Abstract

Diabetes is currently diagnosed by way of blood testing either with elevated glucose levels or elevated A1C levels. 1 Both tests are invasive which makes them inconvenient for many patients. Glucose testing may be subject to preanalytical or analytical errors in measurement. A1C testing may be affected by physiologic perturbations in red blood cell lifespan or by the presence of hemoglobinopathies, which can alter the apparent concentration of A1C so that it does not correspond to mean glycemic levels. A noninvasive diagnostic test for diabetes would be well received at many screening clinics. Such a noninvasive marker would be particularly useful if it could also serve as a marked of diabetic glycation-associated end organ damage.
In 1989 a team from Japan compared the degrees of nonenzymatically glycosylated proteins in the skin nails, hair, and hemoglobin of 51 subjects with diabetes and 20 controls subjects without diabetes.
2
Furosine is an intermediate of nonenzymatic glycation of free amino groups in proteins, a process known as advanced glycation end product (AGE) formation.
3
In this study, A1C levels correlated more closely to the furosine levels in nails (
In 2012 a team from Japan and China measured intermediate products of AGEs in fingernails of patients with diabetes using by ultra-performance liquid chromatography with electrospray ionization time-of-flight mass spectrometry. They noted a statistically greater concentration of one AGE intermediate compound 3-deoxyglucosone (3-DG) (but not two other AGE compounds) in the nails of 20 subjects with diabetes than in 20 controls without diabetes. They concluded that the proposed method provides a good mass accuracy and the trace detection of an AGE intermediate product in the human nail, and that this analytical technique could be a noninvasive technique to assist in the diagnosis and assessment of disease activity in diabetic patients. 4
Last year a team from the Republic of the Congo and Belgium reported that the measurement of glycation in fingernail proteins offers a new approach for actually detecting diabetes. 5 This year the same team also reported that a measurement of glycation in fingernail proteins is a marker of glycation-associated target organ damage. 6
They clipped fingernail pieces from 128 volunteers (approximately half of whom had diabetes) into 2-3 mm pieces. Fructosamine assays measure glycation of all proteins and a fructosamine assay was used to measure the glycation of nail proteins stratified according to whether the subject has known diabetic retinoathy or nephropathy. This amount of glycation was then compared with the age of the subject and also compared with the A1C level of the subject. The amount of glycation was proportionate to age and there was more glycation in the nails of diabetes patients with complications than diabetes patients without complications. The correlation of glycated nail protein values with A1C was glycated nail protein (micromole/gram of nail) = 0.058 × A1c (millimoles per liter) + 4.33;
