Abstract

Keywords
Self-management is the key to diabetes therapy.
1
Information communication technology—including smartphone applications—is recognized as a promising means of allowing timely intervention to support self-management by diabetes patients.
2
We developed a real-time automated interactive system—DialBetics—to help patients achieve that self-management.
3
A randomized control trial for 54 type 2 diabetes patients who were not on insulin injections showed that hemoglobin A1c (HbA1c) of the group using DialBetics improved significantly after 3 months: from 7.1 ± 1.0% to 6.7 ± 0.7% versus the control group’s 7.0 ± 0.9% to 7.1 ± 1.1% (
DialBetics comprises 3 modules:
Data transmission: Patients’ blood glucose, blood pressure, body weight, and pedometer counts are measured at home and sent to the server computer twice a day.
Evaluation: Data are automatically evaluated following the Japan Diabetes Society guideline’s targeted values: optimally, blood sugar <110 mg/dl before breakfast and <140 mg/dl at bedtime; blood pressure <130/80 mm Hg; and pedometer count >10,000. The module determines if each reading satisfies guideline requirements then sends results to each patient’s smartphone. Abnormal readings—defined as blood sugar >400 mg/dl or <40 mg/dl and systolic blood pressure >220 mm Hg—are reported as “Dr. call,” meaning a physician will check the data and interact with the patient if necessary.
Communication: Patients’ voice/text messages detailing meals (with photos) and exercise are sent to the server. Advice on lifestyle modification, matched to the patients’ input about diet and exercise, is returned to each patient (Figure 1).
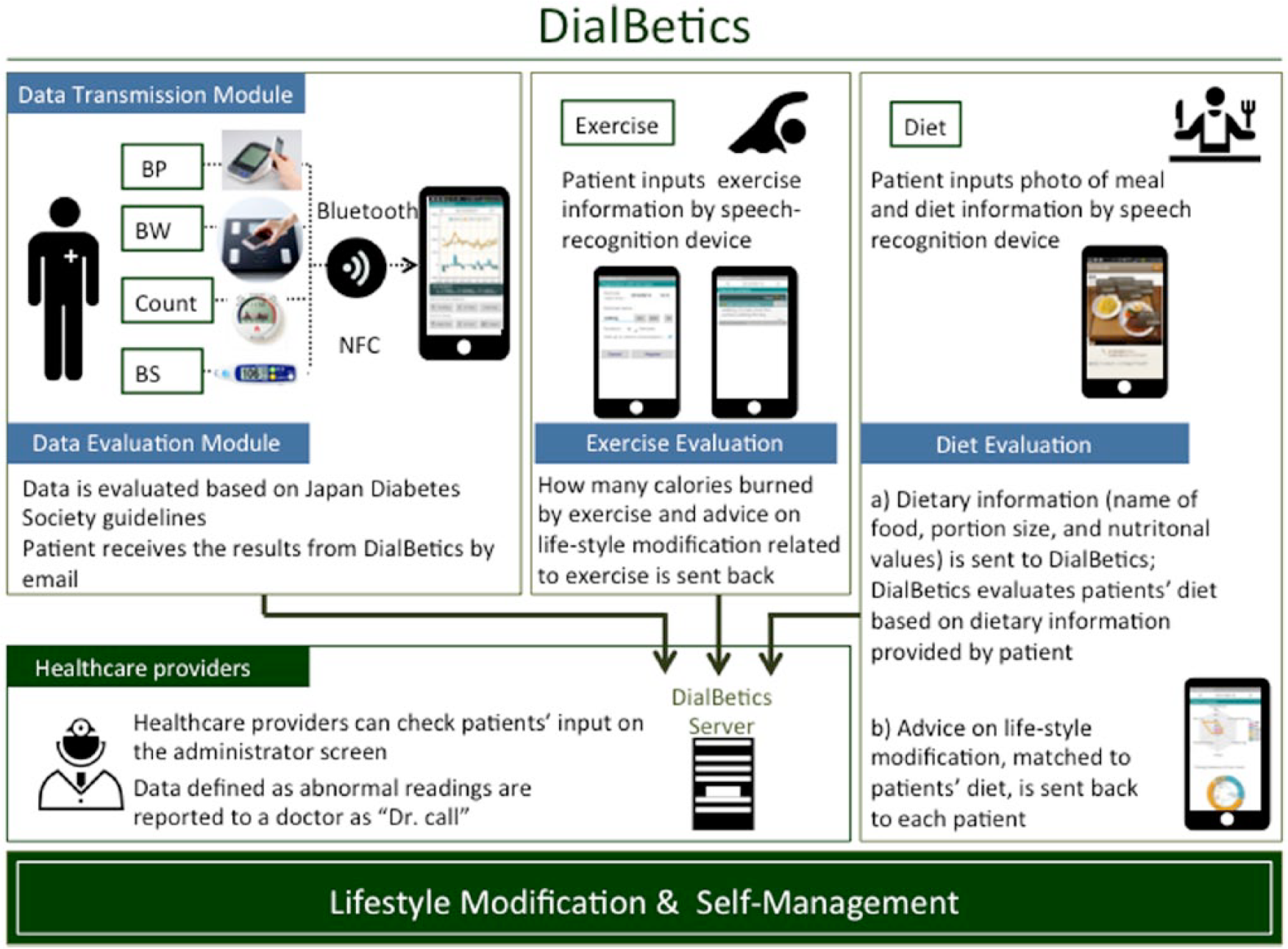
An overview of DialBetics.
Ten patients (2 females) who had been diagnosed with type 2 diabetes more than 5 years ago, and who injected insulin (15.8 ± 10.3 unit/day, 2.2 ± 1.3 times injection/day) were recruited at a university hospital for a 1-month pilot study. Age was 64.0 ± 7.8 years, body mass index (BMI) 26.3 ± 5.0 kg/m2, and HbA1c 7.1 ± 0.9%. To be eligible, patients had to be free of any severe complications—serum creatinine <1.5 mg/dl, no proliferative retinopathy—and had to be able to exercise. They gave written consent, and the Institutional Review Board approved the study. None of the patients experienced hypoglycemia or other adverse events during the study period, with no “Dr. call” readings. DialBetics was found safe for patients on insulin injections. Mean HbA1c (–0.26 ± 0.27%,
Since the purpose of this pilot study was simply to assess the safety and usability of DialBetics for patients on insulin, the 1-month study period was too short, and the 10-patient sample size too small, for evaluation of effects. We plan a longer, randomized study of more participants to validate the long-term effects of DialBetics on clinical outcomes for patients with type 2 diabetes under the Advanced Medical Technology Program of Japan’s Ministry of Health, Labor and Welfare for the system to be covered by health insurance.
Footnotes
Abbreviations
BMI, body mass index; HbA1c, hemoglobin A1c.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KW, K F, SK, and H K are members of the Department of Ubiquitous Health Informatics, which is engaged in a cooperative program between the University of Tokyo and NTT DOCOMO.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by NTT DOCOMO. This work was partially funded by a Grant-in-Aid for Young Scientists (B) 25860478 from the Japan Society for the Promotion of Science.
