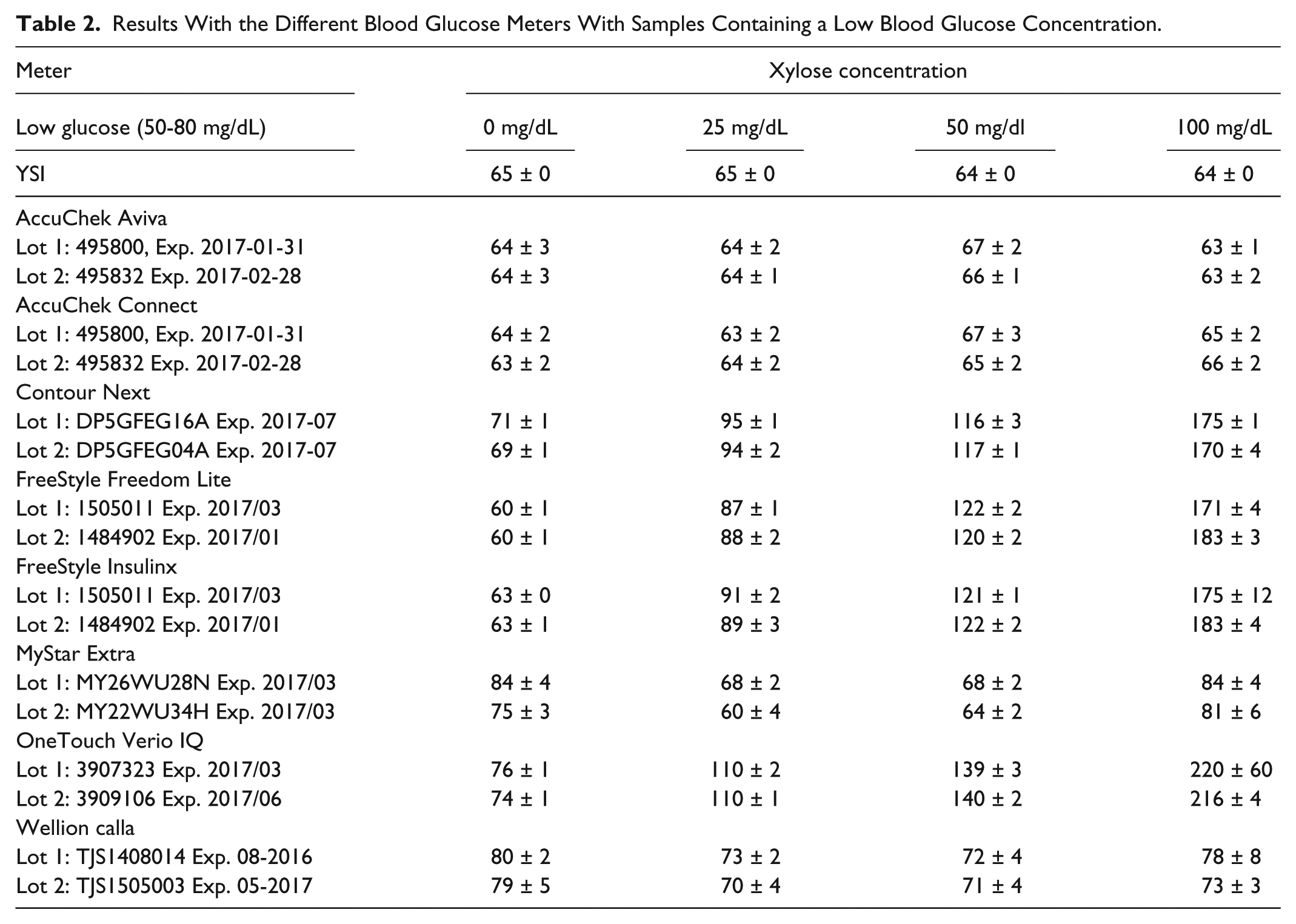
As an example for the general performance of the meters, the results obtained with the 2 lots of the different meters for the samples with low blood glucose are provided in Table 2. It can be seen that the respective 2 lots showed similar results with the respective samples, even in case of major reading deviations The deviations of the readings from the baseline glucose level as assessed without xylose addition, which was set to zero, for the individual meters and strip lots are provided in Figure 1 and Figure 2. There was no substantial xylose interference for AccuChek Aviva, AccuChek Connect, MyStar Extra, and Wellion Calla. The meters with significant additional signal generation by xylose were Contour Next, FreeStyle Freedom Lite, FreeStyle Insulinx, and OneTouch Verio IQ.
