Abstract

Insulin dosing software is increasingly residing on mobile medical devices such as blood glucose (BG) monitors, continuous glucose monitors, and handheld devices. Most of the available software products worldwide are not cleared by the US Food and Drug Administration (FDA). I expect that we will all be seeing even more of such software products in the next few years because of 3 trends: (1) the evidence for these products to provide benefits is accumulating, (2) the public’s demand for mobile medical devices is growing, and (3) the mHealth cloud based software infrastructure is becoming more established. For this type of product to become accepted as mainstream medical products it will be necessary for them to receive regulatory clearance. The regulatory pathway at FDA is evolving. As the demand for assistance with insulin dosing grows, I expect that this type of software will become established as a widely used tool for managing insulin-requiring diabetes.
Potential Benefits
Software can recommend an ideal dose of insulin as well or better than a person if all the important factors determining dosing are known. Examples of software-controlled activities include a self-parking car and an airplane flying on autopilot. In both cases, there must be an operator ready to take over in case the system is heading for trouble. Software for determining a proper insulin dose must incorporate the most important factors affecting the dose. For bolus insulin dosing, 6 factors must be taken into account. Five of the 6 are known to the patient at all times and only 1 of them (the carbohydrate content of the planned meal) varies at different points in time (Table 1). Patients who will use software for determining an insulin dose must be aware of a potential dosing error and then the patient must take over. Self-parking cars will require drivers to be in the cars to be ready to take over and airplanes require pilots to be on board to take over. Likewise computerized insulin dosing require a trained patient who is ready to override the software and take control of insulin dosing if the software instructions are not correct for that situation.
The 6 Factors That Determine the Correct Amount of Insulin for a Bolus Dose.

Computerized software is driven primarily by poor numeracy. A bolus insulin dose is based on a standard dose for ideal or baseline conditions along with a modifying factor for the current situation. Factors which can modify the standard dose include (1) not being at baseline glycemic level when the dose is administered, (2) not having standard insulin sensitivity at the time of a dose, (3) eating a meal which is not predicted to be absorbed at the usual rate, and (4) being in a state where the insulin will not be absorbed at the usual rate. In each case, the physician typically instructs the patient how to modify a dose and the software must be able to incorporate these variables so that a recommended dose is appropriate for the situation where it will be used.
Use of the automated a bolus calculator has been shown to minimize errors in dose determination. 1 There is also an increasing body of evidence that bolus calculators may improve glycemic control and treatment satisfaction in patients who use these devices as intended. 2
Poor Numeracy
Numeracy is defined as the ability to understand and work with numbers. A patient with diabetes is expected to measure carbohydrates, interpret BG values, interpret A1C values, calculate insulin doses for meals, and calculate insulin doses to correct abnormal glycemic levels. Many patients have poor numeracy skills and have difficulty with calculating insulin doses or interpreting glucose or A1C results.3,4 A review of 34 articles discussing numeracy in diabetes in 2013 by Al Sayah et al 5 concluded that low literacy and poor numeracy are associated with poorer diabetes knowledge, but there is insufficient evidence to conclude that these deficits are actually associated with poor outcomes. Since the premise for insulin calculating software is to correct improve dosing inaccuracy caused by poor numeracy, it would be helpful to regulators for such software developers to demonstrate the effectiveness and safety of these products.
Potential Risks
Regarding effectiveness, one might assume that correct dosing will lead to fewer dosing errors and smoother glycemic levels and therefore some combination of improved A1C or fewer hypoglycemic episodes or hyperglycemic episodes. These metrics could be beneficial effectiveness outcomes. The theory here is that the problem of poor numeracy leads to incorrect insulin dosing, which leads to poor glycemic control. An insulin dose calculator can overcome much of the dosing inaccuracy associated with poor numeracy. To the extent that dosing inaccuracy can be corrected by this type of tool, it does not automatically follow that a dose calculator will improve glycemic control. It is not necessarily correct that achievement of correct dosing of insulin leads to either improved control or no worsening of control. Caution is needed for 2 reasons.
The first reason for caution relates to the effectiveness of dose calculators. In some patients with poor numeracy the cause of poor control is not solely the incorrect dosing problems that can be corrected by software, but a higher order problem related to poor understanding of or compliance with all aspects of diabetes therapy. In that case, even if the incorrect insulin dosing is corrected, there could be other behaviors that will overwhelm the improvement in dosing and still continue to lead to poor outcomes such that the software will have minimal effect on mean glycemia. In that case, incorrect dosing would be an epiphenomenon of poor understanding of diabetes and not the factor that requires the most attention. An intervention for poor numeracy in diabetes can result in the desired surrogate endpoint for an improved outcome, which is correct insulin dosing, through the use of insulin dosing software system, however there can still be poor outcomes. This unfortunate situation can be due to either (1) a more fundamental phenomenon linking poor numeracy with factors besides correct dosing or (2) unexpected acute problems related to diabetes that cannot be overcome by correct insulin dosing (Figure 1). Given the lack of an association between poor numeracy and poor control reported by Al Sayah et al, 5 providing a tool to correct poor numeracy will not automatically improve mean glycemia.
