Abstract
Keywords
Intensive glycemic control in type 1 diabetes has been shown to reduce the incidence and progression of microvascular and macrovascular complications.1,2 One validated way to improve glycemic control is the use of insulin pumps and/or continuous glucose monitors (CGMs).3-5 However, many patients do not wear or manage the devices optimally6-9 and clinical penetration is far from complete, even in the top clinical centers. 10 The limited and/or suboptimal use of diabetes technology can be traced, in part, to the “hassle factor,” the substantial degree of time, effort, patience, and appropriate decision-making that are required to monitor, operate, and maintain the devices. Thus, systems that automatically modify insulin delivery based on glucose data could facilitate more effective glycemic management, better quality of life, and wider use of diabetes technologies. Such a partially or fully closed-loop system, often referred to as an artificial pancreas (AP), could also lower the risk of acute hypoglycemia by reducing insulin delivery based on the prediction of the control algorithm. Developing an AP has been identified as a priority by both patient advocacy organizations 11 and federal health agencies. 12 However, for an automated system to improve on the safety and efficacy of current diabetes management, several difficulties must be overcome.
One of the biggest challenges for AP systems is meal time (prandial) glucose control. If the AP system delivers insulin only after the CGM values have started to rise (eg, closed-loop control), then the postprandial glucose excursion will not actually be “covered” by any insulin action for approximately an hour after the start of the meal, due to the slow absorption rate of the subcutaneous insulin injection. Moreover, once the glucose concentration has been lowered back to its target level, some insulin might still be active in the body, due to the slow clearance of currently available insulins when administered subcutaneously. The depot of active insulin can lead to low blood glucose (BG) several hours after the meal (late postprandial hypoglycemia). 13 These postprandial risks do not preclude the possibility of safe, effective fully closed-loop control in controlled clinical settings. 14 However, the risks can be mitigated with manual, “feedforward” delivery of rapid-acting insulin.15,16 Thus in many AP trials, all or part of patients’ meal time insulin has been calculated and delivered in a manual feedforward manner.17-21
The use of manual subcutaneous insulin boluses in an otherwise closed-loop system has several downsides. For rapid-acting insulin analogs delivered by insulin pump, postprandial control is better with a bolus delivered roughly 15 minutes before a meal rather than right at the start of eating. 22 However, preprandial insulin delivery also poses a hypoglycemia risk if a patient eats later or less than expected. Another problem is that both meal announcement and manual bolus require constant diabetes management by the user. This may limit the quality-of-life benefits of an AP system and increase the potential for human error. Also, regardless of when subcutaneous insulin is delivered, its action profile does not closely match that of endogenous insulin release in people without diabetes. 23
Among the key differences between subcutaneous insulin delivery and physiological insulin release is the fact that people without diabetes experience first-phase insulin secretion in response to food stimuli (cephalic insulin response). This spike in blood insulin concentration can begin at, or even in anticipation of, the first ingestion of food.24,25 By contrast, subcutaneously injected rapid-acting insulin analogs show a comparatively smooth ascent up to their peak blood concentration (over 40 minutes after injection) and peak glycemic effect (well over an hour after injection).26,27 The action profile of subcutaneously delivered insulin analogs can be enhanced by a variety of methods under investigation, including coadministration with recombinant human hyaluronidase, 28 coformulation with disodium EDTA and citrate, 29 and heating the site of infusion or injection; 30 each of these methods could be useful in an AP. Even more favorable pharmacokinetics and pharmacodynamics might be achievable through a different route of insulin administration, such as intraperitoneal 31 or—as we describe in this article—pulmonary.
