Abstract
Background:
The most efficacious strategies to improve diabetes control include case management, health care team changes, patient education, and facilitated transmission of patient data to clinicians (“facilitated relay”), but these strategies have not been translated to permit general use in clinical practice.
Methods:
A web-based decision support program was developed to include these features, and assessed in patients who had A1c ≥7.0% despite using metformin with/without sulfonylureas or insulin. Staff entered patients’ glucose data, obtained management recommendations, reviewed the plan with a clinician, and discussed the new plan with patients.
Results:
113 subjects were 96% male and 32% black, with average age 65.6 years and BMI 32.8. During prior primary care, A1c averaged 8.32 ± 0.16% (SEM). In all patients, baseline A1c was 8.18 ± 0.11%, and decreased to 7.54 ± 0.12%, 7.16 ± 0.13%, and 7.54 ± 0.16% at 3, 6, and 12 months, respectively, all P < .001. In 42 subjects who provided glucose data and made requested changes in medications, A1c was 8.12 ± 0.09% at baseline and fell to 7.29 ± 0.11%, 6.98 ± 0.10%, and 7.05 ± 0.10% at 3, 6, and 12 months, respectively, all P < .001. Chart review of 16 subjects followed for 12 months demonstrated that hypoglycemia (symptoms and/or glucose <70 mg/dl) averaged less than 1 episode/patient/month, and there was no severe hypoglycemia.
Conclusions:
A novel decision support program improved A1c with little hypoglycemia. Use of this approach should allow primary care teams to keep patients well controlled, and reduce the need for specialist referrals.
Keywords
Although good metabolic control can reduce the risk of diabetes complications and associated costs, 1 many patients have glucose levels above goal, and hypoglycemia often complicates treatment. 2 A recent meta-analysis has shown that the most efficacious strategies to improve glucose control were case management, health care team changes to provide a role for team members in addition to the primary provider, patient education, and facilitated transmission of patient data to clinicians (“facilitated relay”). 3 However, these approaches have not been combined and translated as needed to permit widespread use.
Translation of diabetes management guidelines can also be hindered by limitations such as (1) lack of specificity in directing management of individual patients, (2) lack of algorithms that emphasize widely used, inexpensive drugs, and (3) difficulty in matching pharmacotherapy with clinical need. For example, therapy should be intensified if A1c levels are high and patients have characteristics which justify more intensive management, 4 but guidelines are not individualized.5,6 The guidelines include multiple choices of drugs, and there are no specific dosage recommendations for individual patients. Primary care providers (PCPs), who provide most diabetes care, often have difficulty navigating the guidelines and algorithms,7,8 and especially in using insulin;9-13 guideline use in primary care is infrequent, and may not lower A1c to goal.14,15 The result is delay in intensifying therapy (“clinical inertia”16,17), inadequate glucose control,18,19 worse health (from diabetes complications), 20 higher costs, 21 and frequent hypoglycemia. 2
Moreover, if patients are using drugs that can cause hypoglycemia, 22 home glucose monitoring can be essential to improving management safely in individual patients, by indicating needs for therapy and risk of hypoglycemia at different times of the day. However, there is often little emphasis in the guidelines on identifying situations where home glucose monitoring should be used, analyzing glucose monitoring data can be labor intensive, and PCPs may have had limited training and experience in using glucose monitoring to guide treatment. As a result, glucose monitoring may not be used adequately; monitoring needs to be structured—to show when treatment should be intensified or reduced—to minimize the risk of hypoglycemia. Although PCPs rarely use monitoring in this way,15,23 PCPs can be trained to use such data to improve A1c levels24,25—but still often make incorrect therapeutic decisions in management of fasting and postprandial hyperglycemia. 26
To achieve translation, we have developed a web-based decision support program which includes the efficacious features for improving glucose control and avoids the common problems which may limit translatability (above). Here we report a proof-of-concept evaluation of efficacy and safety with this approach.
