Abstract
Background:
Using self-measurements of blood glucose (SMBG) is daily routine for patients with insulin-treated diabetes, however measuring ß-ketones in blood is not widespread. How the use of a combined device, which can measure both, is accepted in daily routine by patients and will lead to better glycemic control is not well studied.
Methods:
This multicenter, prospective, noninterventional study assessed the impact of routine use of the GlucoMen® LX Plus on patient acceptance, usage and glycemic control among insulin-treated patients with diabetes mellitus type 1 and type 2. A1c and self-reported frequency of SMBG were evaluated at baseline and also postprandial SMBG, ß-ketone measurements, and use of reminders after 3 and 6 months of use. A total of 631 patients, 254 type 1/350 type 2 (27 no type specified), with mean (SD) baseline A1c 8.5% (1.5), age 54.6 (15.6) years, and 47.3% female were studied.
Results:
Frequent use of SMBG at baseline led to a higher decrease in A1c at 6 month (V3): –0.3% if SMBG measured up to 1/day versus −0.9% in 4-6/day. Increase of SMBG frequency during the study showed also a negative correlation to A1c, 9.2% at V1 versus 7.6% at V3. Postprandial SMBG was done by 77.7% and ß-ketone measurements by 45.5% of all patients; the reminders were used by 33.4% and led to an increased frequency of SMBG at 6 months.
Conclusions:
A combined device for SMBG and ß-ketone measurements is well accepted by patients with insulin-treated diabetes and can lead through the avoidance or detection of ketoacidosis/increased frequency of SMBG and increased awareness of the patients to an improved glycemic outcome.
Keywords
Measuring blood glucose by the patients themselves (SMBG) is an integral part of the treatment of insulin-treated patients with type 1 (T1D) and type 2 diabetes mellitus (T2D). 1 The usefulness of SMBG and its influence on the level of glycemic control and patient empowerment are well established. 2 It has also been shown, that the daily frequency of SMBG and a structured manner of testing with fasting and postprandial (pp) measurements are essential for a positive outcome in glycemic control.3-4 This holds true for patients with T1D and insulin-treated T2D and for patients of all ages.5-7 To our knowledge there is only 1 study reporting on the use of SMBG testing reminder functions build into SMBG devices which shows that these help to overcome some of the challenges to perform pp-SMBG. 8 During the course of insulin therapy the risk of ketoacidosis always exist, the reasons of which can be different, for example, omission or wrong dose of insulin, higher carbohydrate intake, fever or other acute illnesses. However, measuring ketones, especially ß-ketones in blood, is not widespread used yet although often warranted. The seldom use of ketone testing in urine is mostly attributed to the inconvenience for the patients of the urine dipstick technique. It is also well known, that urine testing of ketones is only suboptimal because of the time lag in change of ketone concentrations in the urine compared to ß-ketones in blood as well as urine testing can give only semiquantitative results. 9 Although it is widely recommended by organizations, for example by the ADA, ß-ketone measurements in blood are not common in most countries. How often ß-ketones in blood are actually being measured in daily practice by patients themselves and what influence this has on glycemic control is not clear and not well studied.10-11 How the use of a combined device, which can measure both blood glucose and ß-ketones, is accepted in daily routine by patients and will lead to better glycemic control has not been studied at all to our knowledge.
To elucidate on this question we conducted a prospective, multicenter, noninterventional study of insulin-treated patients with T1D and T2D to assess the acceptance, usage, and resulting glycemic control (as seen in A1c) of a combined device, which can measure blood glucose as well as ß-ketones in blood as a point-of-care (POC) device used by patients.
Methods
The Gold Plus study was a multicenter, prospective, noninterventional study that assessed the acceptance and usage of the different features of the GlucoMen® LX Plus (A. Menarini Diagnostics S.r.l., Florence, Italy) on glycemic control among insulin-treated T1D and T2D diabetes patients on different therapy-schemes at 227 different diabetes clinics in Germany.
The GlucoMen® LX Plus is a combined blood glucose and ß-ketone meter for home glucose testing by patients with diabetes as well as for clinical use by health care professionals (A. Menarini Diagnostics S.r.l., Florence, Italy). The accuracy according DIN ISO 15197:2013 has been documented.
Patients were identified and recruited from the different investigators’ established patient populations (aged ≥ 18 years) diagnosed with T1D or T2D and being insulin-treated with multiple daily insulin doses (MDI) or insulin pump therapy (CSII). All patients who agreed to participate in the study signed an informed consent form before any study data were documented in printed case report forms (CRFs).