We have previously proposed that for type 1 diabetes patients using AP systems, both glucose control and quality of life could improve with the meal time use of inhalable Technosphere® Insulin (TI). 32 TI (Afrezza™; MannKind Corporation, Valencia, CA) consists of recombinant regular human insulin in a dry powder that can be delivered to the lungs using a breath-powered inhaler (Dreamboat™). For decades scientists have recognized that the lungs’ large surface area and high perfusion could allow for rapid pulmonary uptake of insulin, 33 and TI’s profile is particularly favorable. In healthy volunteers without diabetes, TI reached peak insulin concentration in 12-17 minutes and peak glycemic effect at 42-58 minutes, significantly faster than that of regular human insulin; TI’s duration of action was also significantly shorter. 34 A similar TI action profile has been reported in patients with type 2 diabetes, among whom TI suppresses endogenous glucose production faster than insulin lispro.35,36 In patients with type 1 diabetes using insulin glargine, a randomized trial showed that prandial TI led to significantly lower fasting and 1- and 2-hour postprandial glucose values compared to prandial insulin lispro, with a lower hypoglycemia event rate. 37 Several other published clinical studies have further supported TI’s safety and efficacy as a prandial insulin in type 1 diabetes29,38,39 and type 2 diabetes.29,40,41 At the time of the study TI was an investigational drug that was approved on June 27, 2014, as an ultra rapid-acting insulin for oral inhalation indicated for the treatment of adults with type 1 or type 2 diabetes mellitus in the US by the food and drug administration.42,43
Because of its short action profile, TI can significantly blunt the postprandial glucose excursion even if delivered at the start of a meal rather than in advance. Alternatively, we hypothesize that a similar overall insulin action profile (and thus similar glycemic effects) could be achieved by using a “priming” TI dose followed by subcutaneous infusion of rapid-acting insulin. If this subcutaneous insulin delivery were given by an AP system, then the resulting regimen would be conceptually equivalent to adding unannounced (ie, controller’s lack of awareness of the TI action) meal time TI boluses to closed-loop insulin delivery. As an adjunct to a closed-loop system, TI could be delivered in the same dosage at every meal as a prophylactic dose if meal content is >50 g CHO, with the closed-loop system handling all the meal-specific adjustments. This would circumvent a major limitation of TI in open-loop management of type 1 diabetes: the fact that doses are quantized in increments equivalent to 3-4 units of rapid-acting insulin. Such a protocol would also reduce the burden of meal-size estimation for patients. Indeed, the only necessary calculation would be binary: whether a meal is so small that use of TI would introduce undue risk of hypoglycemia.
A combined TI/closed-loop regimen has been tested in silico with the Food and Drug Administration (FDA)-Accepted University of Virginia/Padova Metabolic Simulator. 44 In that experiment, meal time doses of TI equivalent to 4 units of rapid-acting insulin were added to closed-loop subcutaneous insulin delivery as performed in the NIH-funded Control to Zone study. 45 Compared to prandial boluses of subcutaneous insulin lispro, use of TI was projected to increase the time that patients spend in glycemic target without causing extra hypoglycemia risk. Higher doses of TI (20 U of TI, equivalent to 6-8 units of rapid-acting insulin) led to further reductions in hyperglycemia, but they also increased the anticipated rate of hypoglycemia. 32
Herein we describe the first clinical trial of prandial TI as an adjunct to closed-loop insulin delivery.
Methods
Nine subjects with type 1 diabetes were recruited for the study, which was approved by the FDA and the Santa Barbara Cottage Health System’s Institutional Review Board. This study was a follow-up study to a fully automated AP design that did not use TI. The results summary of that study with the identical subjects IDs are discussed by Harvey et al. 45 As discussed in Harvey et al, 46 unannounced meals are still a challenge to an automated AP without a priming meal bolus. The use of TI as a simple and user-friendly priming meal bolus that is not announced to the AP system as an alternative to a prandial meal bolus to assist with meal control is the focus of this follow-up study. All subjects signed the Institutional Review Board–approved informed consent form. Inclusion criteria included age between 21 and 65 years, type 1 diabetes duration of at least 1 year, and use of an insulin pump with rapid-acting insulin for at least 6 months. Exclusion criteria included pregnancy, diabetic ketoacidosis within the past 6 months, A1C > 9.0%, severe hypoglycemia within the past year, concomitant disease or medication use affecting metabolic control, diabetic ketoacidosis within 6 months prior to enrollment, severe hypoglycemia resulting in seizure or loss of consciousness within 12 months prior to enrollment, pregnancy, being a nursing mother, active infection, active gastroparesis, abnormal spirometry, having smoked habitually within the 6 months prior to enrollment, and high insulin sensitivity (insulin-to-carbohydrate ratio greater than 1:12). Subjects were screened with a comprehensive metabolic panel, complete blood count, and thyroid tests, height and weight were measured, and the subject’s insulin pump information was downloaded and confirmed (basal rates, average total daily dose, insulin-to-CHO ratios, and correction factors).