Research Design and Methods
Patients
The decision support program was evaluated with a before-and-after design in outpatient veterans at the Atlanta VA Medical Center. All had baseline A1c levels ≥7.0% despite use of metformin with or without use of sulfonylureas or insulin, and were participants in a trial in which they were randomized, double-blind, 1:1, to receive once-weekly injections of exenatide or placebo. The program was utilized for all participants, as which patients would be receiving active or inactive study drug was unknown; patients were advised that their management would be optimized. The primary study was approved by the Emory University Institutional Review Board and the Atlanta VA Research and Development Committee, and all patients provided informed consent.
Decision Support
All patients were given hard copy forms on which to record glucose levels and use of glucose-lowering medications. Patients were initially instructed to record their prebreakfast glucose levels daily, and pre- and postprandial and bedtime glucose levels (7-point profile) once a week. They were asked to call or fax information to study staff, and to inform staff if there was hypoglycemia. If control improved, glycemic assessment became less frequent.
Study staff entered the glucose values and medication usage into a web-based program, which used algorithms to evaluate metabolic control and recommend changes in management, as needed; the algorithms were designed to mimic the decision making of an experienced endocrinologist. The algorithms included “goal” glucose levels 75-100 mg/dl before breakfast, 80-120 mg/dl before lunch and supper, and 110-150 mg/dl at bedtime. Once metformin dosage was maximized at 2000 mg/day, non-long-acting glipizide was added as needed before meals and/or at bedtime, and dosage changed in increments of 2.5 mg. If glipizide dosage reached 40 mg/day or 20 mg at bedtime, glargine insulin was added next, beginning at a dosage of 0.15 mg/kg body weight, and given before the evening meal to minimize the risk of nocturnal hypoglycemia. Decision support focused on these medications because they are widely used, inexpensive, and available in the VA formulary, but carry a risk of inducing hypoglycemia. The algorithms decreased the recommended dosage of medications if there was hypoglycemia (symptoms or recorded glucose levels <70 mg/dl) or variability in recorded glucose levels, or if a low glucose level was predicted. The algorithms also provided “rationales”—justifications for the recommendations.
Health Care Team Changes, Case Management, Facilitated Relay, and Patient Education
The decision support program was designed to take advantage of the capabilities of different health care team members. While the final management plan remains the responsibility of the clinician, other team members such as the registered nurse (RN) participate in important ways. The use of decision support constitutes case management, and transmission of patient data and management recommendations to the clinician constitutes facilitated relay. Patient education can be provided by staff in discussion of both the glucose data, and the rationale for revised management—including opportunities for education about the impact of diet and physical activity on glucose levels.
Medications
Patients obtained routine diabetes medications (metformin, glipizide, and glargine) through the VA; a small number of patients (less than 5 in each case) required use of alternative medications such as sitagliptin, pioglitazone, repaglinide, or detemir, which were also obtained through the VA.
Measurements
Hemoglobin A1c was measured in the Atlanta VA chemistry laboratory with the Tosoh G7 or later versions of this equipment (Tosoh Bioscience, South San Francisco, CA). Freestyle glucose meters, test strips, and related equipment (Abbott Diabetes Care, Alameda, CA) were provided to all participants through the VA.
Analysis
Control data consisted of each patient’s A1c levels during 12 months of primary care prior to study enrollment, which were obtained from the VA’s electronic medical record (Computerized Patient Record System, CPRS). A1c was also measured at intervals of 3-6 months during the study; since A1c was not a primary endpoint of the primary study, all patients did not have A1c measured at the same intervals. A1c values reported at 3 month intervals reflected all measurements during that period, and values were averaged if subjects had more than one measurement. The primary analysis was intention to treat, involving all 113 participants and comparing A1c levels at enrollment to levels at different times during the study. We also performed a per protocol analysis in 42 participants who provided glucose monitoring data and followed management recommendations as requested for 12 months. In addition, a manual chart review to assess hypoglycemia was performed for the first 16 participants out of the 42; hypoglycemia was defined as a report of typical symptoms and/or glucose monitoring documentation of glucose levels below 70 mg/dl. Differences in A1c levels were evaluated using t tests, and all analyses utilized SAS® statistical software (version 9.2; SAS Institute, Cary, NC).
