Abstract
Exaggerated postprandial glycemic excursions in the first hours after a meal and an increased risk of late postprandial hypoglycemia are commonly observed following bolus injections of regular human insulin due to the delayed peak and the prolonged duration of action of this insulin preparation. 1 In contrast, plasma insulin concentrations peak earlier (generally thought to be within the first hour) and plasma insulin concentrations return to baseline more quickly with rapid-acting insulin analogs (RAIAs). 2 Nevertheless, even with the more favorable absorption characteristics of RAIAs, postmeal hyperglycemia remains difficult to control in insulin-requiring diabetic patients due to delays in the peak action of these analogs to 120 or more minutes after injection. 3
The time to peak insulin concentrations following bolus injections of both human regular and RAIAs are dose dependent, 3 with higher doses leading to delayed peaks and prolonged duration of action of these insulins. Moreover, the FDA-approved prescribing information package inserts for RAIAs state that the time course of action of these insulins may vary in different individuals or at different times in the same individual. 4 Such variations in insulin action are dependent on many conditions, including the site of injection, local blood supply and local skin temperature. 4
The impact of the site of injection on the rate of absorption of regular insulin (ie, abdomen > arm > leg) was first described by Koivisto and Felig in 1980. 5 They also demonstrated that increasing the skin temperature to 30°C to 37°C via sauna use increased the rate of insulin absorption and significantly lowered postprandial glucose excursions compared to control meal studies carried out at standard room temperature. 6
The InsuPatch skin-warming device was designed to provide local warming around insulin infusion sites in insulin pump treated patients. Recent studies with this device have shown that infusion site warming in patients with type 1 diabetes increased local blood flow and reduced postprandial glycemic excursions.7-9 Using the glucose clamp technique, improvements in the pharmacokinetics (PK) and pharmacodynamics (PD) of a standard bolus dose of aspart insulin were observed with infusion site warming in young, pump-treated patients with type 1 diabetes. 10 The InsuPad is an alternative skin-warming device that was designed for patients receiving basal-bolus multiple daily injection therapy. In previous randomized control study, use of the InsuPad device was found to enable patient to improve their glucose control with reduced prandial insulin dose and significantly reduced hypoglycemia rate. 11 In this study, we tested whether local warming using the InsuPad would be effective in lowering postprandial glucose excursions in patients with type 2 diabetes compared to premeal injections of the identical dose of aspart or lispro insulin without skin warming.
Research Design And Methods
Subjects
The study was an open-label, randomized, 2-period, 1-way crossover study. The Hadassah Medical Center institutional review board approved the study; the patients provided written informed consent prior to the study (ICMJE registry number NCT02306343). To be eligible for the study, the subjects had to have type 2 diabetes that was being treated with basal-bolus insulin therapy; age >18 and <75 years and HbA1c values <11%. Exclusion criteria included pregnancy or breastfeeding, alcohol abuse, or other serious medical or psychiatric problems.
Procedures
Each subject participated in the same meal tolerance test (MTT) protocol twice in randomized order, once with (test day) and once without (control day) application of local warming to the skin surrounding the injection site.
On each study day, subjects reported to the clinical research center in the morning after an 8-10 hour overnight fast. An indwelling intravenous catheter was placed for blood sampling and blood glucose levels were stabilized between 100-150 mg/dl. An InsuPad was placed on the abdomen and it was set to warm the skin to 40°C. The study was performed in a sitting position in a temperature-controlled room (24 ± 3°C). On both study days subjects received a bolus dose of the same RAIA (ie, aspart or lispro) that they were receiving prior to the study.
MTTs
On the Test Day, local warming with the InsuPad was activated immediately after an insulin aspart or lispro bolus (0.2 U/kg body weight) was given at t = 0 minutes. The injection was given through the injection window of the warming device on the test day (Figure 1) or directly into the subcutaneous tissue of the anterior abdominal wall without warming on the control day.

Picture of the InsuPad. When opening the InsuPad the injection area is shown.
Immediately after the injection, the patients were asked to drink a standardized liquid meal (Ensure Plus, Abbott, Columbus, OH, USA; 474 ml, carbohydrate 100 g, protein 26 g, fat 23 g) within 10 minutes. Samples for measurement of serum glucose and insulin concentrations were taken from a peripheral venous line at predetermined time points. Total follow up time was 5 hours postmeal. If glucose levels declined during the MTT to <75 mg/dl or increased to >300 mg/dl, glucose or insulin was given and the study was stopped. In studies that were prematurely terminated, the subject’s blood glucose levels were monitored for safety purposes only. If the subject did not complete the first 140 minutes of the MTT procedure, the study was repeated on a different day.