The communication platform for closed-loop control was the Artificial Pancreas System (APS). 47 The CGM used was the Dexcom G4™ Platinum (Dexcom Inc, San Diego, CA), which was unmodified from its FDA-approved version. Continuous subcutaneous insulin infusion (CSII) was performed with the OmniPod® Insulin Management System (Insulet Corp, Bedford, MA). Each volunteer received the same rapid-acting insulin analog used in their typical diabetes management: either insulin aspart (NovoLog®; Novo Nordisk A/S, Bagsvaerd, Denmark), insulin lispro (HumaLog®; Eli Lilly and Co, Indianapolis, IN), or insulin glulisine (Apidra®; Sanofi SA, Paris, France).
Subcutaneous insulin delivery was controlled by a zone model predictive control algorithm (zone-MPC), which modified insulin delivery only if current or predicted CGM values were outside of the prespecified zone: 80-140 mg/dl. 48 The controller was initialized using only each patient’s total daily insulin dose. The system also had knowledge of the individual’s nominal basal profile. Operating simultaneously to the zone-MPC algorithm was a safety algorithm; the Health Monitoring System (HMS), which independently analyzed CGM data. 49 If the HMS predicted that sensor glucose would fall below 70 mg/dl within the next 15 minutes, it would alert the main APS interface and send a text message to the attending physician. The HMS suggested giving the subject roughly 16 g of carbohydrate (CHO).
Two to 3 days before each patient visited the clinical research center (CRC), she or he made an outpatient visit to have 2 CGM sensors inserted by the study staff and to receive training in CGM use. For 2 to 3 days, the CGM was used in blinded mode and calibrated according to manufacturer’s instructions with the OneTouch® Ultra® Blood Glucose Monitoring System (LifeScan, Inc, Milpitas, CA). Two to 3 days after sensor insertion, the patient arrived at the CRC at approximately 4:00
Patients were also studied for a similar time period using the exact same protocol except for the fact that no priming bolus was given. At 11:00
Throughout the CRC visit, reference BG values were measured with the YSI 2300 STAT Plus (YSI Life Sciences, Yellow Springs, OH): every 15 ± 5 minutes during hypoglycemia (YSI BG < 70 mg/dl or CGM-predicted glucose level of 70 mg/dl in the next 15 minutes, until YSI BG > 80 mg/dl), every 15 ± 5 minutes during extreme hyperglycemia (YSI BG > 400 mg/dl, until YSI BG < 300 mg/dl), every 15 ± 5 minutes during and 1 hour after exercise, and every 30 ± 10 minutes otherwise.
Results
The study’s primary endpoint was the comparison of the glucose time in range (70-180 mg/dl) during the 5-hour postprandial period with and without the use of TI. Secondary endpoints included analyses of hypo- and hyperglycemic excursions (minimum and maximum values), time spent outside target range, whether outside intervention was provided, as well as the time in range for the following periods: 70-150 mg/dl during and for 3 hours after exercise and 80-140 mg/dl during the entire study period, including the overnight period (12:00
The 9 subjects with type 1 diabetes participated in both 24-h closed-loop sessions. Demographics and closed-loop details are shown in Table 1. A summary of results is shown in Figure 1, with data comparing cumulative percentage time in range including both YSI BG and CGM. Figure 2 presents percentage time in different glycemic ranges based on YSI BG for both arms (with and without TI) for the full duration of the study (panel A), 5 hours postdinner and postbreakfast (panel B and C, respectively), 3 hours postexercise and overnight (panels D and E, respectively). The median percentage time in range (70-180 mg/dl, YSI BG) during the 5-hour postprandial period with the use of TI was 81.6% (IQ range 72.5-85%, Breakfast 75%, Dinner 93.3%) as compared to percentage time in range without the use of TI 60.0% (IQ range 50.8-75.8%, breakfast 61.6%, dinner 56.6%, P = .059). The median percentage time in range 80-140 mg/dl during the entire study period was 67.5% (IQ range 48.5-73.1%) as compared to percentage time in range without the use of TI of 55.2% (IQ range 52.0-60.1%, P = .032).
Results Summary as Provided by YSI BG and CGM With Demographic and Clinical Parameters of the Individual Subjects.