Results With the Different Blood Glucose Meters With Samples Containing a Low Blood Glucose Concentration.
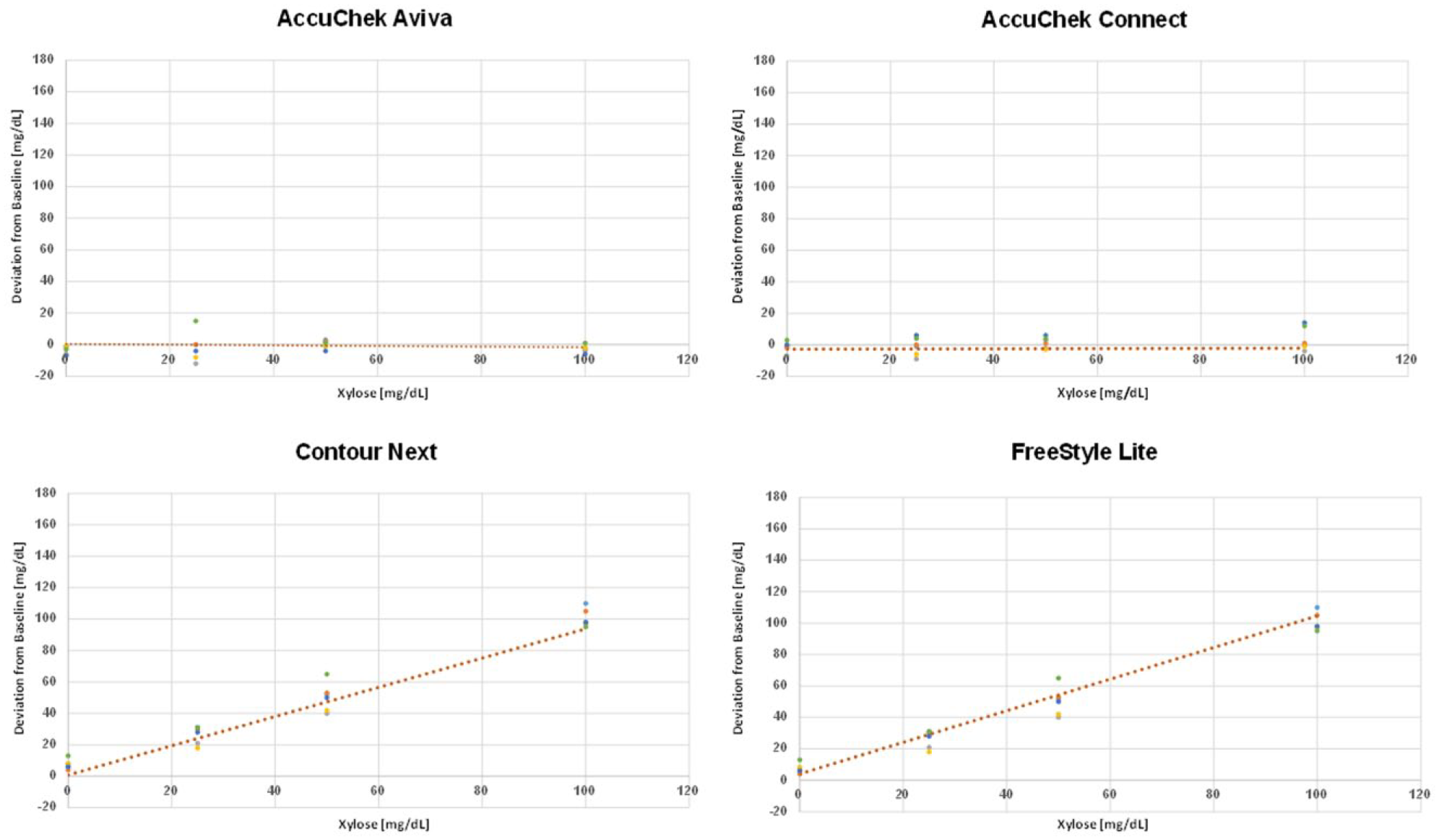
Changes of the absolute meter results when xylose was added to the samples with different glucose levels as assessed by AccuChek Aviva, AccuChek Connect, Contour Next, and FreeStyle Lite. Each data point represents the additional reading increment for 1 strip lot leading to 6 data point per xylose concentration.