Pharmacokinetic and Pharmacodynamic Measures
The PK and PD parameters that were analyzed are shown in Table 1.
Pharmacodynamic and Pharmacokinetic Parameters Without and With Injection Site Warming With the InsuPad.
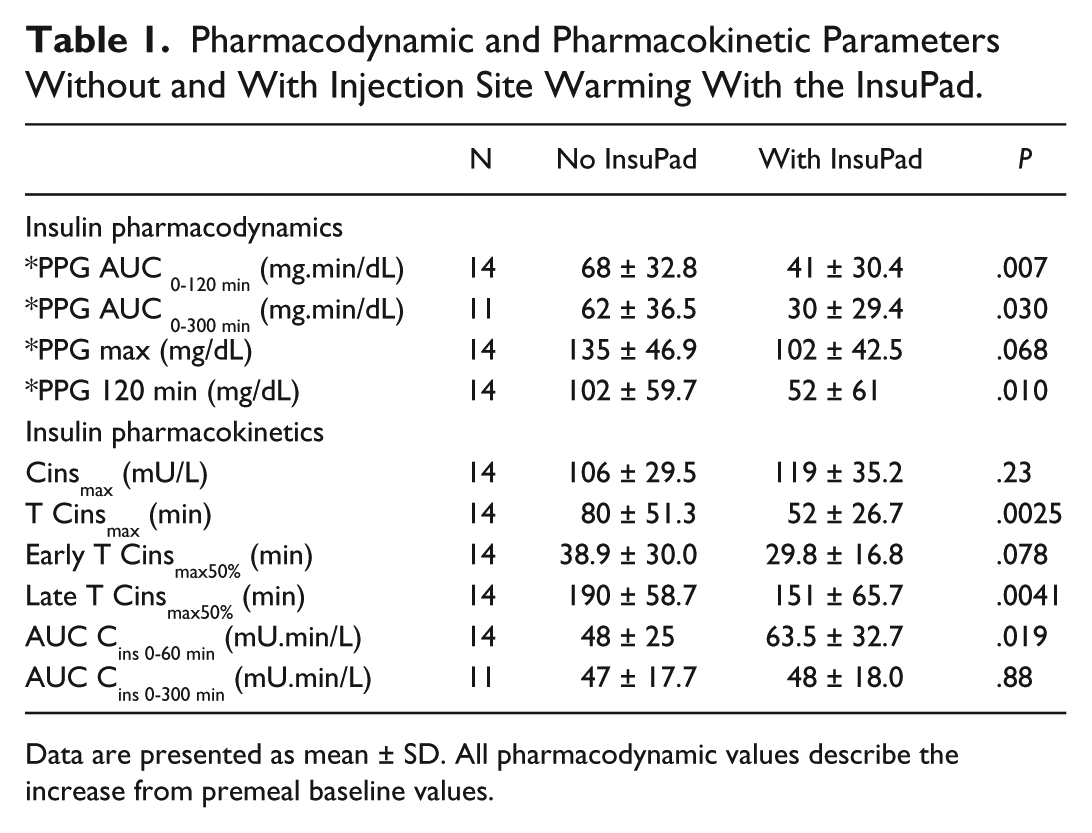
Data are presented as mean ± SD. All pharmacodynamic values describe the increase from premeal baseline values.
The primary efficacy outcome of the study was the area under the postprandial glucose excursion curve during the first 120 minutes after the start of the meal (PPG AUC 0-120 min). Secondary PD endpoints included differences between the 2 studies (warming or no warming) regarding the peak increment in blood glucose after the meal (PPG max), mean blood glucose excursions at 120 minutes and area under the curve (AUC) of the postprandial blood glucose excursion for full 5 hours after the meal (PPG AUC 0-300 min). The peak plasma insulin concentration (Cinsmax), time to peak insulin concentration, time to early 50% of peak insulin concentration, time to late 50% of peak insulin concentration (T Cinsmax, Early T Cinsmax50%, Late T Cinsmax50%,) and area under the insulin response curves (AUC Cins 0-60 min and AUC Cins 0-300 min) were the PK parameters of interest. The primary safety endpoints were evidence of skin inflammation or irritation and mild hypoglycemia (<75 mg/dl) and hyperglycemia (>300 mg/dl).