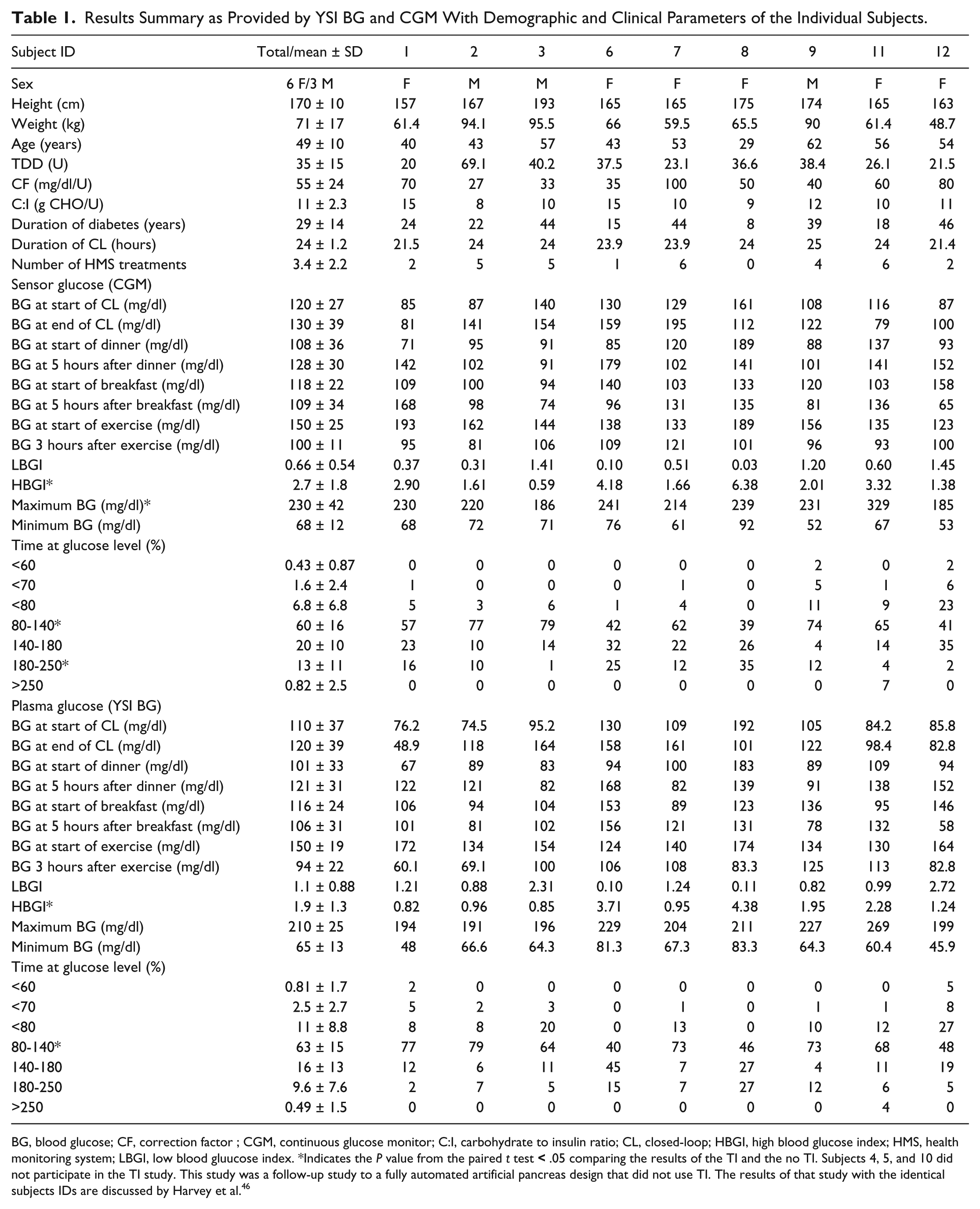
BG, blood glucose; CF, correction factor ; CGM, continuous glucose monitor; C:I, carbohydrate to insulin ratio; CL, closed-loop; HBGI, high blood glucose index; HMS, health monitoring system; LBGI, low blood gluucose index. *Indicates the P value from the paired t test
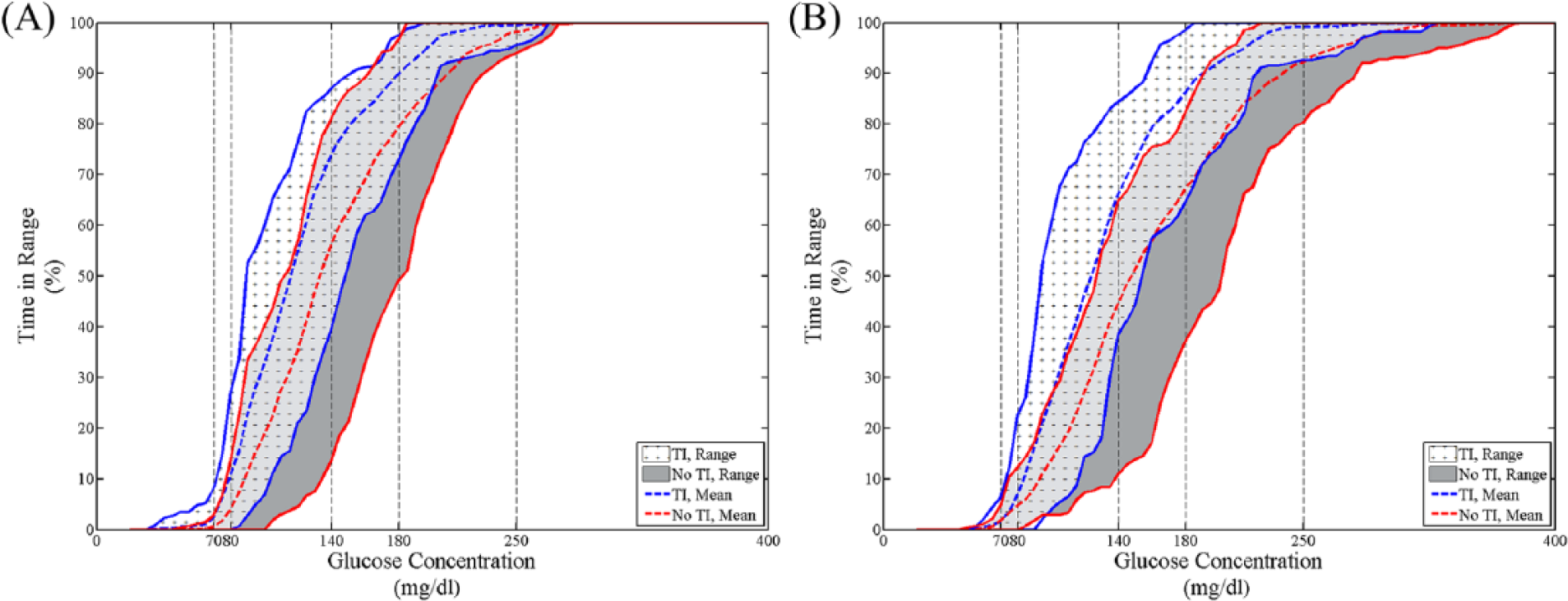
Cumulative percentage time in different ranges for all subjects in the TI case (white with cross marks) and the no TI case (gray) based on YSI BG (A) and CGM (B). The mean values for the TI case and the no TI case are represented by the blue dashed line and the red dashed line, respectively.
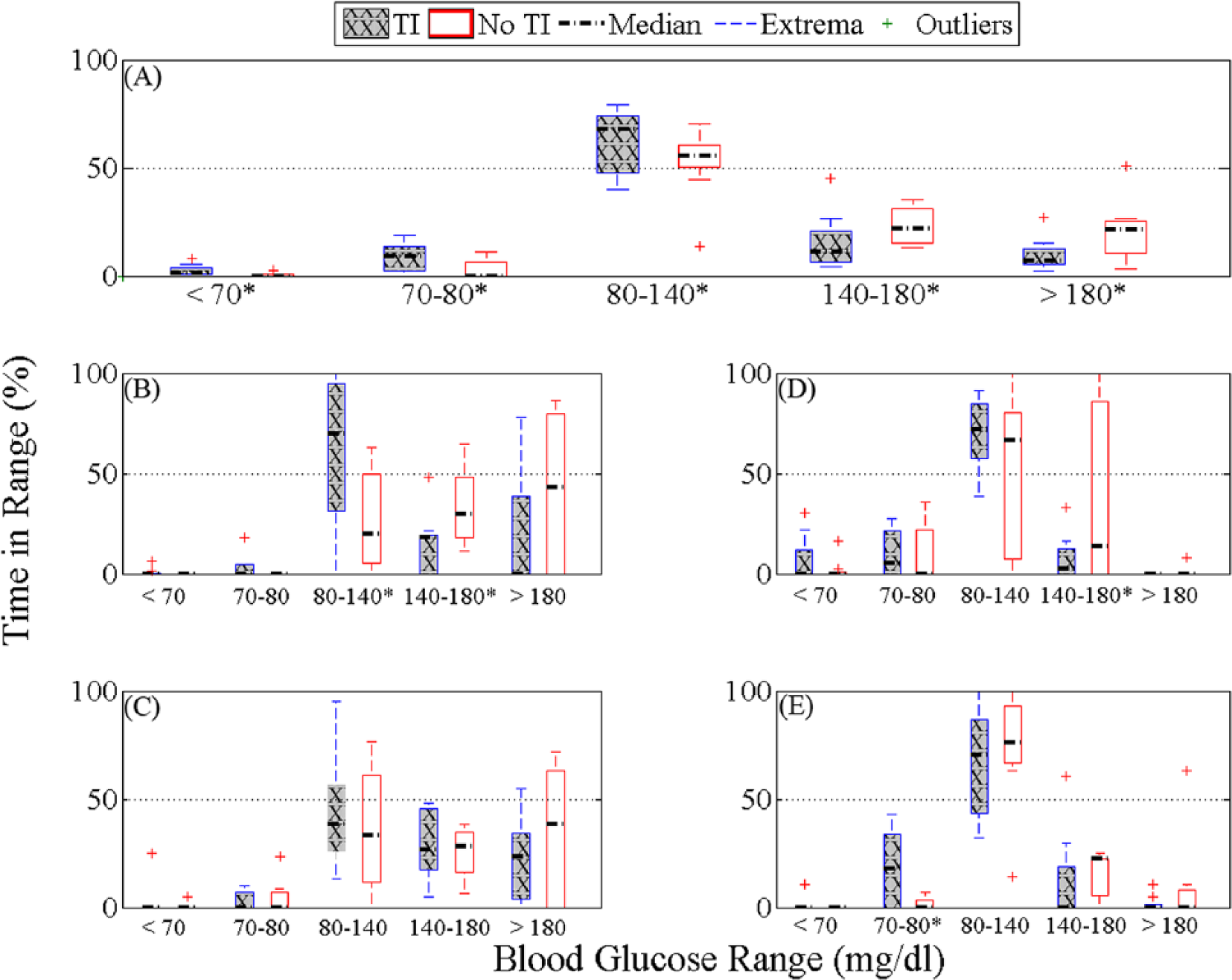
