Abstract

For distribution in the European Union, systems for self-monitoring of blood glucose (SMBG) require a Conformité Européene (CE) mark. Proof of conformity with the international standard ISO 15197, which describes accuracy criteria for SMBG systems, is often performed by manufacturers to obtain a CE mark for their system. A new version of this standard with tighter accuracy criteria was published in 2013 1 and will replace ISO 15197:2003 2 after a transition period of 3 years.
In this investigation, a postmarketing evaluation of 2 CE-marked SMBG systems (TRUEyou [system 1] and TRUEyou mini [system 2]; Nipro Diagnostics Inc, Ft. Lauderdale, FL, USA) was performed following study procedures of ISO 15197:2013. Both systems are available on the European market and were procured by the manufacturer. The evaluation was performed in August 2014 at the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany in compliance with the German Medical Devices Act. The Ethics Committee and the Federal Institute for Drugs and Medical Devices approved the study.
Blood glucose (BG) concentrations of capillary blood samples from 100 different subjects were measured in duplicates with 3 reagent system lots of both systems and these concentrations were compared to values obtained with a glucose oxidase laboratory method (YSI 2300 STAT Plus™ glucose analyzer, YSI Incorporated, Yellow Springs, OH, USA). Regular internal and external quality measures were performed to confirm trueness and precision of the comparison method. The proper function of test systems was ensured by performing control measurements according to the manufacturer’s labeling. The number of values within the accuracy limits of ISO 15197:2013 and ISO 15197:2003 and the relative bias according to Bland and Altman 3 were calculated.
Both systems fulfilled with the 3 tested reagent system lots the accuracy criteria of ISO 15197:2013 with at least 95% of the measured values falling within the stipulated limits (Table 1). For both system 1 and system 2, 96.5% to 98% of the values obtained were within ±15 mg/dl of the comparison measurement for BG concentrations <100 mg/dl or ±15% for BG concentrations ≥100 mg/dl. Both systems showed 100% of the results within zone A of the consensus error grid. Relative bias ranged from −0.6% to 2.7% for system 1, and from 1.1% to 4.3% for system 2.
Numbers and Percentages of Results Within System Accuracy Limits of ISO 15197:2003 and ISO 15197:2013 and Relative Bias According to Bland and Altman.
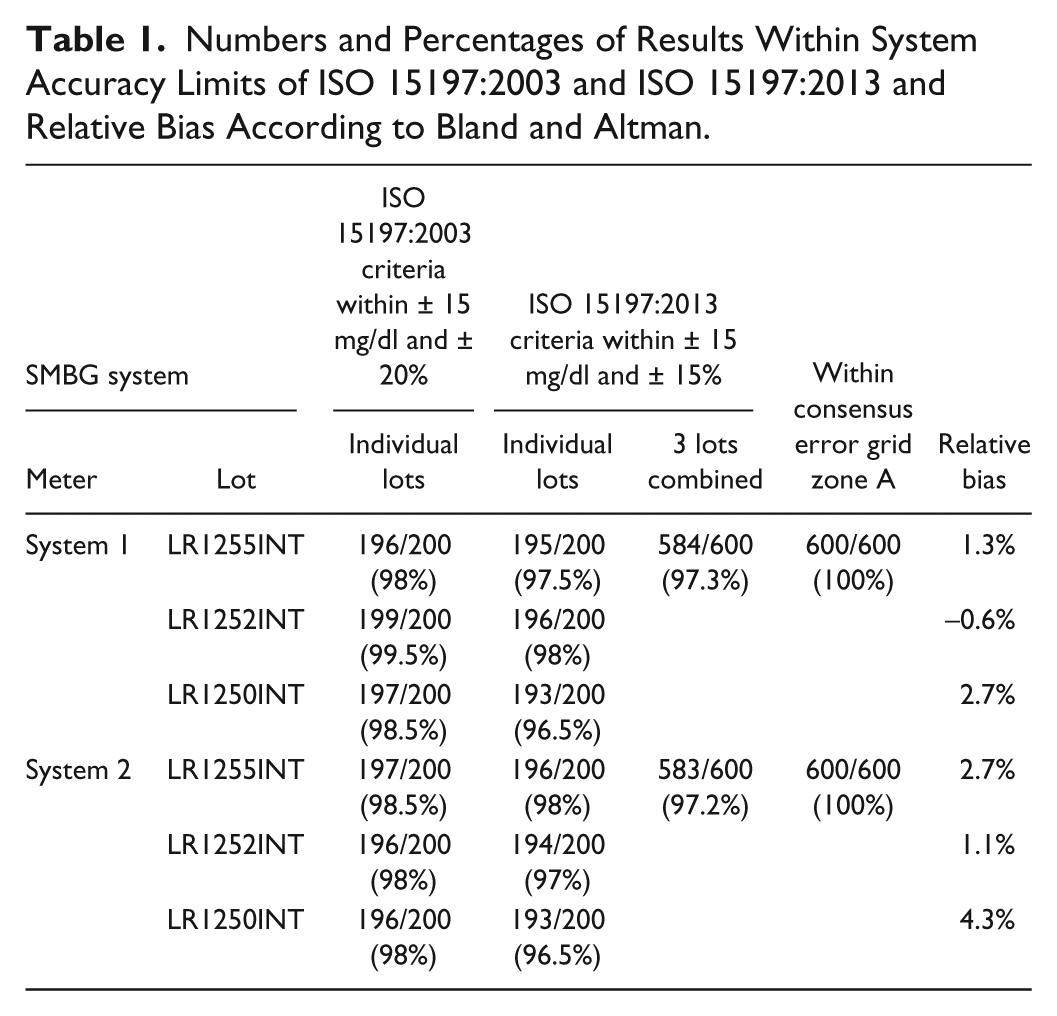
Applying criteria of ISO 15197:2003 (±15 mg/dl / ±20% for BG concentrations <75 mg/dl / ≥75 mg/dl), between 98% and 99.5% of BG values from system 1 and between 98% and 98.5% of BG values from system 2 fulfilled the requirements.
The tested reagent system lots of the 2 BG monitoring systems were shown to meet accuracy demands of ISO 15197:2003 after market release and fulfill the more stringent criteria of the new ISO 15197:2013.
Footnotes
Acknowledgements
The manuscript was prepared by the authors. Nipro Diagnostics, Inc was permitted to review and comment on the manuscript, but the final decision on the content was exclusively retained by the authors.
Abbreviations
BG, blood glucose; CE, Conformité Européene; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm (IDT), Ulm, Germany. GF is general manager of the IDT, which carries out studies evaluating BG meters and medical devices for diabetes therapy on behalf of various companies. GF/IDT received speakers’ honoraria or consulting fees from Abbott, Bayer, Berlin-Chemie, Becton-Dickinson, Dexcom, Menarini Diagnostics, Novo Nordisk, Roche Diagnostics, Sanofi, and Ypsomed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Nipro Diagnostics, Inc, Ft. Lauderdale, USA.