Results
Demographic characteristics are shown in Table 1. The full group of 113 patients was 96% male and 32% black, with average age 65.6 years and BMI 32.8 kg/m2. The subgroups of 42 adherent patients, and the 16 patients whose records were reviewed to assess frequency of hypoglycemia had baseline characteristics that did not differ from those of the full group.
Demographics and A1c Values for 3 Groups of Subjects: All 113 Subjects, 42 More Adherent Subjects, and the 16 Subjects Whose Charts Were Reviewed for Details About Hypoglycemia.
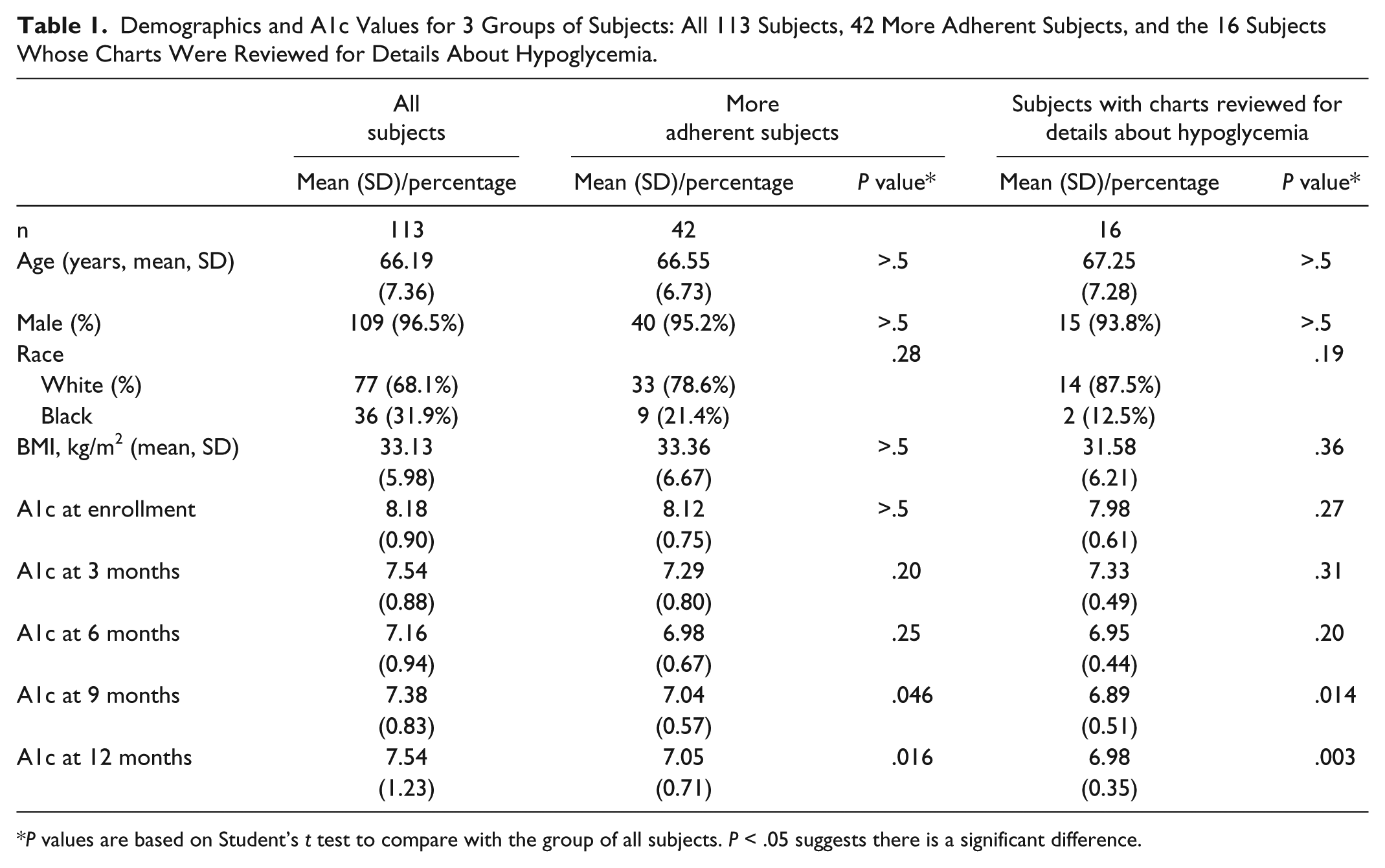
P values are based on Student’s t test to compare with the group of all subjects. P < .05 suggests there is a significant difference.