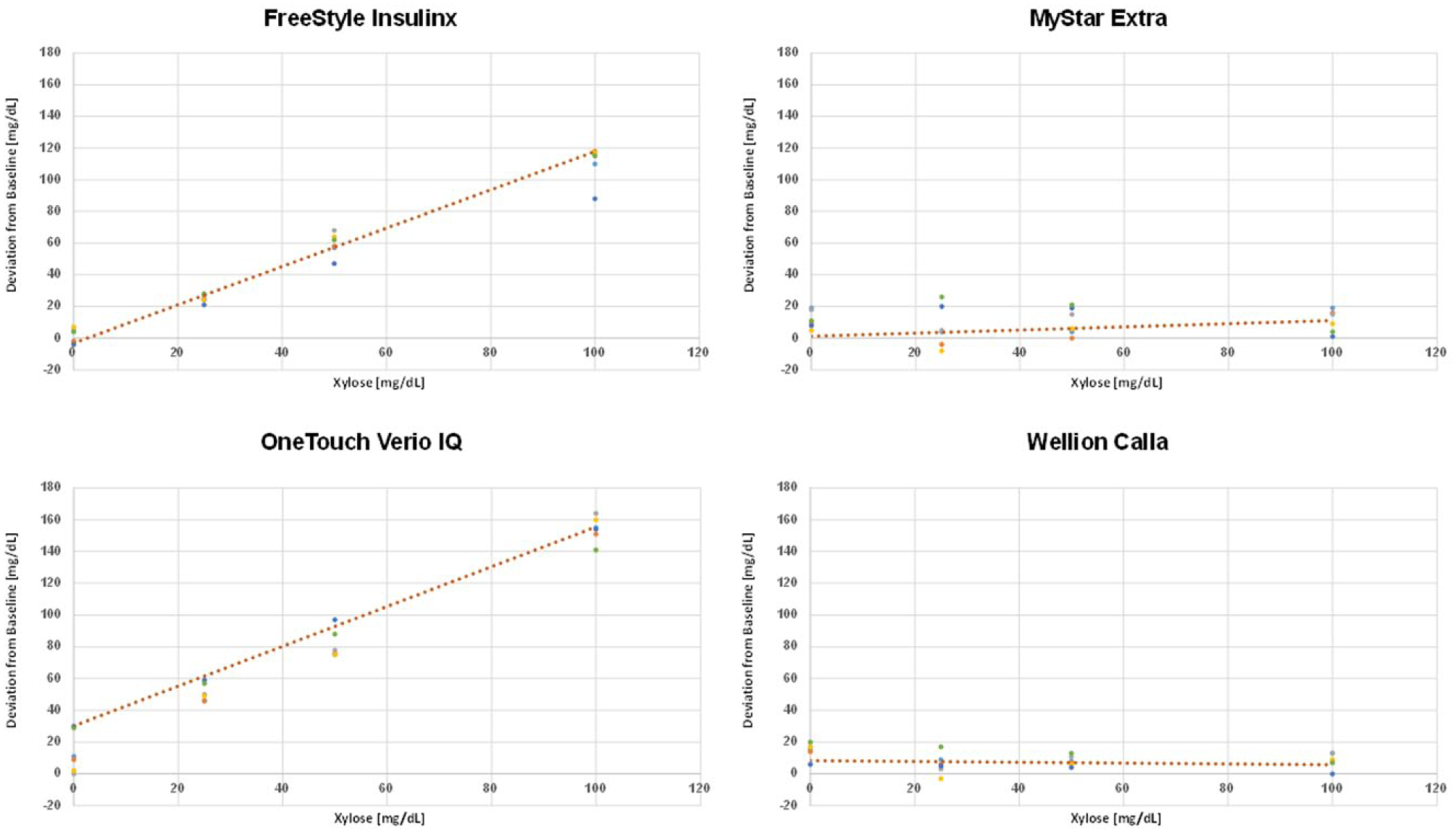
Change of the absolute meter results when xylose was added to the samples with different glucose levels as assessed by FreeStyle Insulinx, MyStar Extra, OneTouch Verio IQ, and Wellion Calla. Each data point represents the additional reading increment for 1 strip lot leading to 6 data point per xylose concentration.
