Abstract
This article discusses the future direction of insulin pump technology and its relationship to the software update process. A user needs analysis revealed that respondents wanted an insulin pump software update process to function much in the same way as smartphone updates. Users of insulin pumps have the same expectations as with other ubiquitous technology such as smartphones, tablets, and laptops. The development of a software update system within a regulated environment that meets the needs of insulin pump users by allowing optional software updates that provide access to pump improvements, feature additions, or access to algorithms that provide therapy-changing technologies is a new way forward for the management of a complicated disease that affects more than 450,000 people using insulin pumps in the United States.
Keywords
In the past, computer software was updated slowly with floppy discs that came in large colorful boxes with hard to understand instruction manuals. Today, modern computers and consumer electronics devices, such as smart phones and tablets, allow users to update their software in a matter of minutes. Remote software updates are so common that they are even used to update automobiles to provide drivers with the latest features. 1 In stark contrast, the process for updating modern insulin pumps, which are small computerized medical devices, has lagged behind. Typically, these pumps use software that remains unchanged for a minimum period of 4 years, per the normal warrantied life span covered by medical insurance payers. Pump users also feel frustration as they are locked into obsolete software from the moment they open the box.
The antiquated medical device paradigm used in the pump space is changing, partly due to the regulatory process inherent in new product development. The new Infusion Pump Improvement Initiative from the FDA and their requirements for human factors testing were launched in early 2010, and as a result the industry is slowly shifting to a user-centered approach to medical device development.2-5 This shift can also be attributed, in part, to a greater awareness of human factors engineering information in the insulin pump industry, which is being used to better understand users’ needs, help reduce harm, and improve quality of life.6-8 Human factors engineering information has become critically important to manufacturers of insulin pumps, and they are beginning to realize that users of insulin pumps have the same expectations from the products they are utilizing to manage their diabetes as they do from other forms of technology such as smartphones, tablets, and laptops. The diabetes industry is hoping to accelerate development and innovation to enable medical devices, such as insulin pumps, to function with the same user experience as consumer electronic devices. New data reported in this article suggest that there is a demand from people with diabetes for an optimized insulin pump software update experience.
The Future of Insulin Pumps
Seven medical device manufacturers have received clearance from the FDA to market their insulin pumps in the United States. Each manufacturer has taken a slightly different approach in providing a solution for those with insulin-requiring diabetes. Tandem Diabetes Care, which manufactures the t:slim® Insulin Pump, has taken a user-centered, forward-looking design approach to create a pump platform that allows for future remote software updates by its users through a direct USB connection to a computer. There are several benefits to this approach. First, it allows for a system in which end users can, if they wish, update the software of their device with new FDA-cleared software in the convenience of their home, prior to the end of their 4-year renewal period. Second, it decouples the software user interface from the hardware of the medical device and allows the system to incorporate new features and interactions as they become available to help users better manage their diabetes. Third, it reduces the burden on the health care provider, pump user, and pump manufacturer by eliminating the need to move through the insurance process to take advantage of updated features. Fourth, it enables today’s pumps to offer vastly different functionality once future technology is available. For example, artificial pancreas systems that seek to automate parts of the insulin delivery process based on continuous glucose sensor data will likely use the same pumping hardware that is available today, but will require updates to the software to incorporate advanced algorithms and control modules. Finally, as the diabetes ecosphere evolves on mobile devices, current software can be updated to allow pumps to communicate with health care applications, providing robust disease management data to patients, providers, and family members.
Human Factors Approach
Tandem Diabetes Care pioneered the use of human factors research as a key differentiator in medical device design. 6 The discipline optimizes the interactions between humans and technology by using evidence-based data to develop systems and products that minimize the risk of injury or illness to the people who use them. 7 Safety is always a central focus of this research, and as a result of this activity products are created that are safe, effective, intuitive, and highly usable. 8
The first step in the human factors research process is to understand the intended users involved in the process of updating insulin pump software. During this phase, detailed use scenarios are developed to aid designers, software developers, and the project team to better understand how the intended users will interact with the system. It is also important to investigate the intended use environments for the software update process. Understanding where users intend to update their software, whether at home or in clinic, is critical to inform the development of early prototypes. Uncovering insights about intended users, such as expectations and motivations for updating software and intended use environments, allows designers and developers to create an experience that is optimized for the majority of users. Fortunately for the diabetes community, the importance of understanding both health care provider and patient needs in the use of insulin pumps, continuous sensors, and artificial pancreas technology is beginning to make its way into published literature.9-11 Below is a summary of a user needs analysis conducted to understand the needs and limitations of those who would be updating insulin pump software.