Percentage time in different glycemic ranges based on YSI BG in the TI case (gray with diagonal line) and the no TI case (white) for the whole study (A), start of dinner to 5 hours after (B), start of breakfast to 5 hours after (C), start of exercise to 3 hours after (D), and the overnight period (12:00
The median percentage time in range (70-150 mg/dl, YSI BG) during and for 3 hours after exercise was 94.4% (IQ range 91.6-94.4%) as compared to percentage time in range without the use of TI 83.3% (IQ range 30.5-94.4%, P = .19). Three hours after exercise the median glucose was 100 mg/dl (IQ range 82.8-107 mg/dl) with TI and 120 mg/dl (IQ range 112-127 mg/dl) without TI (P = .02). There was no significant difference during the overnight period (12:00
The total median insulin/24 hours was 29.6 U for the TI group (21.6 U of SC + 8 U of TI) and 20.9 U for the group without TI (Figures 3B and 3F, respectively). The percentage of subjects experiencing HMS alerts per 30 minutes is shown in Figure 3C and Figure 3G for the TI arm and the non-TI arm, respectively. There was no statistical difference in 30-minute alarms. Information regarding the starting time of each of the 3 unannounced challenges for both studies (dinner, breakfast, exercise), if shown as the percentage of subjects who have commenced the challenge (Figure 3D and Figure 3H for the TI and non-TI groups, respectively). All the results of the individual subjects are available in Online Supplementary Figures 1 to 9.