The mean influence of xylose on the overall meter signal can be expressed as a xylose recovery rate. The xylose recovery rates for the different patient self-test meters are provided in Figure 3. It was highest in the OneTouch Verio IQ meter (162%), followed by the FreeStyle devices (Insulinx: 120% and Lite: 104%) and Contour Next (100%).
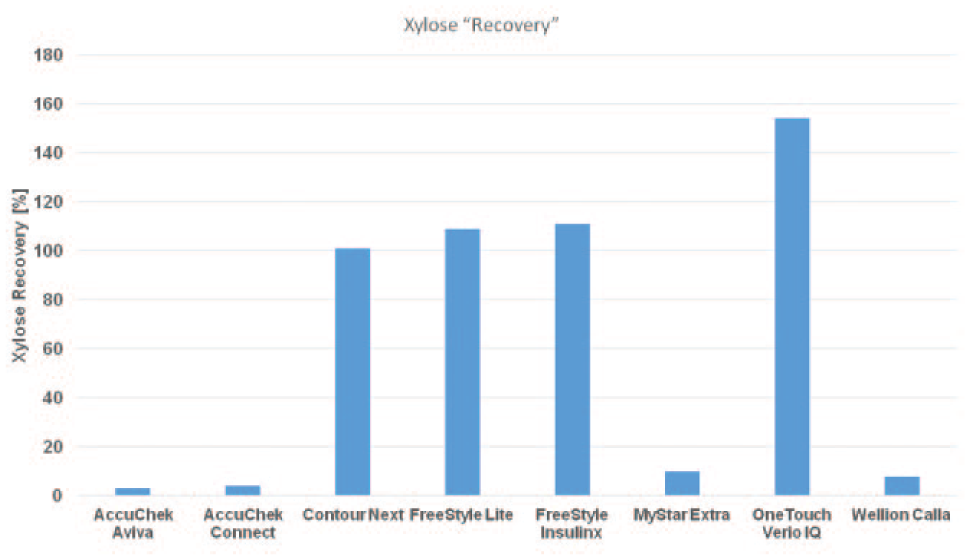
Sensitivity of the different blood glucose meter systems to xylose interference. Calculated as mean xylose recovery rate in comparison to the unspiked samples.
Discussion
The concept of foods specifically developed to promote health or reduce the risk of disease was introduced.in Japan in the 1980s. It was recognized that an improved quality of life must accompany increasing life expectancy for the expanding number of elderly people in the population, if health care costs were to be controlled. Functional foods have not been legally defined in the United States or in Europe yet. Generally, they are considered as foods that are intended to be consumed as part of the normal diet and that contain biologically active components, which offer the potential of enhanced health or reduced risk of disease. Examples of functional foods include foods that contain specific minerals, vitamins, fatty acids or dietary fibers, foods with added biologically active substances, such as phytochemicals or other antioxidants and probiotics that contain live beneficial bacterial cultures.12,13
The sweetness of xylose is about 70% of cane sugars. D-Xylose is accepted by the US Food and Drug Administration as a safe food ingredient (GRAS). In the EU, xylose’s status is also food ingredient. In functional foods, xylose provides a multifunctional solution. It is a food application, feed application, sweetener, flavor agent, browning agent and is 100% natural, while containing 0 calories. 4 D-Xylose is used in food industries, in the production of savory flavors, as it is highly effective at inducing Maillard reactions, which helps to produce golden brown color in food applications such as batters and breadcrumbs.14,15
Next to its negligible caloric value, there are several reasons why polysaccharides containing xylose as a major component represent attractive candidates for functional food products:
Xylose is an antibacterial and antifungal, particularly with-gram negative organisms and candida. 16
It is proving to help prevent cancer of the digestive tract.17,18
Absorption rate of Xylose is decreased in some patients with intestinal disorders, including those with colitis, diabetes, and chronic alcoholism, suggesting that when absorption problems are corrected, these conditions might also reverse themselves.19,20
Unlike sucrose or artificial sweeteners, Xylose promotes the growth of “friendly flora” in the intestines, thus increasing the manufacture and absorption of all nutrients. This aspect is necessary in strengthening the immune system to help fight off any type of illness. 21
Xylose is safely consumed in daily amounts as great as 35 grams for a 150-pound adult and a minimum amount for substitution has not yet been determined. It can, therefore be expected that with the success of functional food introduction into daily nutrition, xylose will be more prevalent in the blood stream of patients with diabetes using insulin and other antidiabetic drugs who are performing frequent blood glucose measurements on a daily basis. Xylose has on the other hand been identified as a potential interfering substance in blood glucose meters employing glucose-dehydrogenase based strip technologies.10,11 According to our knowledge, the extent of this effect has not been explored in a comparative investigation yet.
