Abstract
Background:
Hemoglobin A1c (HbA1c) is routinely used to monitor long-term glycemic control and for diagnosing diabetes mellitus. However, hemoglobin (Hb) gene variants/modifications can affect the accuracy of some methods. The potential effect of Hb variants on HbA1c measurements was investigated using a high-performance liquid chromatography (HPLC) method compared with an immunoturbimetric assay.
Methods:
Fasting plasma glucose (FPG) and HbA1c levels were measured in 42 371 blood samples. Samples producing abnormal chromatograms were further analyzed to characterize any Hb variants. Fructosamine levels were determined in place of HbA1c levels when unstable Hb variants were identified.
Results:
Abnormal HPLC chromatograms were obtained for 160 of 42 371 samples. In 26 samples HbS was identified and HbA1c results correlated with FPG. In the remaining 134 samples HbD, Hb Louisville, Hb Las Palmas, Hb N-Baltimore, or Hb Porto Alegre were identified and HbA1c did not correlate with FPG. These samples were retested using an immunoturbidimetric assay and the majority of results were accurate; only 3 (with the unstable Hb Louisville trait) gave aberrant HbA1c results.
Conclusions:
Hb variants can affect determination of HbA1c levels with some methods. Laboratories should be aware of Hb variants occurring locally and choose an appropriate HbA1c testing method.
Hemoglobin A1c (HbA1c) is a biochemical marker widely used in the management of patients with diabetes mellitus (DM) to monitor the adequacy of long-term glycemic management.1-3 Levels of HbA1c reflect average blood glucose levels over the previous 2 to 3 months 1 and correlate well with both microvascular and, to a lesser extent, macrovascular complications.1-3
Highly standardized HbA1c assays are available. A report from the International Expert Committee on the Role of HbA1c in Diagnosis of Diabetes in 2009, recommended stand-alone use of the HbA1c test to diagnose diabetes (with a threshold level of ≥6.5%) in addition to the current use for monitoring the effectiveness of glycemic control. 4 For diagnosis, HbA1c tests must be performed using a method that is certified by the National Glycohemoglobin Standardization Program (NGSP) and standardized to the Diabetes Control and Complications Trial. This study set the stage for establishing specific diabetes treatment goals using HbA1c as a marker of mean blood glucose. 1 However, it has been shown that the accuracy of some HbA1c tests can be adversely affected by the presence of Hb gene variants and hemoglobin (Hb) modifications. This can result in method-specific anomalies when measuring HbA1c leading to under-, over-, or nonestimation of the HbA1c fraction 5 and subsequently to inappropriate clinical management. It has been estimated that approximately 7% of the world’s population are heterozygous carriers of Hb disorders. 6 Of the estimated 366 million individuals with DM in 2011, around 26 million also had a Hb disorder, and these numbers are expected to double by 2030.7,8 As a result, the NGSP advises that laboratories should consider the likely prevalence of specific hemoglobinopathies in their population when selecting an HbA1c assay. 9 In addition, since the measurement of HbA1c is dependent on a normal erythrocyte life span, conditions with abnormal red cell turnover, such as anemia due to hemolysis and iron deficiency, may interfere with HbA1c measurements. In such cases the diagnosis of diabetes must be based solely on glucose criteria. 1
A variant Hb fraction may be an incidental finding during HbA1c analysis. In this study we investigated the potential effect of Hb variants on HbA1c measurements when using a high-performance liquid chromatography (HPLC) method compared with an immunoturbidimetric assay.
Methods
Samples and testing
Between November 2009 and January 2012, blood samples from 42 371 diabetic patients were analyzed, with samples for fasting plasma glucose (FPG) and HbA1c measurements drawn in the morning following overnight fasting. FPG levels were determined using the hexokinase method on the cobas® 6000 analyzer (Roche Diagnostics, Rotkreuz, Switzerland). HbA1c measurements were performed in ethylenediamine tetra-acetic acid (EDTA) anticoagulated blood samples using the ADAMS™M HA-8160 HPLC method (A Menarini Diagnostics, Florence, Italy; HPLC method 1), in diabetic mode and calibrated in accordance with the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Reference Measurement Procedure. Chromatograms from each sample were visually inspected for abnormal patterns suggesting the presence of Hb variants. As a comparison, HbA1c levels were also tested using the immunoturbidimetric Tina-quant® HbA1c Gen.2 and Gen.3 assays (Roche Diagnostics, Mannheim, Germany) on the cobas 6000 analyzer, also in accordance with IFCC standardization.