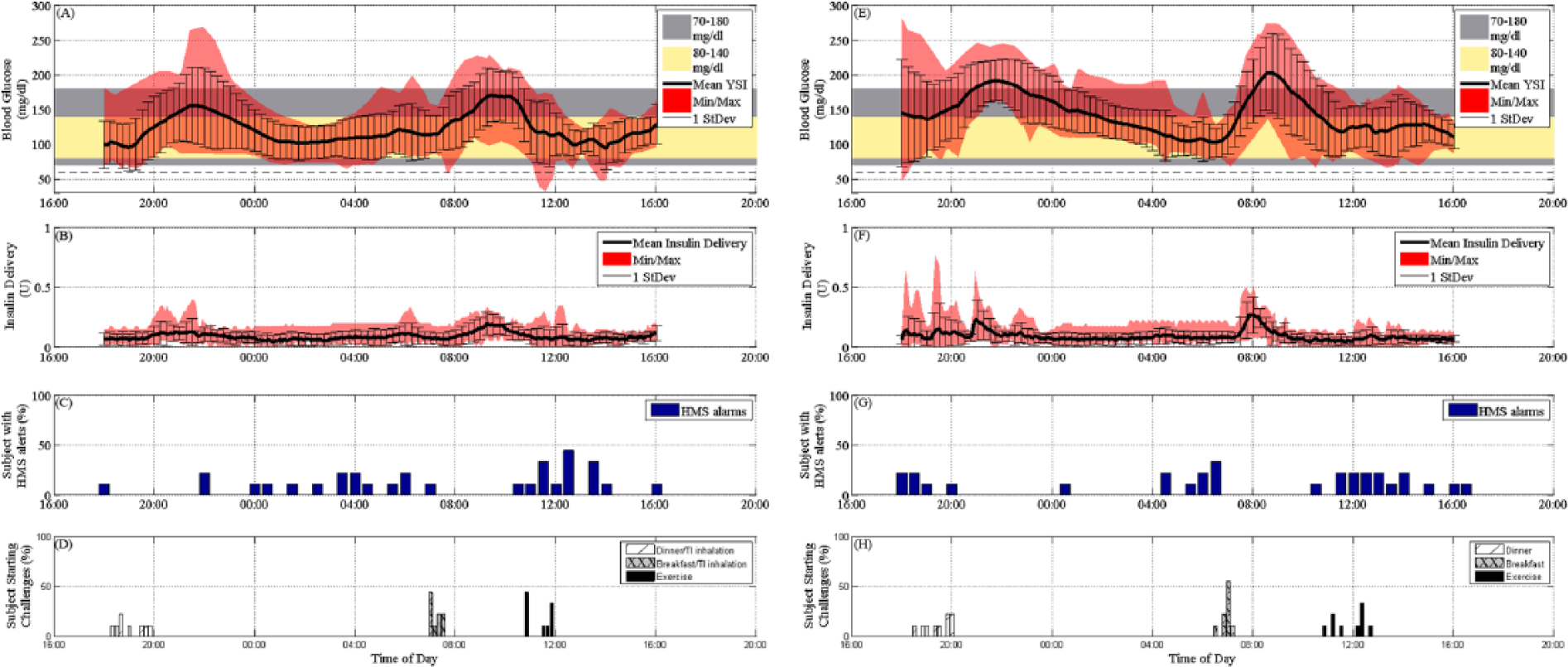
Glucose summary results of all TI trials are summarized in (A) as mean YSI BG, 1 standard deviation (SD), and minimum and maximum YSI BG values. The control objective (80-140 mg/dl) is shown as the light yellow band and the clinically accepted region (70-180 mg/dl) as the gray band. The mean insulin delivery via CSII for all trials is shown in (B) along with 1 SD and the minimum and maximum of the insulin delivery values. The percentage of subjects received HMS alarms in a 30-minute interval is shown in (C). The time of starting the challenges and TI inhalation is shown in (D). Glucose summary results of all no TI trials are summarized in (E) as mean YSI BG, 1 SD, and minimum and maximum YSI BG values. The control objective (80-140 mg/dl) is shown as the light yellow band and the clinically accepted region (70-180 mg/dl) as the gray band. The mean insulin delivery via CSII for all trials is shown in (F) along with 1 SD and the minimum and maximum of the insulin delivery values. The percentage of subjects received HMS alarms in a 30-minute interval is shown in (G). The time of starting the challenges and TI inhalation is shown in (H).
As can be seen in the glucose tracing in Figure 3A, there was an overall a mild postprandial rise in glucose concentrations as compared to the non-TI case (Figure 3E). Some subjects had a higher postprandial peak, generally when they had started at a higher value at meal time. Nevertheless, a significant reduction in postprandial excursion was observed with the TI group compared to the control group (median delta YSI BG of 57 mg/dl and 95.5 mg/dl from the baseline at meal time by YSI BG in the with and without TI cases, respectively, P = .006). After dinner, the median delta glucose peaks by YSI BG were 64 mg/dl (IQ range, 30-120 mg/dl) and 85 mg/dl (IQ range, 50-112 mg/dl) in the with and without TI cases, respectively, with P values of .15, and the median peak times by YSI BG were at 150 (IQ range, 120-180 minutes) and 121 minutes (IQ range, 105-159 minutes) in the with and without TI cases, respectively, with P values of .18. Median baseline predinner glucose values by YSI BG were 94 mg/dl (IQ range, 89-100 mg/dl) and 124 mg/dl (IQ range, 91-134 mg/dl) in the with and without TI cases, respectively.