Data were collected on 3 visits of patients and health care professionals (HCPs) at the time of enrollment (visit 1) and visit 2 /visit 3 (after 2.4 ± 0.6 months/5.5 ± 0.8 months), with a written structured questionnaire regarding use (and recommended use) of the device and gylcemic control, including occurrence of hypoglycemia and ketoacidosis (at baseline only) and elevated ketone measurements.
A1c analyses were performed at the local laboratories of the sites and were documented by the local staff. Self reported data of the patients were documented in the CRFs, which included frequency and timing of SMBG, detection of hyperglycemic glucose values, and the use of the reminder and blood ketone measuring functions of the device.
Statistical Analysis
Mainly descriptive statistical analysis was performed for this noninterventional study. Quantitative data were analyzed by the statistical parameters valid N, missing N, mean, standard deviation (SD), and selected quantiles (1%, 25% [lower quartile], 50% [median], 75% [upper quartile], 99%). Qualitative and categorical variables were presented by means of (absolute and relative) frequency distributions. In addition, statistical tests were performed on an exploratory basis. A P value < .05 was considered to denote statistical significance. For quantitative variables the Wilcoxon–Mann–Whitney test (comparing 2 independent groups) and the Kruskal–Wallis test (comparing more than 2 independent groups) were applied, and for categorical variables the chi-squared test was used.
All analyses were performed using SAS® (version 9.2).
Results
In total 908 patients were enrolled into the study, 10 had to be excluded because they did not meet the inclusion criteria, of 631 all data were available for analysis (FAS), 254 with T1D and 350 with T2D, in 27 patients the diabetes type was not recorded. All patients were insulin-treated, 31 patients with T1D used CSII, 117 patients with T2D started with MDI therapy at V1 and 233 were experienced in MDI with these mean (SD) baseline (V1) characteristics: A1c 8.5% (1.5), age 54.6 (15.6) years, and 47.3% were female. There was no difference in A1c regarding the therapy form (different types of insulins or CSII) (P = .7726).
At V1 59% of the patients measured SMBG 4 or more times a day whereas at the end of the observation period (V3) 76.2% did so. During the study from V1 to V3 7.6% (n = 48) of the patients reduced the frequency of SMBG, 63.7% remained unchanged (n = 402) and 28.7% increased (n = 181) their frequency. Patients with frequent use of SMBG at V3 showed a higher decrease in A1c at 6 month (V3) from baseline: −0.8% in 3/day (−0.9%/SMBG 4-6/day; −0.8%/ SMBG >6/day), compared to only −0.3% if measured up to 1/day (P = .0723), although there was no additional benefit if frequency of SMBG was higher than 3/d as shown. Increase of SMBG frequency during the study showed also a negative correlation to A1c. Those patients who increased their frequency started out with a higher A1c at baseline (9.2%) but experienced a greater improvement in A1c of −1.5 compared to unchanged frequency (8.3%/−0.6) and decreased frequency (8.1%/–0.3) at V3 (P < .0001) (Table 1).
A1c at Baseline and at the End of Observation Period and Use of SMBG, Reminders, and ß-Ketone Measurements According to Good, Suboptimal, and Nonoptimal Glycemic Control at Baseline.