Samples that produced an abnormal chromatogram were further analyzed using the D10 Dual Program HPLC method (Bio-Rad, Hercules, CA, USA; HPLC method 2) in an extended program designed to characterize abnormal Hb variants. Samples containing Hb variants were also investigated at a hemoglobinopathy reference center in Madrid, Spain. Molecular characterization was performed by DNA genomic extraction (BioRobot EZ1; Qiagen, Hilden, Germany) and selective amplification of the Hb β-chain gene using an Applied Biosystems® 2720 thermal cycler (Life Technologies, Applied Biosystems, Foster City, CA, USA). The β-chain gene was sequenced using an ABI Prism® dRhodamine Terminator Cycle Sequencing Ready Reaction Kit (PE Applied Biosystems, Foster City, CA, USA) following the manufacturer’s instructions and analyzed using an ABI Prism 310 Genetic Analyzer (PE Applied Biosystems, Foster City, CA, USA).
Fructosamine levels were determined with a colorimetric test using nitriblue tetrazolium in alkaline solution (Biosystems S.A., Barcelona, Spain) when unstable Hb variants were present and HbA1c measurements were not possible.
Statistical Analyses
Statistical analysis was performed using SPSS 12.0 for Windows (Microsoft, Seattle, WA, USA). Values were expressed as mean ± standard deviation. Normality was tested using the Shapiro–Wilk test and the Student’s
Results
A normal chromatogram produced using HPLC method 1 shows a characteristic peak for HbA1c at 60 seconds (Figure 1). Abnormal chromatograms were obtained from 160 of 42 371 patient samples tested; 26 of these showed HbA1c results in accordance with the FPG results and HPLC method 2 testing showed that the patients were carriers of the HbS