In our laboratory study, the YSI reference method and 4 of the patient self-test meters (AccuChek Aviva, AccuChek Connect, MyStar Extra, and Wellion Calla, all <10% of xylose interference) were uninfluenced by xylose. In contrast, Contour Next (100%), FreeStyle Freedom Lite (104%), FreeStyle Insulinx (120%) and OneTouch Verio IQ (162%) were markedly influenced by this interference factor. It is noteworthy that all affected meters are employing a glucose-dehydrogenase-based technology. MyStar Extra and Wellion Calla are based on glucose oxidase technologies and are calibrated versus glucose oxidase-based reference methods (as is the YSI point-of-care glucose analyzer). Glucose oxidase does not bind xylose, which may be the main obvious reason for the stability against xylose interference.
The 2 AccuChek devices, which are also employing a glucose dehydrogenase technology like the affected devices, showed also no interfering potential of xylose. The major difference between the AccuChek technology and the affected technologies is that the AccuChek strips are calibrated with a glucose hexokinase laboratory reference method, while the other devices are calibrated against glucose oxidase or glucose dehydrogenase-based reference methods. The exact mechanism by which the AccuChek devices avoid or correct for xylose interference has not been disclosed, but it can be speculated that the manufacturer has identified and is using a glucose-dehydrogenase mutant, which is not subject to xylose interference.
Xylose is a known substrate of glucose dehydrogenase, and it is therefore, for example, recommended to not use these devices in the timely context of a malabsorption test procedure with application of 15 g of xylose. While lower absorption levels indicate a potential malabsorption syndrome, and in particular celiac disease in children, normal absorption of this xylose quantity results in 1 h blood xylose concentrations of up to 50-60 mg/dL.22-24
Our study has several limitations. It was a laboratory experiment and performed with manipulated venous blood samples containing artificially produced xylose concentrations. Before drawing an ultimate conclusion regarding the clinical relevance of our findings, it is required to perform confirmatory clinical studies with oral xylose uptake and use of the devices with capillary whole blood and test performance in accordance with the instructions for use.
However, in light of increasing xylose concentrations in functional food products, our findings may already now have some clinical relevance. In case of a substantial impact of xylose on the meter readings, the clinically most dangerous condition would be the presence substantial amounts of glucose during a period of low glucose values. This risk should be discussed in diabetes education programs and may be considered when selecting meters for patients who intend to use functional food as part of their daily nutrition.
Footnotes
Abbreviations
GDH, glucose dehydrogenase; GLP-1, glucagon-like peptide 1; GRAS, generally recognized as save; HPLC, high-pressure liquid chomatography; IRB, institutional review board; PEPCK, phosphoenolpyruvate-carboxykinase; pO2, oxygen pressure in the blood; YSI, Yellow Springs glucose analyzer.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AP has received consultancy fees, travel support, and research grants from Sanfoi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by Sanofi.