Laboratory Methods
Blood samples for serum glucose and insulin measurements were collected in serum separating gel tubes and kept for 20 minutes on ice to allow for clotting and then were centrifuged and stored in -20 degrees Celsius until measurement. Glucose levels were measured using hexokinase enzymatic method (Glucose HK gen.3 on Cobas 800 system, Roche Diagnostics, Manheim, Germany). Insulin assays were performed using an enzyme-linked immunosorbent assay test kit (Iso-Insulin 10-1128-01, Mercodia, Uppsala, Sweden).
Data Analysis
Postprandial glucose excursions were calculated by subtracting the baseline levels at time 0 from all subsequent measurement results. AUC values were calculated using the linear trapezoidal method. A paired t test was used for comparing the results obtained on both study days. All subjects included in the analysis of the primary end point (PPG AUC0-120 min)) completed at least 140 minutes, subjects that did not complete the 5 hours follow-up in both test and control conditions were not included in the analysis of the secondary endpoint of PPG AUC0-300 min. PK and PD data are given as mean ± SD.
Results
Patient Demographics
Ten males and 4 females participated in the study: mean (± SD) age was 61.6 ± 8.4 years, HbA1c: 8.42 ± 1.13%; BMI 29.10 ± 5.61 kg/m2 and diabetes duration: 20.1 ± 9.0 years; prandial daily insulin dose: 34.6 ± 14.6 IU; daily basal insulin dose: 47.3 ± 15.0 IU. Four patients were on antidiabetic oral drug (Metformin) which was the same on both test and control visits.
Glucose and Insulin Profiles During Meal Tolerance Testing
As shown in Figure 2, baseline blood glucose levels were similar on test and control days (128 ± 16 vs 134 ± 12 mg/dl, respectively, P = .42). While the initial rise in serum glucose levels was similar during the first 30-40 minutes on both study days, the 2 curves diverged thereafter. Consequently, the AUC of the postprandial glucose excursion during the 2 hours following the meal (PPG AUC0-120 min ) was reduced by 40% (P = .007) (Table 1) when local warming was applied to the injection site. As shown in Table 1, other PD parameters were also significantly improved by injection site warming.
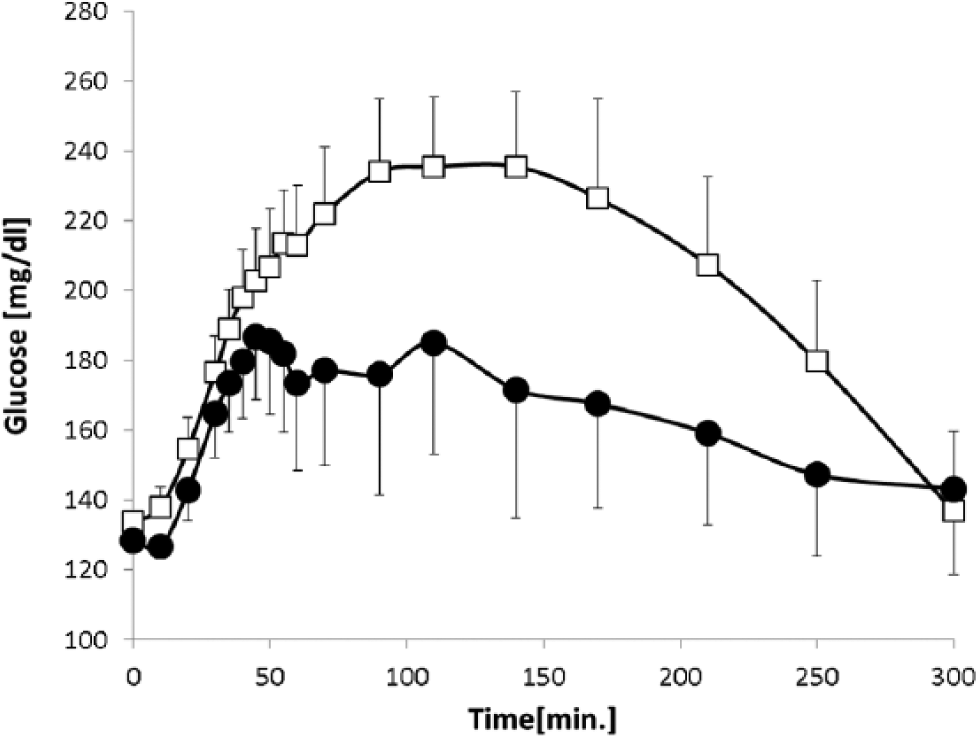
Postprandial glycemic excursions after a liquid meal with and without local application of heat in patients with type 2 diabetes. Data are presented as mean ± SE.