variant. For the remaining 134 patients, HbA1c levels were abnormally low (42 patients) or gave no result (92 patients). Molecular characterization of these samples identified 5 heterozygous Hb variants: HbD (27 patients), Hb Louisville (3 patients), Hb Las Palmas (98 patients), Hb N-Baltimore (4 patients), and Hb Porto Alegre (2 patients). The patients who presented with abnormally low HbA1c levels were found to be carriers of HbD (11 patients), Hb Louisville (3 patients), Hb Las Palmas (24 patients), or Hb N-Baltimore (4 patients). No HbA1c result was reported for 16 patients who were carriers of the HbD variant, 74 patients with Hb Las Palmas and 2 patients with Hb Porto Alegre.
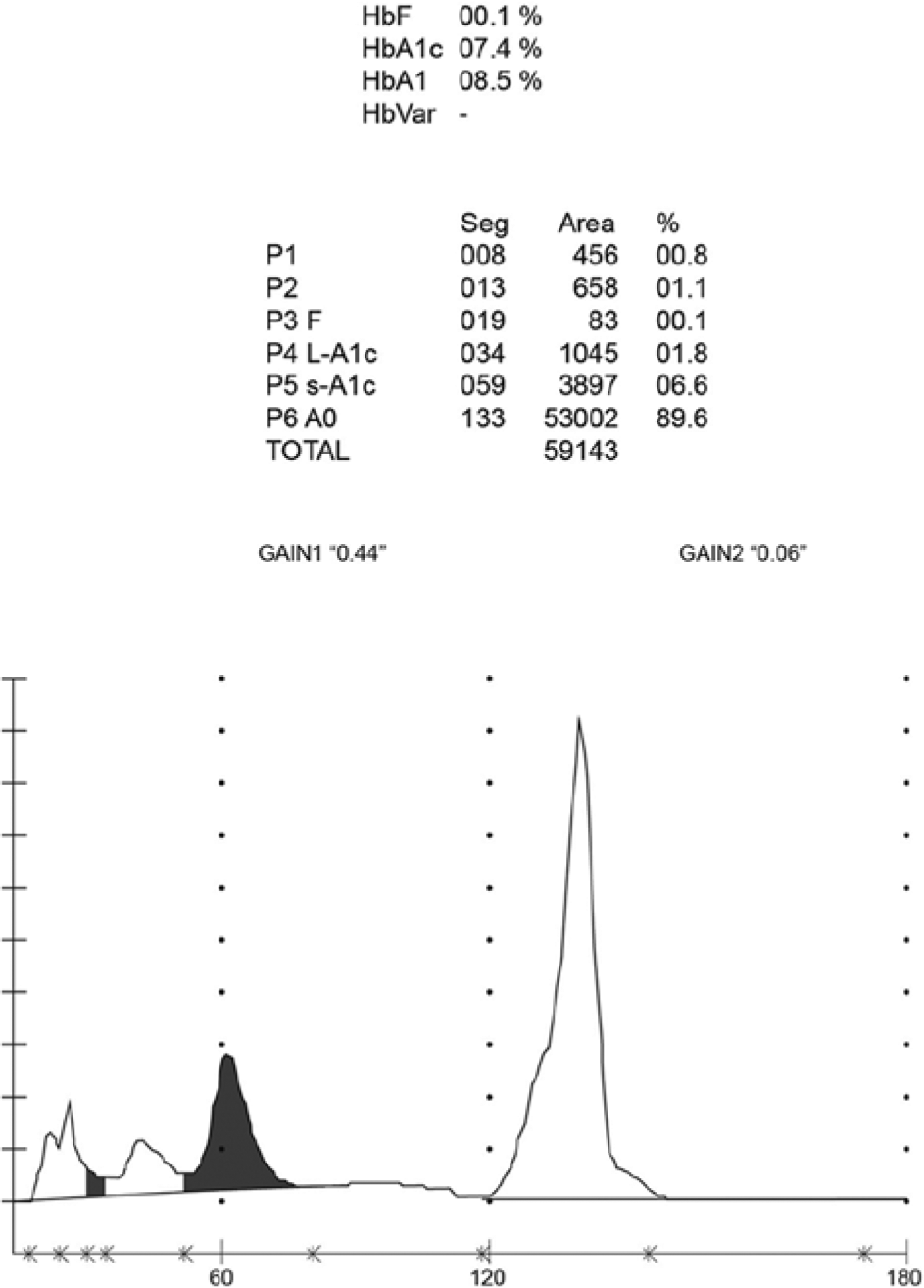
Normal chromatograms obtained using the HA-8160 HPLC method showing the characteristic HbA1c peak that elutes at 60 seconds.
Hemoglobin S
Figure 2A shows an example of the HPLC method 1 chromatogram obtained with the 26 samples from patients with HbS and contains an additional peak. This Hb variant has a valine for glutamic acid substitution at position 6 of the β-globin chain.
10
There was no statistically significant difference between the HbA1c levels reported by HPLC method 1 (5.90 ± 0.60%) and the immunoturbidimetric assay (5.91 ± 0.57%), and there was strong correlation between the results (
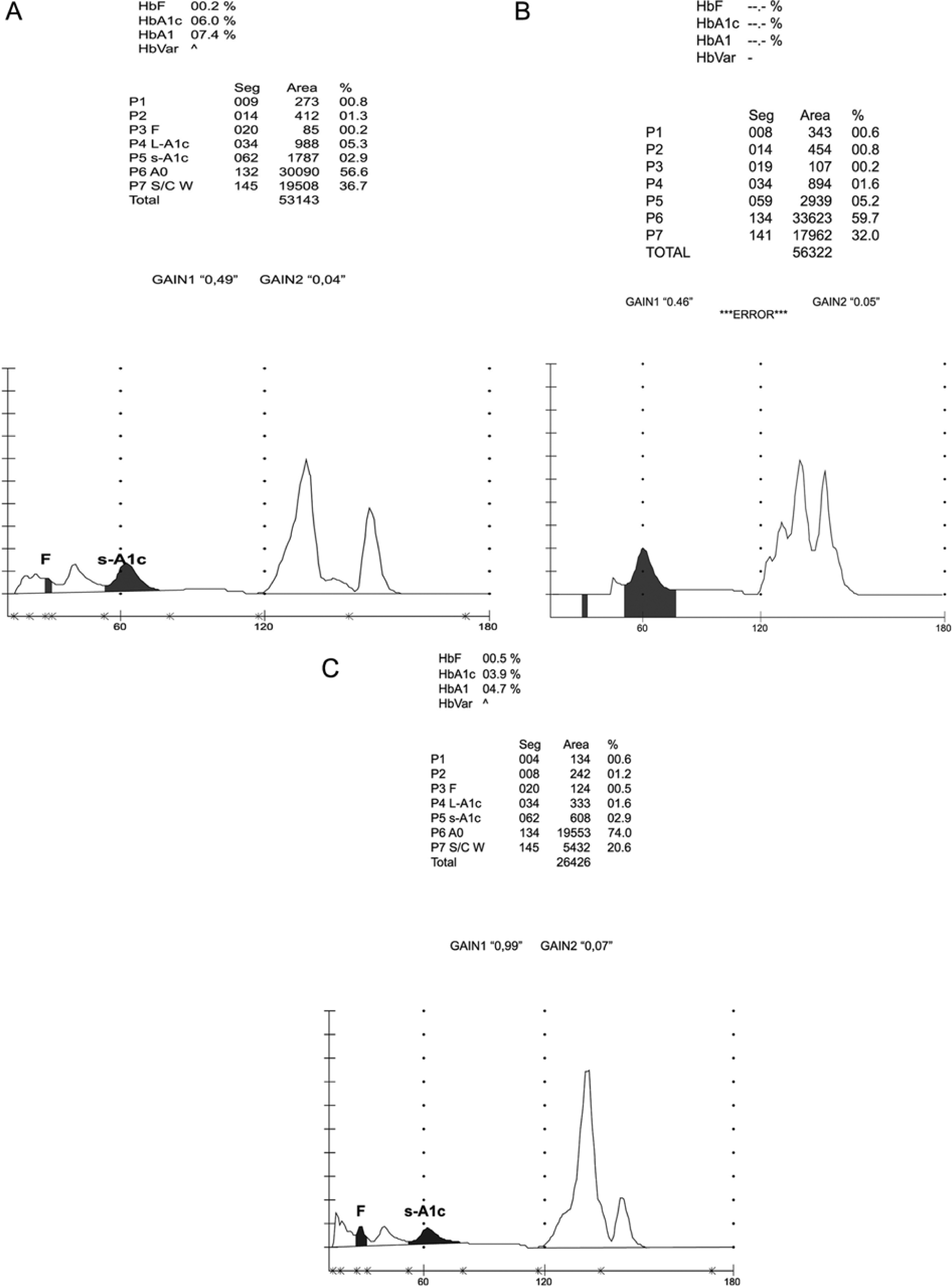
Chromatograms obtained using the HA-8160 HPLC method showing an extra peak due to the presence of (A) HbS, (B) HbD, (C) Hb Louisville.
Hemoglobin D