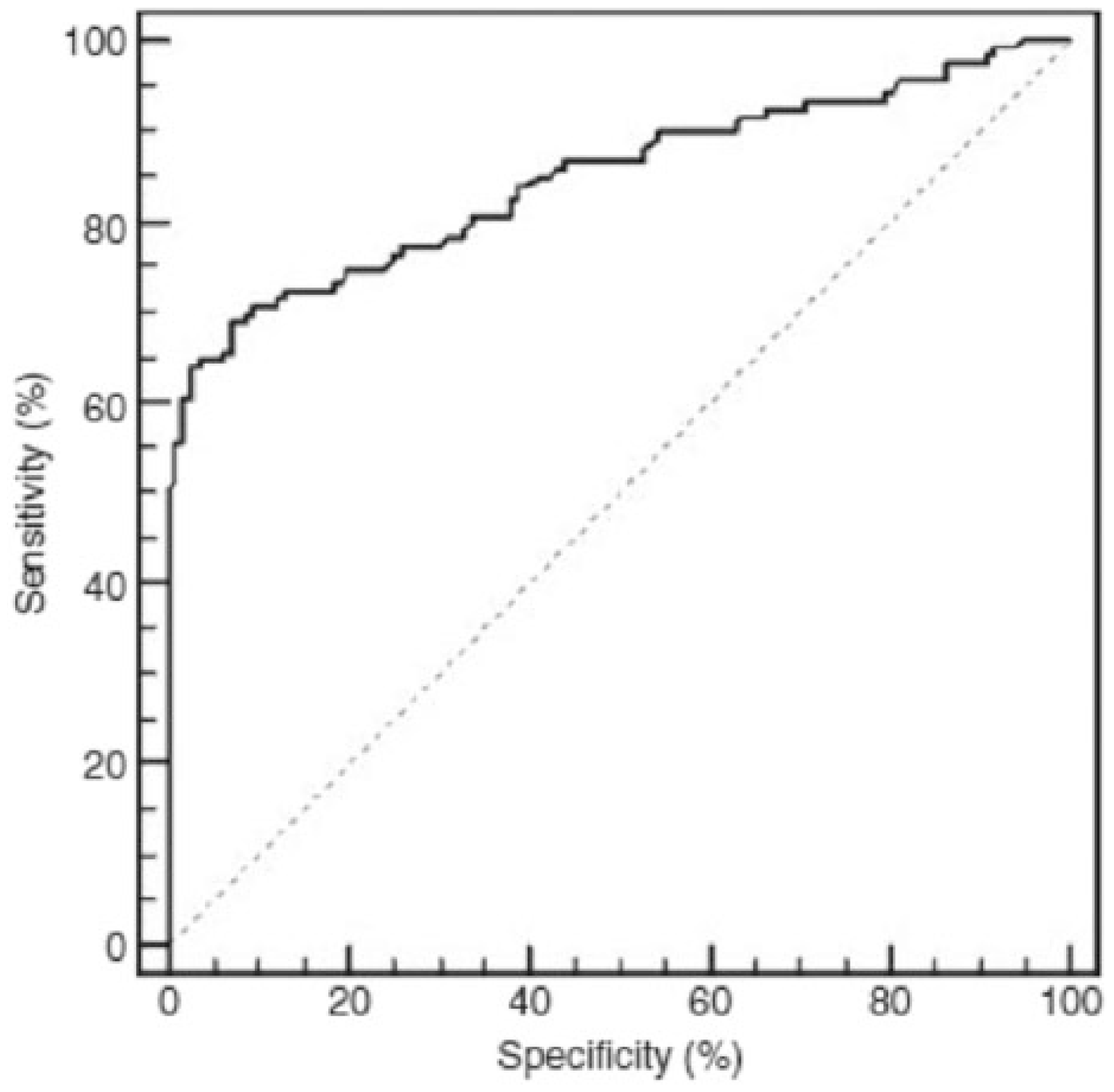
ROC curve of glycated nail proteins. Area under the curve (AUC) = 0.848, cutoff point = 3.14 μmol fructosamine/g nail; sensitivity = 68.9%; specificity = 93.1%). Reproduced with permission from
The investigators concluded that their test provided information about the period that the nail was exposed to extracellular fluid glucose. The increased glycation content with age was attributed to slower growth of nails with aging. There was poor correlation between absolute blood A1C levels and nail fructosamine levels. This discordance was attributed to physiological differences between processes raising levels of these two glycated analytes as well as to possible variations between patients in the glucose diffusion rates from blood vessels to nail beds. They concluded that this method could be considered to be an alternative test to assess long-term levels of mean glycemia and might provide useful information for management of diabetic complications where blood sample analysis is not possible. The investigators cautioned against harvesting toenails for diagnostic purposes because of the risk of causing a diabetic foot wound to develop from the clipping procedure. 5
This year the same group reported a study comparing the amount of glycation of fingernail proteins with the amount of glycation of eye lens proteins. They studied a cohort of 216 subjects (29% of whom had type 2 diabetes) who underwent cataract excisions and provided an eye lens protein and produced fingernail clilppings. 7
The eye lens proteins contained a greater concentration of glycated proteins in the subjects with diabetes mellitus type 2 patients (n = 63, mean = 3.80 ± 1.57 μmol/g eye lens) compared to the subjects without diabetes (n = 153, mean = 3.35 ± 1.34 μmol/g eye lens) (
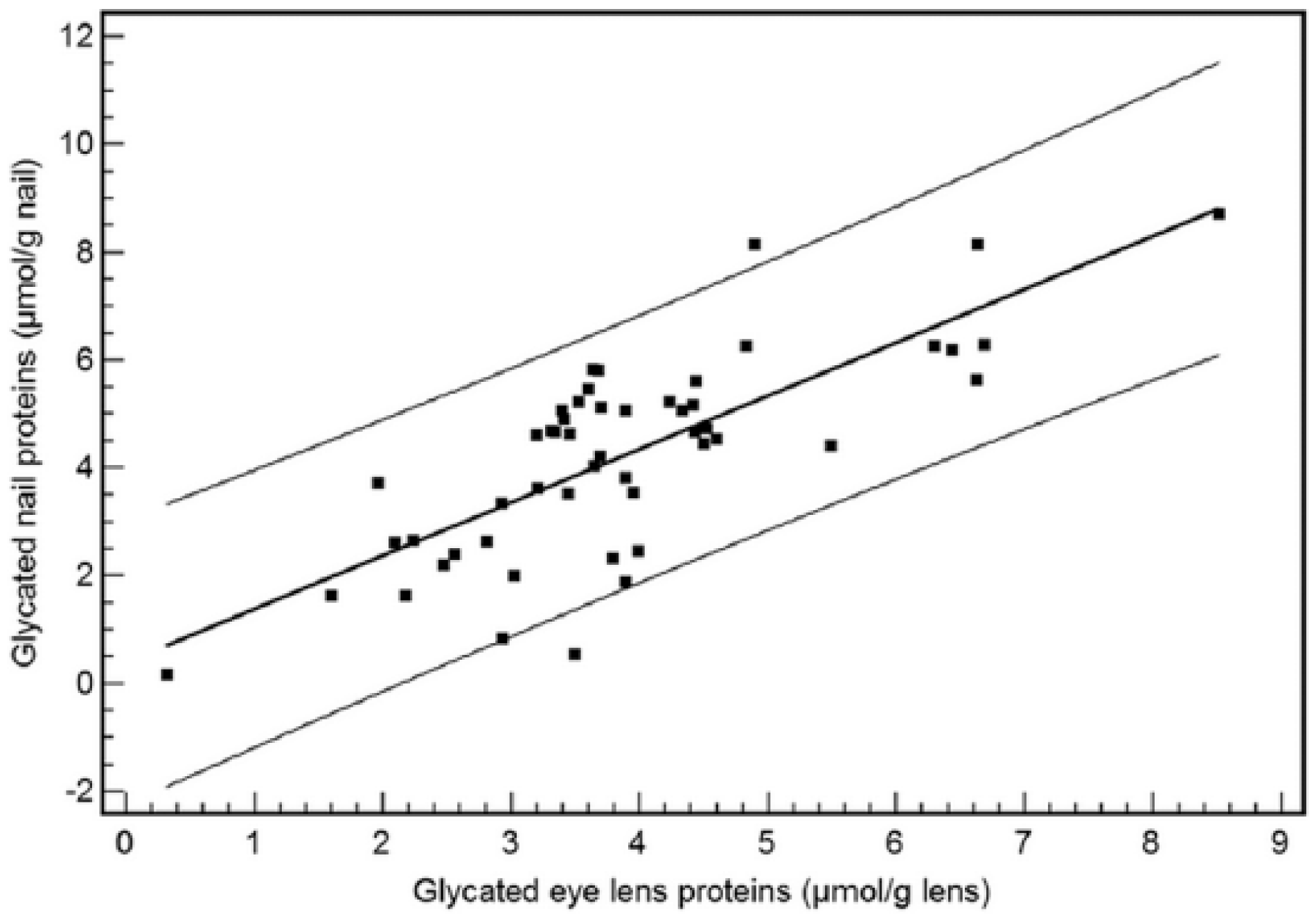
Correlation between glycated nail proteins and glycated eye lens proteins in the group of type 2 diabetes patients (n = 35). The equation of linear regression is y (glycated nail proteins) = 0.26 + 1.12 × (glycated eye lens proteins) (
A nail can take up to 6-9 months to be replaced. 7 This means that nail glycation fluctuations occur over a 6- to 9-month period. Eye lens proteins do not turn over (like nail proteins do) and are susceptible to lifelong glycation. 8 Therefore, AGE accumulation in the lens contributes to formation of cataracts. The correlation observed between glycated nail proteins and glycated eye lens proteins in this study supports the role of glycated nail proteins as a risk factor for predicting cataract disease and the conclusion that glycation of nail proteins can be regarded as a marker for diabetic glycation-associated target organ damage.