User Needs
To better understand the user’s perceptions of software updates for insulin pumps, an electronic survey was sent to current t:slim pump users between November 6, 2014, and November 10, 2014. Respondents were invited by email to provide their feedback on the concept of remote pump software updates. The survey was comprised of a mixture of 43 qualitative and quantitative questions and was administered through the SurveyMonkey platform. Questions covered participant demographics, general questions about software updates (e.g., related to consumer electronics), and questions that specifically addressed the pump software update concept. There were 1,315 responses to the survey. The majority of respondents (56%) fell within the 25- to 54-year-old age range, with responses also coming from younger (≤17 years of age) and older (≥75 years of age) users. Respondents were 55% female and 45% male. The average time since diagnosis was 18.9 years, with a range of 1 to 66 years.
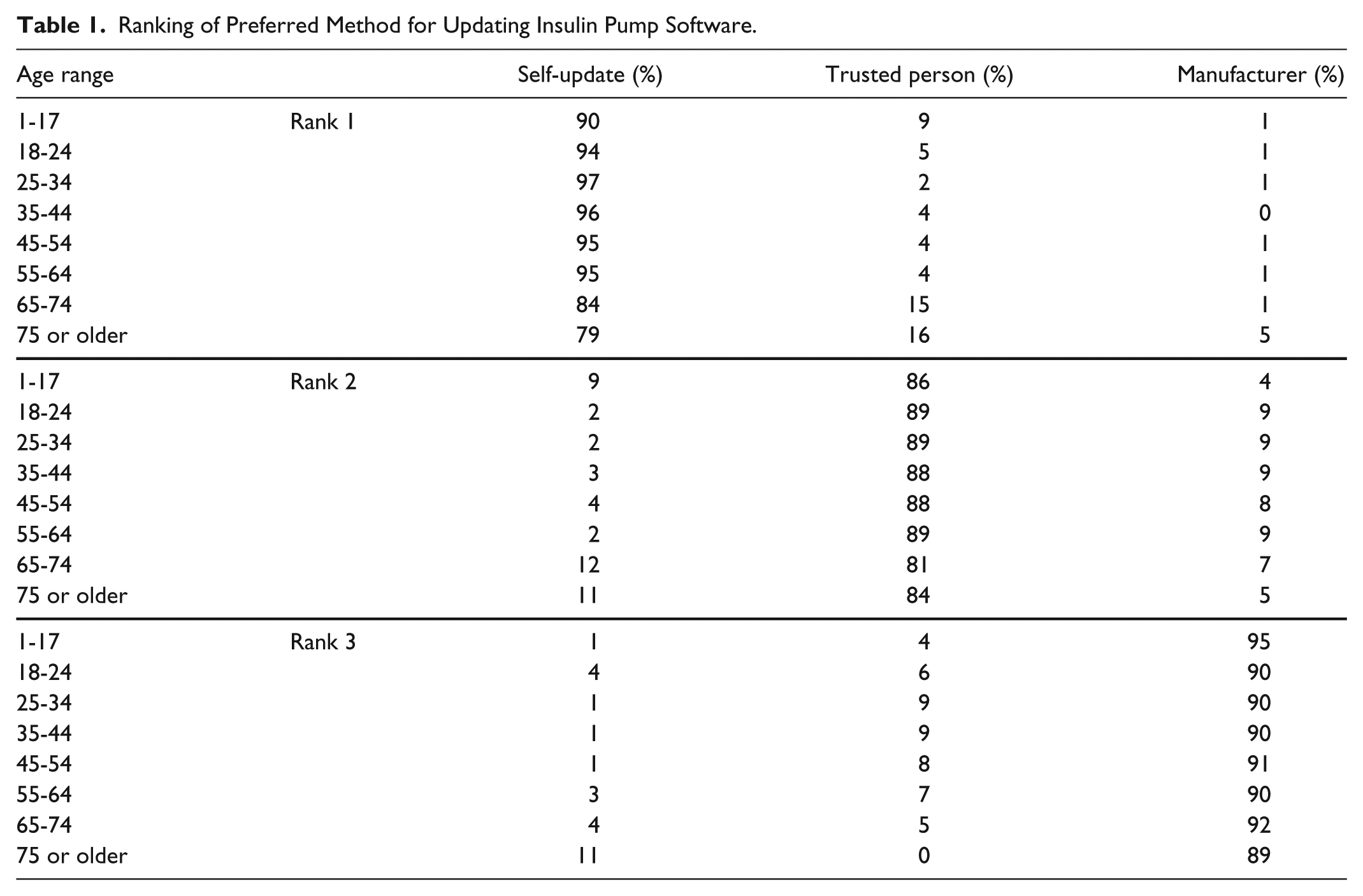
Survey respondents were asked to rate their comfort with technology (using a Likert-type scale from 1 =
Ranking of Preferred Method for Updating Insulin Pump Software.