Twenty-seven samples produced abnormal chromatograms with the HPLC method 1 that included an extra peak in the S/C window that hindered the accurate quantification of HbA1c (Figure 2B). For 16 patients no result was given for HbA1c; the peaks were not labeled and the chromatogram indicated “abnormal separation.” For the remaining 11 patients, the results were abnormally low and not consistent with the corresponding FPG concentrations. These samples were retested using the immunoturbidimetric assay, which gave results consistent with the FPG concentrations for each patient. 11 The extra peak was labeled as HbD by HPLC method 2. DNA sequencing revealed the presence of HbD Punjab, also called HbD Los Angeles, which contains a substitution of glutamine for glutamic acid at position 121 of the β-globin chain. 10
Hemoglobin Louisville
Table 1 shows the results for 3 patients with a history of type 2 DM and consistent elevation of FPG but aberrantly low HbA1c values. The HPLC method 1 chromatogram showed an abnormal pattern, consisting of an extra peak in the S/C window (Figure 2C). All 3 patients had evidence of hemolytic anemia and their average total Hb levels were 9.41 g/dl, 10.9 g/dl, and 8.6 g/dl, respectively. In addition, fructosamine levels indicated the presence of hyperglycemia (reference range <285 μmol/l). Further investigations revealed the patients belonged to the same family and all 3 were heterozygous for Hb Louisville, an unstable Hb differing from HbA by the substitution of a phenylalanine residue for a leucine residue in position 42 of the β-globulin chain. 12 This causes instability in the Hb molecule with increased erythrocyte destruction, clinically manifesting as hemolytic anemia. 12
Results From the 3 Patients With Hb Louisville.
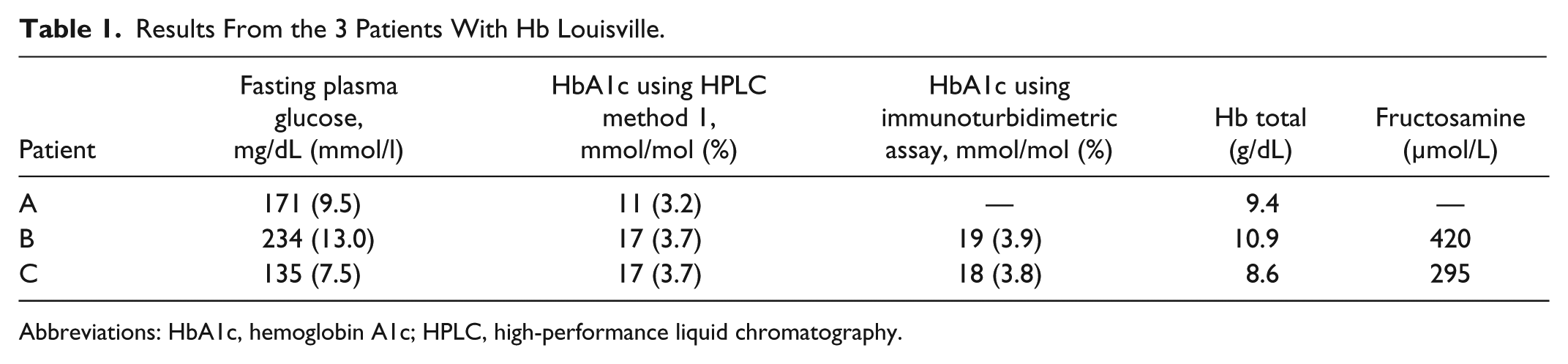
Abbreviations: HbA1c, hemoglobin A1c; HPLC, high-performance liquid chromatography.
