Abstract

For people with diabetes mellitus, self-measured blood glucose (SMBG) using a portable blood glucose (BG) meter plays a key role in controlling BG level and is an important component of diabetes management.1,2 In contrast with SMBG systems based on traditional static electrochemical technology, the BGStar® (AgaMatrix, Salem, New Hampshire, USA) SMBG meter uses dynamic electrochemistry and the glucose oxidase measurement principle, and has been shown to correct for hematocrit interference on BG measurement within the system’s operating limits. 3 This SMBG meter was not approved in China when the present study was initiated, and we evaluated its clinical accuracy and safety for SMBG measurement in diabetic and nondiabetic Chinese people.
In this study, accuracy assessments were performed in accordance with the International Organization for Standardization (ISO) 15197:2003 standard (the current version at the time of study initiation), and the China Food and Drug Administration’s (CFDA) requirements. 4 Reference BG measurements were acquired from 2 systems; the manufacturer’s recommended reference, the YSI 2300 STAT PlusTM Glucose and Lactate Analyzer (YSI Life Sciences, USA), and a CFDA-approved system, the StatStrip® Glucose Connectivity Meter (Nova Biomedical, USA). Two BG measurements were made using each reference method before and after duplicate measurements using the SMBG meter and system accuracy was assessed as described in ISO 15197:2003. A post hoc evaluation using ISO 15197:2013 was also performed. 5
Of 412 individual SMBG meter results obtained from 206 evaluable participants, >95% deviated within ±15 mg/dL for BG values <75 mg/dL and ±20% for BG values ≥75 mg/dL in comparison with the mean YSI (99.5%) and StatStrip (99.0%) measurements, and therefore exceeded the minimum ISO 15197:2003 accuracy requirement (Table 1). Passing and Bablok regression analysis correlation coefficients were 0.99 and 0.98 against YSI and StatStrip, respectively. Consensus Error Grid analysis revealed that 100% of data points fell in zones A and B. Using ISO 15197:2013; >95% of individual SMBG meter values deviated within ±15 mg/dL for BG <100 mg/dL or ±15% for BG ≥100 mg/dL in comparison with the mean YSI (98.5%) and StatStrip (95.6%) measurements (Table 1). The SMBG meter was safely operated; no device-related AEs were reported during the study. In addition, no product technical complaints were reported during the study.