As new diagnostic tests for diabetes are being sought, a measurement of fingernail glycation could prove useful for this purpose. Several barriers must be overcome, however, for this method to gain traction. First, the assay in the two recent studies was not rigorously validated to ensure that the method is accurate, specific, and reproducible over the specified range that glycosylated protein will be analyzed. This validation process will provide evidence that the method does what it is supposed to do. At this time the method has low correlation of glycated nail protein with A1C. Therefore, for this assay to be adopted for screening for diabetes, it will need to demonstrate good correlation of glycated nail protein with some other measure of mean glycation besides A1C such as estimated average glucose, 9 mean glucose based on continuous glucose monitoring, 10 or estimated A1C. 11 Second, a screening test for diabetes must be fast and inexpensive. This reported method is labor-intensive, and no economic analysis was presented for the method in any of the articles describing this type of assay. Third, the assay method must be scalable to remain rapid and practical for large numbers of subjects, but no information has been provided on this aspect of the described potential mass screening program. Therefore, three important dimensions of this screening method (accuracy, cost, and scalability) have not been reported to date.
This fingernail test could avoid some of the problems that have been reported when the A1C level does not conform to mean glycation because of abnormalities of hemoglobin structure or lifespan. Because a fingernail test is noninvasive and can be performed in the nonfasting state, this type of measurement could lend itself to screening programs where there are barriers to drawing screening blood tests. Ultimate adoption of this screening method for glycated nail protein will require additional data to support the accuracy, economic value, and scalability of this assay.
Footnotes
Acknowledgements
I thank David Sacks, MD for his helpful comments about the technology described in this article.
Abbreviation
AGE, advanced glycation end product.
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK has served on advisory panels for Insuline, Lifecare, Novartis, Sanofi, Tempramed and Voluntis, has received research support from Eli Lilly, Halozyme, Janssen, and Novo Nordisk, and owns stocks/shares in Tempramed.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