Hemoglobin Las Palmas
Samples from 98 patients with DM showed anomalous chromatograms using HPLC method 1. No result was obtained for 74 samples (the “abnormal separation” warning was observed), while unexpectedly low HbA1c results were obtained for the remaining 24 (ranging from 12 mmol/mol [3.3%] to 25 mmol/mol [4.4%]) that were not consistent with the corresponding FPG results (Table 2). In all cases the chromatogram contained an additional small peak following the characteristic HbA1c peak (Figure 3A) that hindered the accurate quantification of HbA1c. However, results obtained after retesting the samples using the immunoturbidimetric assays did correspond with the FPG results. DNA sequencing of blood samples from 10 patients revealed a heterozygous point mutation at base 2 of codon 49 (CD8; Ser→Phe HBB:c.149C→G) which identified the presence of the Hb Las Palmas variant. 10
HbA1c Results in Patients with Hb Las Palmas (with HbA1c Results by HPLC Method 1), Hb N-Baltimore, and Hb Porto Alegre.
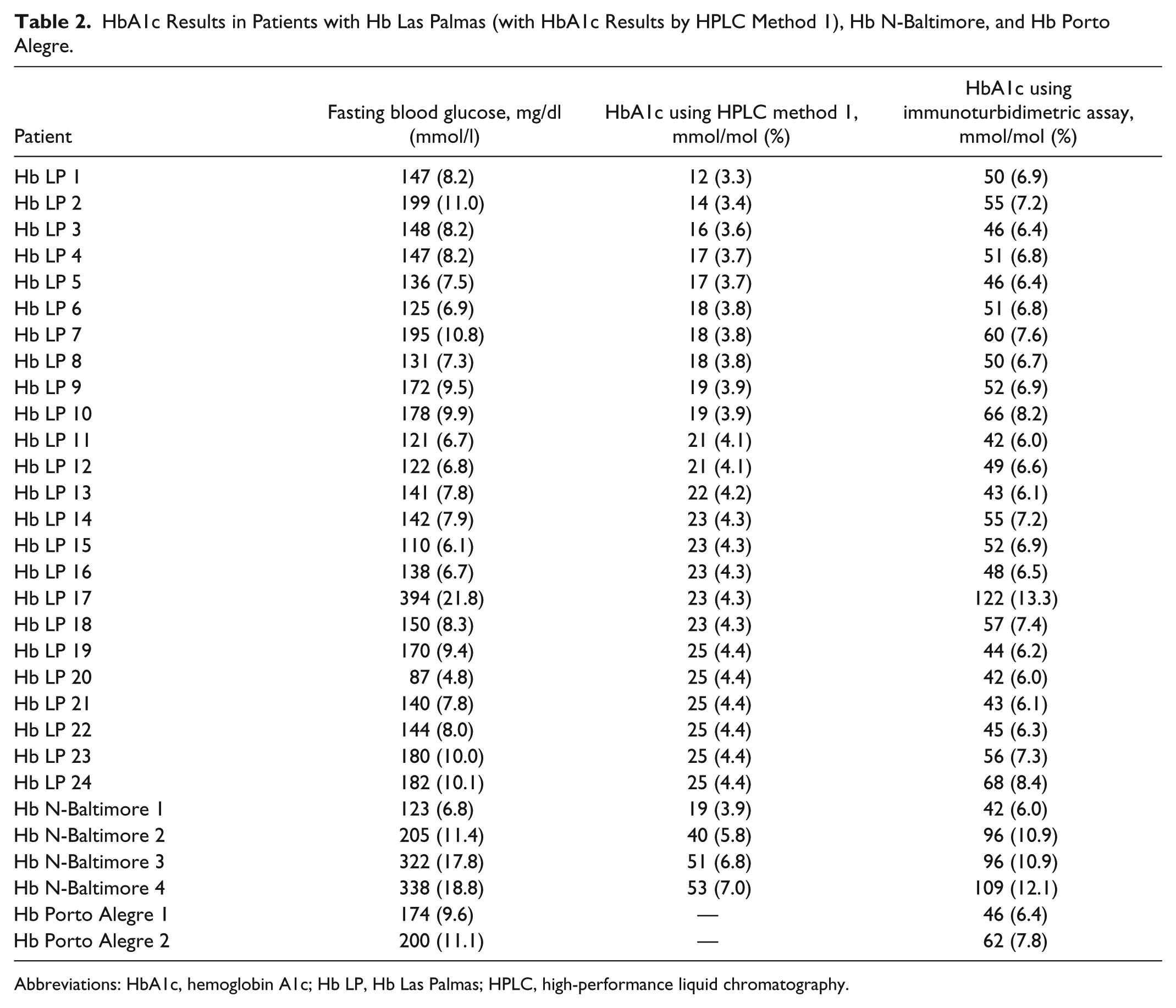
Abbreviations: HbA1c, hemoglobin A1c; Hb LP, Hb Las Palmas; HPLC, high-performance liquid chromatography.