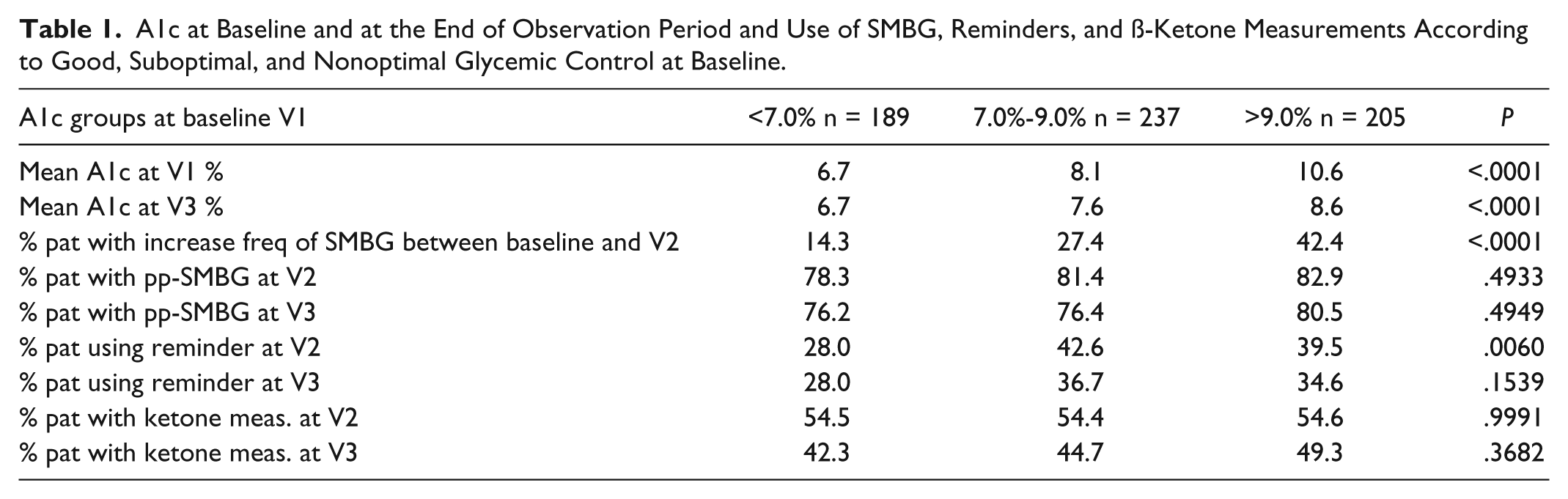
pp-SMBG were done by 77.7% of all patients, rising depending on the frequency of SMBG that was done (eg, SMBG 1/d than pp-SMBG 45.5% vs 93.5% if SMBG >6/d (P < .0001). There was no difference between the groups of patients who reported performing pp-SMBG related to A1c at V3 (76.2% with A1c <7.5% vs 76.4% with A1c 7.5-9.0% vs 80.5% in the group with A1c >9.5% (P = .4949). But there was a difference regarding the therapy form used: those patients using analogue insulins in MDI were measuring pp-SMBG more often (82.4% MDI analogues vs 76.5% CSII vs 66.7% MDI nonanalogues) (P = .0140).
The reminders for SMBG were used by 33.4% of the patients with no correlation to A1c at V3 (P = .1539) or therapy form (P = .3602). There was a difference in use of reminders for patients with different diabetes types (38.6% T1D vs 29.1% T2D, P = .015) and different age groups (age 18-30 years 40.4%, 31-60 years 39.3%, >60 years 25.4%, P = .003). The use did lead to an increased frequency of SMBG and pp-SMBG at 6 months. Those who increased the SMBG frequency during the study used more often the reminders before 3 months visit (V2) (but not at V3) compared to those who reduced the SMBG frequency or led it unchanged (45.9% vs 34.1%/31.3%, P = .0165). The usage of reminders correlated positively to the frequency of SMBG from 18.2% if SMBG 1/d to 52.2% if SMBG >6/d (P = .0002) (Table 2). Nearly half of the patients who did pp-SMBG used the reminder function (41.8%). More than twice as many patients, who measured ß-ketones also reported the use of reminders at V3 (47.0 vs 22.1%, P < .0001).
Change in Frequency of SMBG According to Use of Reminders, ß-Ketone Measurements, and pp-SMBG.
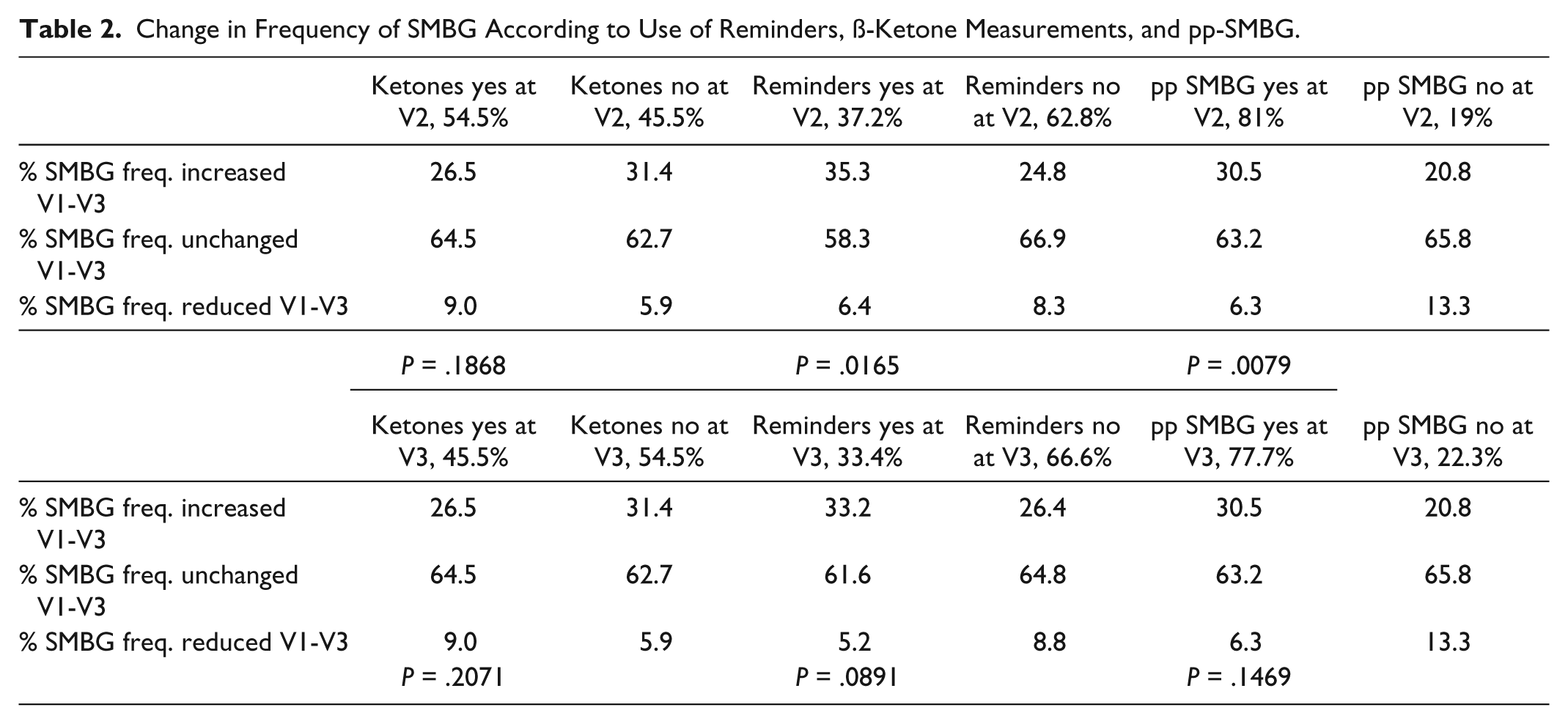