After breakfast, the median peak postprandial glucose excursion was 61 mg/dl (IQ range, 56-85 mg/dl) and 99 mg/dl (IQ range, 75-118 mg/dl) by YSI with and without TI, respectively, with a P value of .008, and the median time to peak occurred at 120 minutes (IQ range, 90-150 minutes) and 107 minutes (IQ range, 95-131 minutes) in the with and without TI cases, respectively, with a P value of .22. Median baseline prebreakfast glucose values by YSI BG were 106 mg/dl (IQ range, 95-136 mg/dl) and 110 mg/dl (IQ range, 89-131 mg/dl) in the with and without TI cases, respectively.
Mild activity was undertaken for all subjects for 30 minutes beginning between 11:10
Conclusions
A major rate-limiting factor in the development of an AP is the delay in insulin action associated with currently available rapid acting insulin analogs. Previous investigations have demonstrated that meal control is one of the major challenges of an automated insulin management and even a premeal bolus has its limitation in preventing postprandial hyperglycemia. We have previously demonstrated that fully automated AP closed-loop systems can safely control glucose concentrations in patients with type 1 diabetes under limited CHO consumption. The postprandial performance of this type of system as currently designed is less than ideal, since if one wants optimal control, meal sizes would need to be restricted or one would need to have a system with the addition of glucagon. The addition of an ultra-rapid insulin at meal time allows for superior postprandial control. Using this hybrid semiautomated approach has several advantages. First the patient would have to do only a rough estimation of the CHO content. The controller will take care of any remaining insulin requirement. Second, the use of TI mimics first phase secretion of insulin with a fast-peak and rapid clearance, which results in lower postprandial peaks and less late postprandial hypoglycemia. Third, its use also results in both lower insulin delivery and glucose concentration variability.
The convenience/ease of use was commented on by all of the study participants. Adding TI to an automated closed-loop AP system results in superior postprandial control as demonstrated by lower postprandial glucose exposure by 81% and 60% for dinner and breakfast, respectively, without additional hypoglycemia. There is less variability in both insulin delivery and glucose concentrations in the TI arm. As seen in most AP studies to date there was no difference in glucose control during the overnight period.
We acknowledge that we are giving a partial premeal bolus for the TI arm, but we wanted to demonstrate that the addition of an ultra-rapid insulin to an automated AP system improves postprandial control without the need to estimate the meal content. We acknowledge that glucose control in the postbreakfast period is still difficult to control. We anticipate the in future studies there would be a proportional dose escalation around periods of increased insulin resistance and meals with increased CHO intake. We also acknowledge that using a full meal announcement in combination with closed-loop control will most likely result in more time in range. This study was designed to demonstrate a proof of concept that giving a small, consistent bolus of ultra-rapid insulin that provides a first phase meal correction could improve the postprandial glucose profile as compared to fully automated closed-loop control with very minimal user intervention or need to count CHOs and enter estimation to the closed-loop system.
Footnotes
Acknowledgements
MannKind Corporation provided Technosphere pharmacokinetics and pharmacodynamics data. Joseph P. Shivers (Sansum Diabetes Research Institute) assisted with the writing of the manuscript. We also acknowledge product support from Insulet Corp, Dexcom Inc, and Lifescan Inc. Clinicaltrials.gov NCT01874392. Video supplement: ![]() .
.
Abbreviations
AP, artificial pancreas; APS, Artificial Pancreas System; BG, blood glucose; CGM, continuous glucose monitor; CHO, carbohydrate; CRC, clinical research center; CSII, continuous subcutaneous insulin infusion; FDA, Food and Drug Administration; HMS, Health Monitoring System; T1DM, type 1 diabetes mellitus; TI, Technosphere® Insulin; Zone-MPC, Zone Model Predictive Controller.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HZ was a consultant to Mankind during employment at Sansum Diabetes Research Institute.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the JDRF (17-2011-765) and the National Institutes of Health (DP3 DK094331 and R01 DK085628).