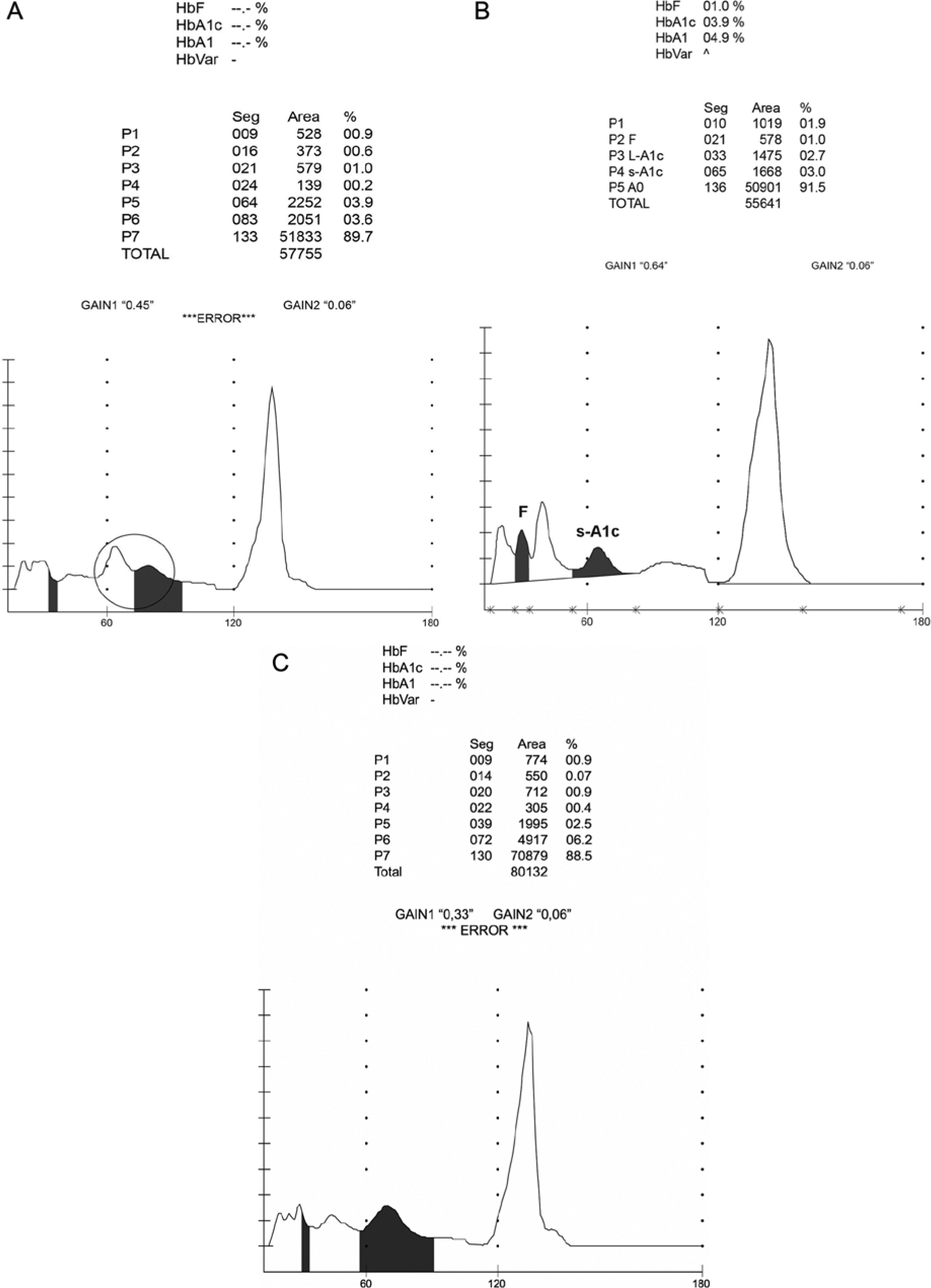
Chromatograms obtained using the HA-8160 HPLC method in the presence of (A) Hb Las Palmas, a small peak is present following the characteristic HbA1c peak (enclosed in circle); (B) Hb N-Baltimore, a small elevation is present following the characteristic HbA1c peak; (C) Hb Porto Alegre; the HbA1c peak is wider and delayed.