System Accuracy of a SMBG Meter Assessed Using the ISO 15197:2003 Standard Stratified by Blood Glucose Levels < 75 mg/dL or ≥ 75 mg/dL, and the ISO 15197:2013 Standard Stratified by Blood Glucose Levels < 100 mg/dL or ≥ 100 mg/dL.
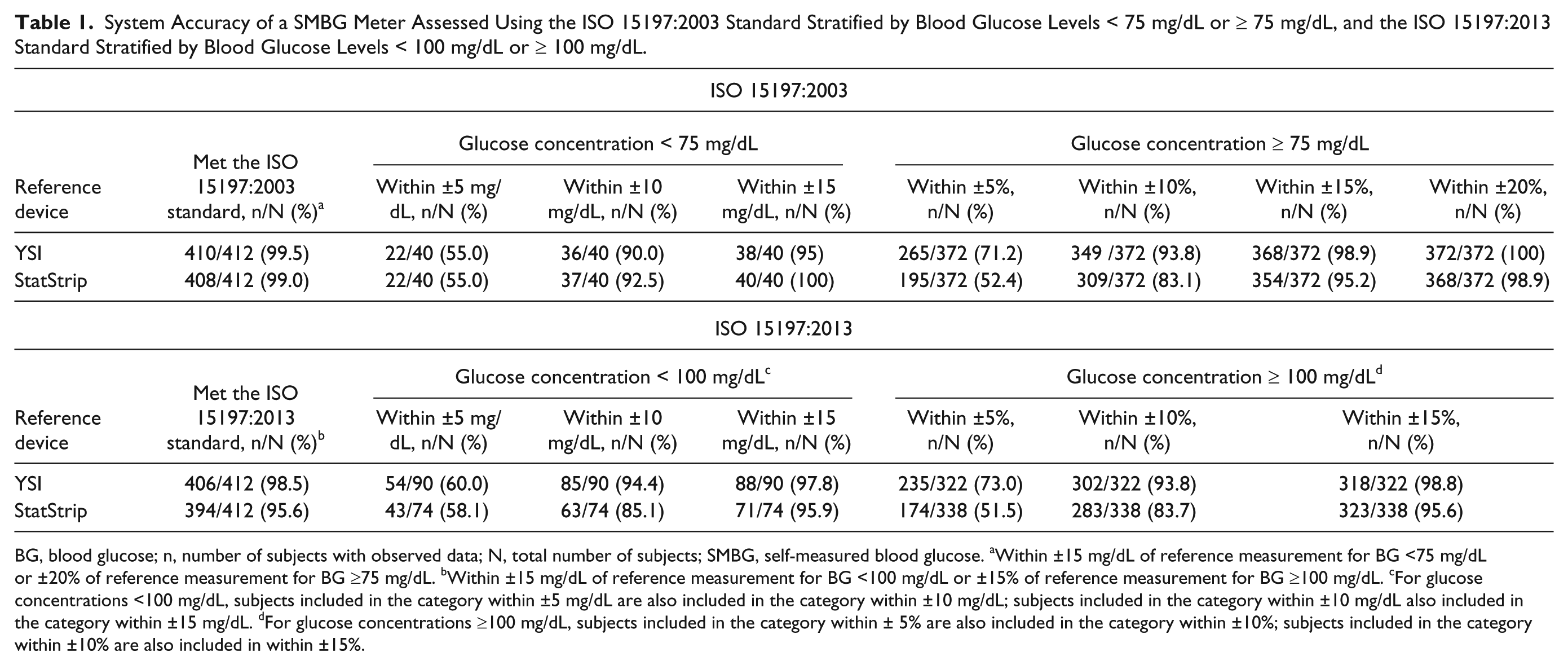
BG, blood glucose; n, number of subjects with observed data; N, total number of subjects; SMBG, self-measured blood glucose. aWithin ±15 mg/dL of reference measurement for BG <75 mg/dL or ±20% of reference measurement for BG ≥75 mg/dL. bWithin ±15 mg/dL of reference measurement for BG <100 mg/dL or ±15% of reference measurement for BG ≥100 mg/dL. cFor glucose concentrations <100 mg/dL, subjects included in the category within ±5 mg/dL are also included in the category within ±10 mg/dL; subjects included in the category within ±10 mg/dL also included in the category within ±15 mg/dL. dFor glucose concentrations ≥100 mg/dL, subjects included in the category within ± 5% are also included in the category within ±10%; subjects included in the category within ±10% are also included in within ±15%.
Although our study protocol was designed with reference to ISO 15197:2003, a post hoc analysis showed that >95% of SMBG meter measurements agreed with the mean YSI reference measurements within the more stringent tolerances set out in the current ISO 15197:2013 standard.4,5 Unfortunately, further changes in ISO 15197:2013 such as acquiring data from 3 different reagent system lots (3 test strip lots) and testing by the intended users were not included in the present study design. Despite these limitations, the strengths of our study include the careful study design and conduct, and the use of 2 reference methods to fulfill and overcome specific regulatory requirements.
In summary, this study confirms that the SMBG meter evaluated exceeds the minimum accuracy standards for BG measurement as defined by ISO 15197:2003 in diabetic and nondiabetic Chinese people.
Footnotes
Acknowledgements
The manuscript was prepared by the authors with medical writing support from BC-biostat and Jake Burrell at Adelphi, which was paid for by Sanofi. Sanofi was permitted to review and comment on the manuscript, but the final decision on the content was exclusively retained by the authors.
Abbreviations
AE, adverse event; BG, blood glucose; CFDA, China Food and Drug Administration; DM, diabetes mellitus; ISO, International Organization for Standardization; POC, point-of-care; pO2, pressure of oxygen; SMBG, Self-monitoring of BG.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AB and LD are employees of Sanofi. The other authors have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Sanofi.