The ß-ketone measurements were done by 45.5% of the patients with a similar difference regarding diabetes type (T1D 64.6% vs T2D 30.9%, P < .0001) and age groups (age 18-30 years 63.8%, 31-60 years 49.7%, >60 years 32.5%, P < .0001) but no difference regarding to A1c at baseline or V3 (P = .3682) compared to those not measuring ß-ketones. Of those who had measured ß-ketone before V3 only 1.7% measured SMBG 1/d but 71.1% measured SMBG 3-6/d (P = .0007), but there was no statistical significant difference in changing frequency of SMBG during the observation period in this group (Table 2).
Patients using CSII performed ß-ketones measurements nearly twice as often than all other patient groups (82.4%, P < .0001).
Measuring pp-SMBG or ß-ketones by itself did not result in a more favorable A1c at 6 months with 7.7% versus 7.6%, P = .1976 and 7.7% vs 7.6%, P = .3168, respectively.
Looking at the results regarding A1c, SMBG, use of reminders and measuring ß-ketones separated by A1c at baseline in 3 groups of patients with good, medium, and unfavorable metabolic results refer to Table 1.
Discussion
We investigated the impact of the use of a combined device for SMBG and ß-ketone measurements by patients with insulin-treated diabetes in daily routine care. The study population was a mixture of different patients with insulin-treated diabetes, both T1D and T2D, treated with MDI (newly or experienced) or CSII over a great age range. All were treated in diabetes clinics in daily routine without an intervention protocol. Of the 631 patients of the FAS nearly three-quarters measured SMBG 4 or more times a day at baseline (59%) and at the end of the observation period (76.2%). A higher frequency of SMBG was associated with a better glycemic control expressed in A1c as it has already been shown in different studies and for various age groups before.6,7,12 In our study, patients who increased their testing frequency during the observational period improved their A1c, independent of their baseline frequency and their baseline A1c. This increase of frequency happened already during the first 3 months and stayed nearly stable afterward, suggesting that the patients experienced an advantage of frequent testing already at the beginning of the observational period.