Mean serum insulin concentrations on the 2 study days are shown in Figure 3, and PK analyses are shown in Table 1. Fasting serum insulin concentrations were nearly identical on both the test and control days (16.45 ± 8.54 vs 13.93 ± 5.62 mU/L, respectively, P = .35). It is noteworthy that skin warming with the InsuPad had no effect on the peak insulin concentration achieved following injection (Cinsmax) or the AUC Cins 0-300 min (Table 1). Nevertheless, mean serum insulin levels were increased during the first 60 minutes with InsuPad versus No InsuPad, resulting in a significant increase in the AUC Cins 0-60 min (P = .006).
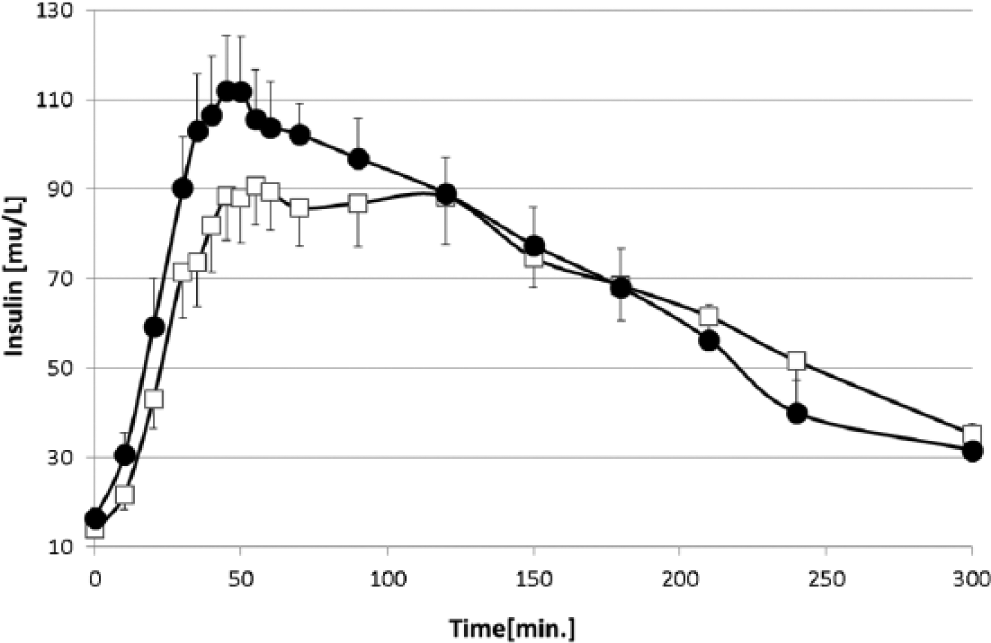
Circulating insulin concentrations after a liquid meal with and without local application of heat in study subjects. Data are presented as mean ± SE.
Figure 4 shows the T Cinsmax for individual subjects under each experimental condition. In the control experiments, T Cinsmax varied widely, occurring after 50 minutes in 10 of the 14 subjects. Indeed in 2 of these subjects, T Cinsmax was markedly delayed to 140 and 210 minutes. In contrast, with InsuPad, only 4 of the 14 subjects had T Cinsmax ≥ 50 minutes.
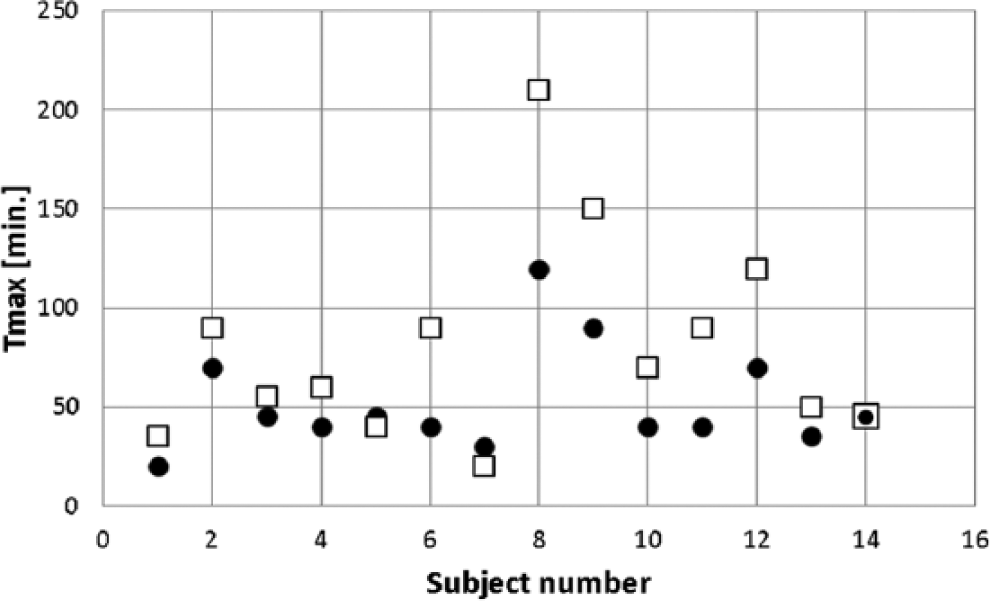
Differences in the time to maximum insulin concentration following the 0.2 U/kg bolus dose of aspart or lispro insulin. White squares indicate T Cinsmax on control days and black circles on test days.