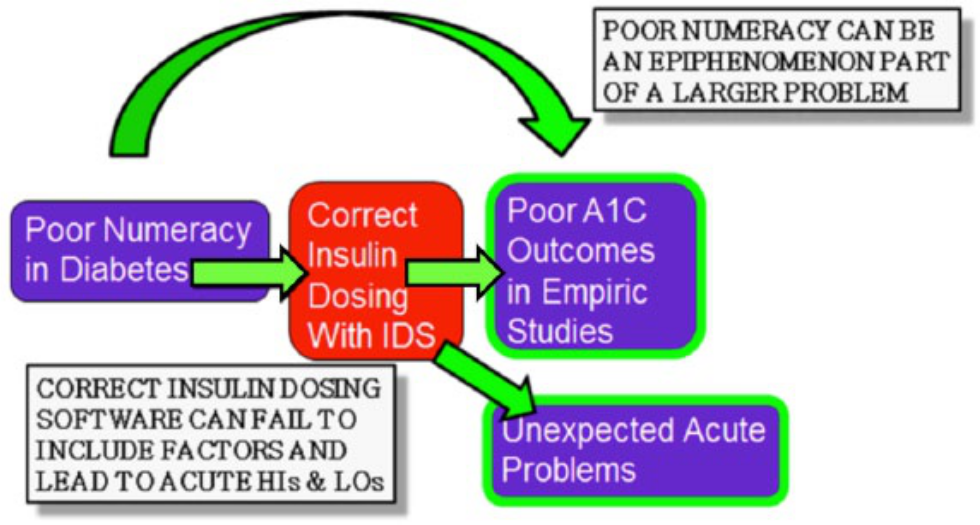
Two mechanisms whereby an intervention for poor numeracy in diabetes intended to achieve the surrogate endpoint of correct insulin dosing through the use of insulin dosing software can result in the desired surrogate endpoint along with a poor clinical outcome.
The second reason for caution relates to the safety of dose calculators. In some patients with poor numeracy, it is possible that such software could lead to some adverse short-term glycemic excursions if the software is used improperly. Stacking is a problem caused by repeated correction blouses while insulin from other recent boluses is still on board. If patients fail to program insulin on board into their software system, then there is a risk of the software recommending excessive insulin dosing resulting in short-term hypoglycemia. During periods of increased insulin resistance insulin doses must be increased, and during periods of decreased insulin resistance insulin doses must be decreased. If a patient becomes excessively reliant on bolus dose calculation software and does not manually program short-term events that can alter resistance, then there is a risk of the software recommending inadequate or excessive insulin dosing resulting in short-term hyperglycemia or hypoglycemia. Furthermore, the metabolic effect of insulin is nonlinear. Most bolus advisors do not take this physiologic factor into account and they fail to reduce the insulin sensitivity factor when the BG is below 90-100 mg/dl. 6
Buyer Beware or Be Where the Cleared Products Are
Given the uncertainty as to whether insulin dose calculation software will truly improve outcomes in terms of effectiveness and safety, it is advisable to conduct empirical trials of insulin calculation software to determine the effectiveness and safety of these products. I believe that superiority endpoints for safety and noninferiority endpoints effectiveness are worthy regulatory goals for medical software.
Hundreds of products are available as mobile diabetes applications providing various functions with little objective evidence to recommend most of them. In 2013 Eng and Lee 7 studied 100 randomly selected marketed mobile medical applications for diabetes from the iTunes store. Eight of them were for insulin dosing, and all 8 were not cleared by FDA. The peer-reviewed literature contains few studies of the safety and effectiveness of most diabetes software products and even the published studies are often underpowered for showing a statistically significant endpoint.
In September 2014 Roche launched its ACCU-CHEK Aviva Expert BG meter as the first ever FDA-cleared BG monitor providing insulin dosage calculation. Before patient use, a health care professional must prescribe the ACCU-CHEK Aviva Expert system and provide the patient-specific target BG, insulin-to-carbohydrate ratio, and insulin sensitivity parameters to be programmed into the ACCU-CHEK Bolus Advisor. Once programmed, a patient must consult with the health care professional before making any changes to these ACCU-CHEK Bolus Advisor settings. 8 I expect that before long, other BG monitors will also receive FDA clearance for BG monitors with insulin dosing capabilities.