There are always different influences, which contribute to the frequency of SMBG and the structured use of these (pp measurements, paired testing), for example, type of treatment and types of insulin used (risk of hypoglycemia). Also an increase of SMBG frequency could result from a change to insulin therapy after oral medication before. Another reason can be the impact of reminders to perform SMBG as shown here. The difference of frequency of pp-SMBG could be explained by the fact, that patients with analogue insulins (in MDI or CSII) are being taught the risk of possible hypoglycemia due to a faster kinetic and therefore are more akin of pp-SMBG. Reminders of SMBG measurements were used more often in the group of patients with suboptimal A1c at V1 and led to an increase in frequency of SMBG. Bergenstal et al demonstrated in their study with a SMBG device that also had as an advanced feature reminders for glucose testing that the patients increased their SMBG frequency and the use of pp-glucose measurements. 8 This shows that the use of reminders can help patients to optimize their treatment regime. One has to admit though, that especially younger patients and patients with T1D are more keen to use reminders, as shown in our study. This could be explained by the assumption that younger patients are aware of their problem of forgetting to measure SMBG because of time restraints and that patients with T1D generally perform SMBG more often than patients with T2D.
The need of ß-ketone measurements, or any ketone measurements, is well proven, but the acceptance varies between patients. 13 Using ß-ketone measurements with a blood test strip is by nature of the technique much more convenient and the results more timely and correctly available, which makes a response to a positive result more meaningful to the patients and leads to a better outcome.9,14,15 We have already reported, that patients accepted the advice from their HCP to measure ß-ketones if these would feel it to be appropriate, as was in 74% of the patients. 16 The ß-ketone measurements were used mostly with high blood glucose values (ca. 60%), when the device gave a ketone reminder (50%) or with ketoacidosis symptoms (15%). This demonstrated a high acceptance of ß-ketone measurements in daily routine but also shows together with the results presented here how often patients experience situations when ß-ketone measurements are warranted and they actually do so. Again it was shown here that younger patients and patients with T1D performed more often ß-ketone measurements. This could be explained by the fact that patients with T1D often experience more glycemic variability than patients with T2D. For older patients the possibility of checking their ketones while measuring SMBG should be encouraged to make it easier for them compared to urine measurements of ketones. Therefore devices to measure ß-ketones should be made available to patients with diabetes using insulin.
Several limitations are noteworthy. First, this study was designed as a post marketing survey in a descriptive way and not as a randomized trial and consequently there was no control group that used a device without option of beta ketone measurement. Therefore some question could not be answered. Data were self reported or evaluated by examining the logbook or analyzing downloaded data by the physicians (except A1c). The number of patients of whom the data were downloaded by the physicians was not separately analyzed. There was no clear definition for high BG values or ketoacidosis and the frequency of these were not assessed, which would have required ß-ketone testing. Only the actual use of ß-ketone measurements were documented in a qualitative way, not quantitative. The frequency of hypoglycemia was not being recorded except at baseline in a qualitative way. HbA1c values and references had not been harmonized according to DCCT standards. Furthermore duration of diabetes had not been asked in a definite way.
Conclusions
The use of a combined device for SMBG and ß-ketone measurements by patients with insulin-treated diabetes in daily routine care is feasible, easy to use and well accepted by patients with insulin-treated diabetes. The measurements of ß-ketones in blood or pp SBMG by itself does not improve glycemic outcome as determined by A1c but only together with an increased frequency of daily SMBG. Randomized trials with better defined different patient groups and more detailed documentation of SMBG and usage of ß-ketone measurements and reminders as well as the results of such measurements would help to better understand the impact of the use of a combined device for SMBG and ß-ketone measurements in daily routine in a diabetes population.
Footnotes
Acknowledgements
We wish to thank the participating patients and diabetes clinics with their staff for their assistance and commitment to this research effort.
Abbreviations
ADA, American Diabetes Association; CRF, case report form; CSII, continuous subcutaneus insulin infusion; HCP, health care professional; MDI, multiple daily insulin doses; POC, point-of-care; pp-SMBG, postprandial self-measurement blood glucose; SMBG, self-measurement blood glucose; T1D, type 1 diabetes mellitus; T2D, type 2 diabetes mellitus; V1, visit 1; V2, visit 2; V3, visit 3.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RZ has served on advisory boards for Animas and Roche Diagnostics, received honoraria from Animas, Menarini, and Roche Diagnostics, and received research funding from Roche Diagnostics. RE has worked as a consultant for A. Menarini Diagnostics, Germany. BS is as medical advisor employee of A. Menarini Diagnostics Germany.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study was provided by A. Menarini Diagnostics Germany, Division of Berlin-Chemie AG, Berlin, Germany.