Local warming was well tolerated by all study participants. Comparison of the skin area at the injection site with or without use of the warming device showed no occurrence of irritation or inflammation at the site. Three patients in the control visits needed intervention before 300 minutes, 2 due to hypoglycemia (BG < 75 mg/dl) and 1 due to hyperglycemia (BG > 300 mg/dl). Two patients in the test visits needed intervention before 300 minutes due to hypoglycemia.
Conclusions
The InsuPad device was developed to take advantage of the increase in the rate of absorption of subcutaneously injected insulin that accompanies increases in local blood flow due to skin warming, demonstrated by Kiovisto more than 30 years ago. As we hypothesized, use of the InsuPad in adults with type 2 diabetes was associated with a substantial reduction in postprandial glucose excursions during the first 2 hours of a MTT compared to control experiments without skin warming. In addition to significant differences in the primary efficacy outcome of the study, all of the other secondary PD outcomes were also improved with injection site warming.
It is well recognized that a number of factors including local skin temperature and local blood flow contribute to between subject variations in the rate of absorption of subcutaneous injections of RAIAs. These factors undoubtedly played a role in the wide variations in the time to maximum insulin concentrations in the control studies. Indeed, contrary to conventional wisdom, the T Cinsmax exceeded 50 minutes in the majority of control studies but was less than 50 minutes in all but 4 of the 14 studies with skin warming. It is also noteworthy that the greatest decreases in T Cinsmax with the InsuPad were observed in those subjects with the longest delay in T Cinsmax during control studies.
Postprandial hyperglycemia was much better controlled during test versus control studies, even though the overall bioavailability of insulin (as reflected by the AUC Cins 0-300 min) and the peak serum insulin levels (Cinsmax) were not affected by skin warming. Thus, better control of postprandial glucose with the InsuPad was explained by consistently earlier peaks rather than by increases in the magnitude of peak serum insulin levels.
A number of other approaches are currently under study to accelerate that time-action profile of RAIAs. These include micro-needle, intradermal infusion sets for pump therapy,12-15 changes in excipients in RAIA formulations to accelerate insulin molecule monomerization 16 and treating the insulin infusion site with hyaluronidase to breakdown interstitial barriers to dispersion of injected liquids. 17 In comparison to these approaches, local warming relies on a relatively simple physiological approach which, in this study, we have demonstrated is suited for injection as well as pump therapy. Moreover, these approaches are not mutually exclusive; local warming could be added to other measures to increase the rate of insulin absorption even further. 18
Day-to-day and dose-to-dose variations in the time action profiles of premeal boluses of RAIAs contribute to both marked hyperglycemic excursions and sudden, unexpected hypoglycemia even when the same patient gives the same premeal bolus dose of RAIA for identical meals and under similar conditions. Use of the InsuPad may reduce unpredictable changes in local blood flow and skin temperature and should, in turn, reduce the risk of sudden hyper- and hypoglycemia; making optimal control of diabetes less difficult to achieve. 11
Footnotes
Abbreviations
AUC, area under the curve; early T Cinsmax50%, time to early 50% of peak insulin concentration; FDA, Food and Drug Administration; IU, insulin units; late T Cinsmax50%, time to late 50% of peak insulin concentration; MTT, meal tolerance test; PD, pharmacodynamics; PK, pharmacokinetics; PPG, postprandial glucose; RAIAs, rapid-acting insulin analogs; SD, standard deviation; SE, standard error; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IR is a consultant and shareholder in Insuline Medical Ltd, AP and WVT are Insuline Medical Ltd consultants. DF, and TA are Insuline Medical Ltd employees. GB is an Insuline Medical Ltd employee and shareholder.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by Insuline Medical Ltd, Petach Tikva, Israel.