An illustrative patient is shown in Figure 1. The patient was initially taking both metformin and pioglitazone, along with glipizide. With decision support, the glipizide dosage was readjusted and glucose levels fell, but glucose was still above goal, so glargine insulin was begun. As the glargine dosage was increased, the glipizide dosage was lowered progressively (to avoid hypoglycemia), and most of the later glucose levels were in goal range. Delayed initiation of basal insulin is a common problem in primary care,9-13 and this patient shows how decision support can assist in initiation and safe titration.
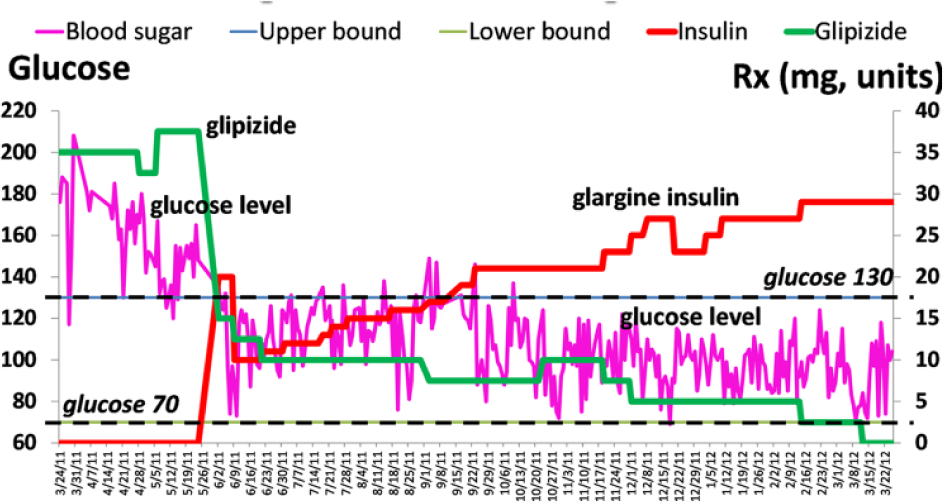
Illustrative example. Over a period of about a year, dosages of non-long-acting glipizide are shown as mg/day in green, dosages of glargine insulin are shown as units/day in red, and the 3-day rolling average of home glucose monitoring levels is shown in purple.
A1c levels during the year prior to study initiation—with management in primary care—were 8.21 ± 0.15% at -12 months, 8.28 ± 0.18% at -9 months, 8.37 ± 0.15% at -6 months, and 8.32 ± 0.14% at -3 months (mean ± SEM), respectively. A1c at study initiation was 8.18 ± 0.11%, slightly but not significantly lower than A1c levels at -12, -9, -6, and -3 months prior to baseline (all P > .05).
With decision support, A1c levels improved, and in all 113 patients (Figure 2), were significantly lower than levels at baseline at +3, +6, +9, and +12 months (all P < .001). Since some patients would not monitor their glucose levels as requested and/or make recommended changes in their medications, we also performed a per protocol analysis in 42 adherent patients. This group had baseline A1c levels of 8.12 ± 0.12% (P = ns vs the full group), and as shown in Figure 3, had A1c levels at +9 and +12 months that were significantly lower than those of the full group at 9 months and 12 months (both P < .05).