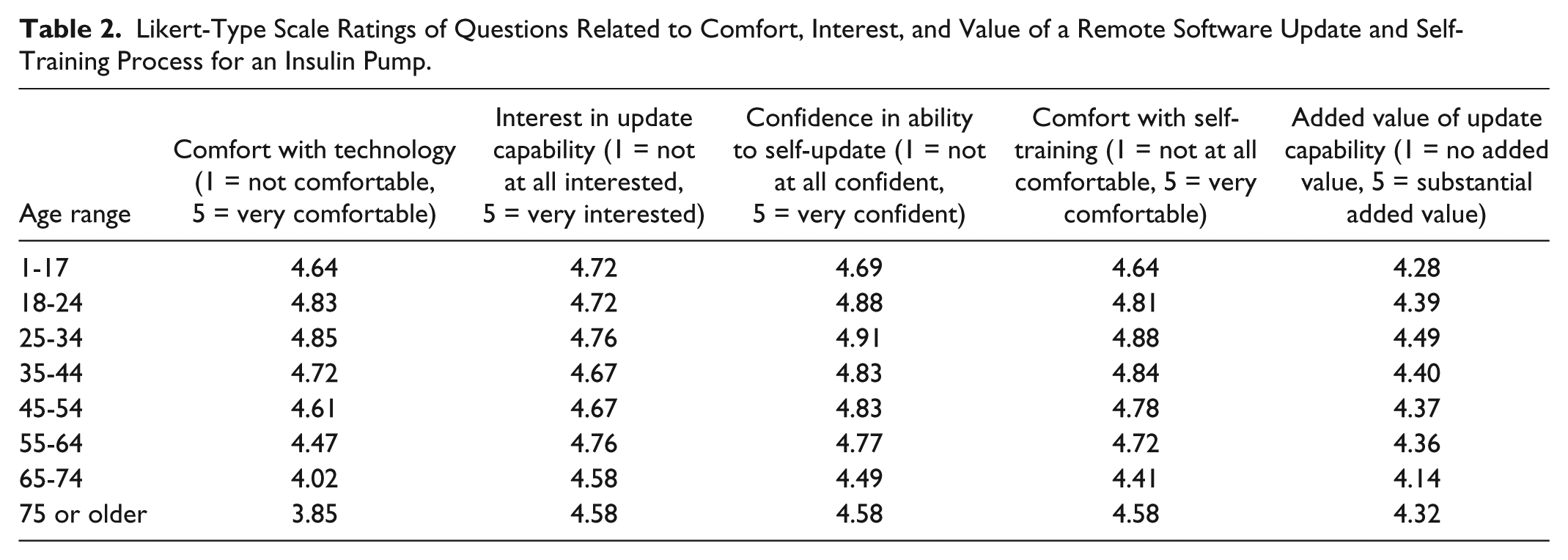
Likert-Type Scale Ratings of Questions Related to Comfort, Interest, and Value of a Remote Software Update and Self-Training Process for an Insulin Pump.
In terms of receiving notifications on available updates, almost 90% of respondents reported that they would prefer to receive an email with information that included (1) the amount of time required for the update process, (2) a description of the new features available in the update, (3) system requirements, (4) well-defined update instructions, and (5) contact information for customer service in case there is a problem. Respondents were also asked questions regarding the amount of time they felt an insulin pump software update should take. Of respondents, 63% indicated that they would expect the update to take 10 minutes or less.
Survey respondents were asked several questions regarding their comfort with self-training on new software features. Using a Likert-type scale of 1 (
When respondents were asked to rate their level of agreement on a 1
We recognize there are numerous limitations with this project. First, the study is not necessarily representative of wider views of people with diabetes who are not familiar with insulin pumps or may not be comfortable with modern technology. Also, the results are subject to self-selection bias as the survey was only sent to current t:slim pump users and those with access to internet or email. Finally, respondents were reacting to conceptual images and descriptions on a computer screen as opposed to a final commercial implementation of a remote software update system. Thus, further human factors research and testing is warranted to uncover real-world use cases necessary to optimize the final product configuration.
Conclusion
The current model in which software remains static on an insulin pump for up to 4 years is obsolete. There is a desire from users and from medical device manufactures to create an ecosystem of technology which allows insulin pumps to be updated with the newest software in a timely manner. In general, users want the update process to be uncomplicated and fast, and indicate a comfort level with self-training on new features via online training modules.
The analysis described in this article points to the smartphone update process as the preferred model for insulin pump software updates. If implemented correctly, the commercialization of this technology can have profound implications for how insulin pumps are used to manage a person’s diabetes. No longer will a person have to decide between purchasing a device today, or waiting for a new feature to become available. No longer will manufacturers have to engage in costly and inefficient pump return procedures to update the installed base. Manufacturing of the pump hardware becomes streamlined, customer and technical service becomes more consistent, advances in diabetes research (like the artificial pancreas) can be deployed much more quickly to the people who will benefit from it, and the process of continual product improvement becomes established because it’s simple and expected. The product life cycle is extended. The user experience is optimized. It becomes a new way forward.
Footnotes
Acknowledgements
The authors would like to thank Adam Noar, Bob Anacone, Eric Shearin, and Susan Morrison for their support and contributions to this effort.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NES, ETV, CAM, and MS are full-time employees at Tandem Diabetes Care, Inc, San Diego, CA. LJP, is a full-time employee at Senseonics, Germantown, MD.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Tandem Diabetes Care, Inc.