Bolus Software Embedded in Insulin Pumps
One reason why bolus calculating software is potentially safer in a pump than in a BG monitor is that pumps keep track of repeated correction doses that are administered in a short period of time and these products can be programmed an with insulin-on-board feature (the duration of action of an insulin bolus) such that a series of bolus doses for hyperglycemia cannot become excessive because the pump keep track of prior doses and not recommend a dose of insulin to be administered in excess of what is still on board but that has not yet taken effect. A second reason why bolus calculating software is potentially safer in a pump than in a BG monitor is that if a patient develops a problem with their glucose level, it is possible to review the dosing records and learn whether an unintended dose of insulin was administered and determine how to avoid the problem in the future. In contrast, with a BG monitor containing software, the actually administered insulin dose cannot be determined. A pen that can keep track of administered insulin would be very useful for patients who are using bolus software because the software would then be able to keep track of insulin on board and avoid stacking. 9
Future Directions in Bolus Calculator Regulation
From the technical perspective, cleared bolus calculator software will need to be safe. In 2014 Rees 10 recommended 5 required elements for future bolus calculator software: (1) in vitro testing of insulin dosing algorithms utilizing an in vitro simulator such as the University of Virginia simulator; (2) risk management activities by global regulatory agencies in accordance with either AAMI TIR32:2004, Medical Device Software Risk Management and Guidance for Industry and FDA Staff, Total Product Life Cycle: Infusion Pump-Premarket Notification (510(k)) Submissions, Draft Guidance, Guidance for Industry and FDA Staff: Guidance for the Content of Premarket Submissions for Software Contained in Medical Devices, or similar updated documents as an international standard; (3) standardized algorithm documentation for submission of stand-alone insulin dosing calculators, as well as those that are a constituent of a medical device; (4) a worldwide database of registered products to improve regulatory transparency around the world, as well as to monitor and track device safety throughout the device life cycle; and (5) avoidance of required standardization of insulin dosing algorithms to preclude the stifling of scientific and technologic advances in the area. 10
Traditionally FDA includes a benefit–risk determination in deciding whether to clear a product, such as bolus software. On March 28, 2012, FDA issued a document on this topic: “Guidance for Industry and Food and Drug Administration Staff—Factors to Consider When Making Benefit-Risk Determinations in Medical Device Premarket Approvals and De Novo Classifications.” In it FDA explained that any probable benefit to health from the use of a device is weighed against any probable risk of injury or illness from such use. 11
The FDA patient Preference Initiative was launched in 2013 to identify and develop methods for assessing patient valuations of benefit and risk related to specific device types and specific illnesses and conditions. 12 This emphasis on patient involvement was in evidence at the recent Public Workshop—Regulatory Science Considerations for Software Used in Diabetes Management, November 13, 2014, at FDA Headquarters. 13 The purpose of this meeting was to seek input from the clinical community, academia, government, industry and other stakeholders regarding the makeup of the intended use population for insulin bolus calculators, and the technical considerations for their design and use. The views of patients were carefully considered through patient participation in the discussion and public comment by patients. Many participants expressed the view that they look to FDA more to regulate safety than effectiveness for insulin dosing software. On January 8, 2015, FDA announced that in 2015 they plan to develop a draft guidance for medical device decision support software. 14
On February 9, 2015, FDA released a guidance on Medical Device Data Systems (MDDS). 15 FDA stated that this agency does not intend to regulate hardware or software products that transfer, store, and convert formats, medical device data, or convert information produced by a different device. This guidance specifically mentioned that BG monitors transmitting glucose data remotely will be exempted from regulation, but it did not intend to exclude insulin dosing software from regulation.
Meanwhile the future of FDA regulation of insulin dosing software might be affected by pending legislation. In January 2015 the US House of Representatives Energy and Commerce Committee released an initial discussion document outlining proposals of its 21st Century Cures initiative to modernize medical research and medical product regulation. The plan is to pass this legislation in Congress and have it signed by the President this year. The draft bill contains the latest version of the Sensible Oversight for Technology Which Advances Regulatory Efficiency (SOFTWARE) Act, which was first proposed in October 2013. The SOFTWARE Act would distinguish 2 types of medical software: medical software, which would be regulated by FDA and health software, which would remain unregulated. The draft bill would require FDA to develop a new regulatory program to oversee medical software within 2 years of the bill’s passage.16,17
Conclusions
Increasingly more types of legal insulin dosing software are being developed and many types will be coming to the United States in the next few years. The future availability of such applications will depend on how safe, effective, and convenient these products will be as well as how much regulation or how little regulation is demanded of FDA by the public and by the US Congress.
Footnotes
Abbreviations
BG, blood glucose; FDA, US Food and Drug Administration; MDDS, Medical Device Data Systems; SOFTWARE, Sensible Oversight for Technology Which Advances Regulatory Efficiency.
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant for Google, Insuline, Lifecare, Roche, Sanofi, Tempramed, and Voluntis. He is also a stockholder in Tempramed.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