Hemoglobin N-Baltimore
Table 2 includes 4 patients with a history of type 2 DM who experienced consistently elevated FPG concentrations over the previous 6 months but had low HbA1c values. The HPLC method 1 chromatograms from the 4 patients’ blood samples revealed an abnormal pattern (Figure 3B): a small elevation following the characteristic HbA1c peak but no warning signal. HbA1c levels measured using the immunoturbidimetric assay corresponded with the FPG concentrations for each patient. Sequencing the Hb β-chain gene from all 4 patients revealed them to be heterozygous for the HbN-Baltimore variant, a structural variant that contains a substitution of lysine for glutamic acid at position 95. 10
Hemoglobin Porto Alegre
Two patients with a history of type 2 DM and warning signs of diabetic complications, but for whom an HbA1c measurement could not be obtained using HPLC method 1, are shown in Table 2. The HPLC chromatograms revealed an abnormal pattern with no extra peaks but a delay in the appearance and width of the HbA1c peak. In these 2 patients the HbA1c peak appeared at approximately 70 seconds, the “abnormal separation” warning was observed and no result was obtained for HbA1c (Figure 3C). HbA1c measurements using the immunoturbidimetric assay were consistent with FPG levels for each patient. Sequencing revealed heterozygosity for the Hb Porto Alegre variant in both patients, and also an intragenic polymorphism at codon 27 (GCC→GCT) that has been linked with the Hb Porto Alegre variant. 13 This structural Hb variant contains a substituted cysteine rather than a serine at position 9 of the β-globulin chain and causes polymerization of the Hb molecule.
In total HPLC method 1 gave no result for 57.5% of the screened patients, while 26.25% of the results were too low without a warning in the flag field. Only 16.25% of the samples identified as from HbS carriers presented correct HbA1c results. All of the 157 samples that were retested using the immunoturbidimetric assays corresponded with the FPG levels for each patient.
Discussion
HbA1c levels strongly correlate with blood glucose levels 14 and this is the most widely used marker for monitoring glycemic control. In view of the evidence that HbA1c levels are a strong predictor of new-onset DM, 15 the American Diabetes Association includes the use of stand-alone HbA1c testing for diagnosing DM and identifying pre-DM in its clinical practice guidelines. 16 The growing importance of HbA1c as a tool for diagnosis and monitoring of diabetes means that awareness of associated conditions that can affect laboratory HbA1c measurements is essential.
HbA1c can be measured using a variety of tests, each of which can be influenced by several factors leading to aberrant results.8,9,17 Hemoglobinopathies can affect HbA1c values in 3 ways: by influencing the binding of glucose to Hb, affecting chromatography peak measurements, and increasing the risk of hemolysis and hence decreasing the life span of red blood cells.17,18
HPLC methods for HbA1c quantification separate Hb species based on charge differences and are known to be susceptible to interference from Hb variants. Approximately 1175 Hb variants have been identified to date 10 with several known to coelute with HbA0. Failure to resolve the variant Hb components from HbA1c and/or HbA0 may cause problems with peak integration and lead to anomalous HbA1c results. Inaccurate HbA1c values can be observed when Hb variants, or their glycated derivatives, cannot be separated from HbA or HbA1c. The presence of relatively common Hb variants is often easily recognized when using HPLC methods by the presence of an additional peak in the chromatogram. However, further studies are required to characterize the Hb variant.
The presence of Hb variants has a lesser effect when immunoassay methods are used for HbA1c measurements. The immunoturbidimetric test from the current study uses monoclonal antibodies that recognize an epitope within the first 6 amino acids of the N-terminal of the Hb β-globin chain. Therefore, only the 17 known Hb variants with mutations in this susceptible region will affect HbA1c measurement.1,19 These Hb variants are extremely rare and are only found in individuals (eg, Hb Okayama) or families (eg, Hb Graz) so are unlikely to affect the majority of results obtained with this immunoturbidimetric test.
In a previous study, with 622 whole blood samples without hemoglobin variant, we found excellent correlation between the HPLC method 1 and inmunoturbidimetric assay for measuring HbA1c levels in whole blood.20-21
In the current study, 3 of the 6 Hb variants detected by analyzing HPLC method 1 chromatograms, HbS, HbD and Hb Louisville, presented with an extra peak in the S/C window.
HbS is the most widespread Hb variant, with a high frequency in West and North Africa, the Middle East and the Indian subcontinent. In a series of publications between 2000 and 2008, it was noted that HbS does not interfere with HPLC method 1 and immunoturbidimetric methods,22-24 an observation that this study has confirmed.
The presence of lower HbA1c levels measured by HPLC in patients carrying the HbD trait has been reported previously.25-27 In our study, no results were obtained for 59% of samples from patients carrying the HbD trait and abnormal results in the remaining 41% using HPLC method 1. However, presence of the HbD trait had no clinical effect on the results reported by the immunoturbidimetric assay, which were found to be in accordance with the patient’s clinical situation. 11
Chromatograms from patients carrying the Hb Louisville trait also contain an extra peak, but in this case the variant produces instability in the Hb molecule leading to increased erythrocyte destruction. Hb Louisville (42(CD1)Phe→Leu) heterozygotes suffer from mild anemia, jaundice and hemolytic crisis. 12 Because Hb Louisville is unstable HbA1c measurements are unreliable by any method, so fructosamine, a product of serum protein glycation, has been suggested as an alternate measure of glycemic control. 18 It is neither affected by disorders of red blood cells nor influenced by Hb variants, thus serving as an indicator of blood glucose concentrations over the previous 2 to 3 weeks. 28 In patients with DM who are Hb Louisville carriers, glycemic control should be monitored with serial glucose determinations or by fructosamine level. 29
In addition to Hb variants that produce an extra peak, we detected 3 other anomaly causing variants in HPLC method 1 chromatograms following the characteristic peak of the HbA1c that elutes at 60 seconds: Hb Las Palmas (β49 Ser→Phe; 10), Hb N-Baltimore (β95 Lys→Glu; 12) and Hb Porto Alegre (β9 Ser→Cys; 10). Hb Las Palmas was initially described in 1988 as a silent β-chain variant 30 and no new cases have been reported until now. In our 98 patients this variant hindered accurate quantification of HbA1c using HPLC method 1, with either no result (along with a warning of abnormal separation) or abnormally low results that were inconsistent with the FPG value for each patient. 31 Smith and Torbert initially described Hb N-Baltimore in 1958. 32 The 4 variant carriers in the current study had abnormal HPLC method 1 chromatograms, consisting of a small elevation following the characteristic HbA1c peak but no warning of abnormal separation. If the clinical management of these patients had depended solely on these results they may have received inappropriate treatment. 33 The Hb Porto Alegre variant, which is clinically asymptomatic, was discovered by Tondo et al in a large Brazilian family of Portuguese origin in 1963. 34 In the current study, the heterozygous presence of the Hb Porto Alegre variant in 2 patients with type 2 DM produces a glycated version of this variant that interferes with HPLC method 1 through abnormal separation of HbA1c, resulting in no reported HbA1c value. 35 In patients heterozygous for the Hb Las Palmas, 31 Hb N-Baltimore, 33 and Hb Porto Alegre variants, 35 the results of HbA1c determined by the immunoturbidimetric assay were consistent with the FPG concentration for each patient.
Conclusions
The results described highlight the importance of local Hb variant and hemoglobinopathy awareness and the implications for the testing method utilized. A list of known interfering factors for specific commercial assays is maintained on the NGSP website 9 and manufacturers should include these limitations in the instructions for use for each assay. Laboratories should use this information to select HbA1c testing methods that are appropriate for the region. 36 In addition, HbA1c results should always be assessed within the clinical context of the patient in case unexpected Hb variants are present. When inconsistencies are observed between a patient’s home blood glucose monitoring and laboratory-measured HbA1c levels, a falsely elevated or lowered HbA1c result should be considered. Any result that does not correlate with the clinical situation should also be investigated. 17 Conditions that shorten erythrocyte age (eg, hemolytic anemia) also falsely lower HbA1c test results, regardless of the assay method. 37 In these situations additional testing should be carried out using a test based on a different analytical method, such as an immunoturbidimetric assay, that can help to avoid the issues of Hb variant interference observed with HPLC-based methods as they employ highly specific antibodies that target a conserved region of the Hb β-globulin chain.
Footnotes
Abbreviations
DM, diabetes mellitus; EDTA, ethylenediamine tetra-acetic acid; FPG, fasting plasma glucose; Hb, hemoglobin; HbA1c, hemoglobin A1c; HPLC, high-performance liquid chromatography; IFCC, International Federation of Clinical Chemistry and Laboratory Medicine; NGSP, National Glycohemoglobin Standardization Program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: English language editing was provided by Elements Communications Ltd, Westerham, UK and supported by Roche Diagnostics International Ltd, Rotkreuz, Switzerland.