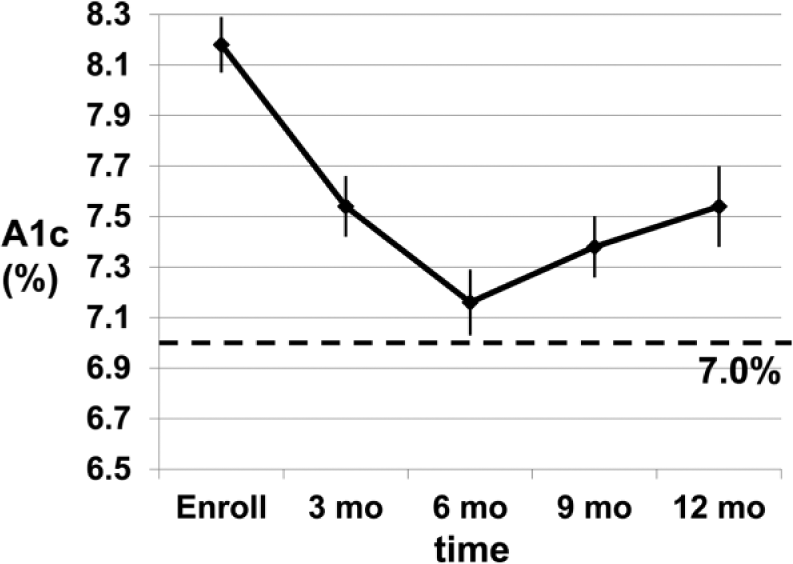
Levels of A1c in all subjects (n = 113). Mean ± SEM.
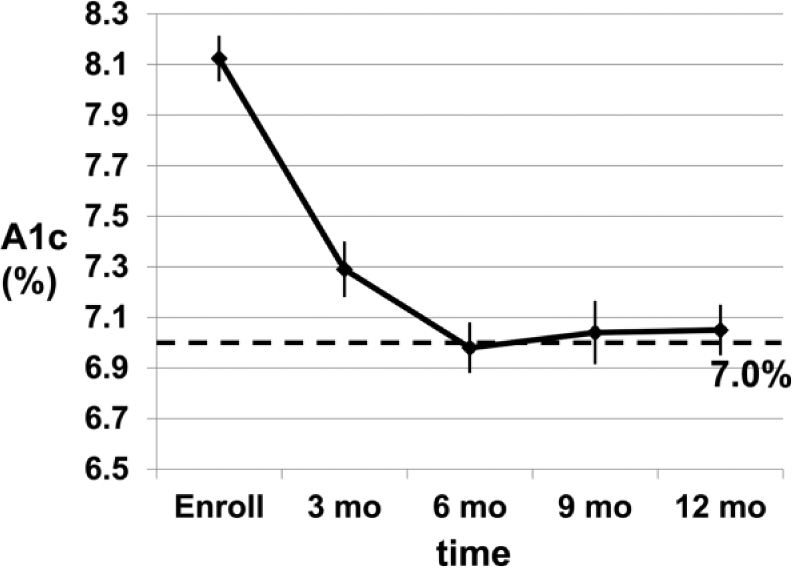
Levels of A1c in more adherent subjects, all of whom provided glucose monitoring data as requested and made requested changes in dosages of medication (n = 42). Mean ± SEM.
Hypoglycemia was assessed by chart review in the first 16 adherent patients. Their baseline A1c was 7.98 ± 0.08%, and fell to 7.33 ± 0.06%, 6.95 ± 0.06%, 6.89 ± 0.07%, and 6.98 ± 0.05% at +3, +6, +9, and +12 months, respectively (not shown), also lower than values in the full group at 9 and 12 months (both P < .05). Despite the improvement in A1c levels, recorded hypoglycemic symptoms and/or home glucose monitoring values <70 mg/dl averaged less than 1 episode per month (Figure 4).
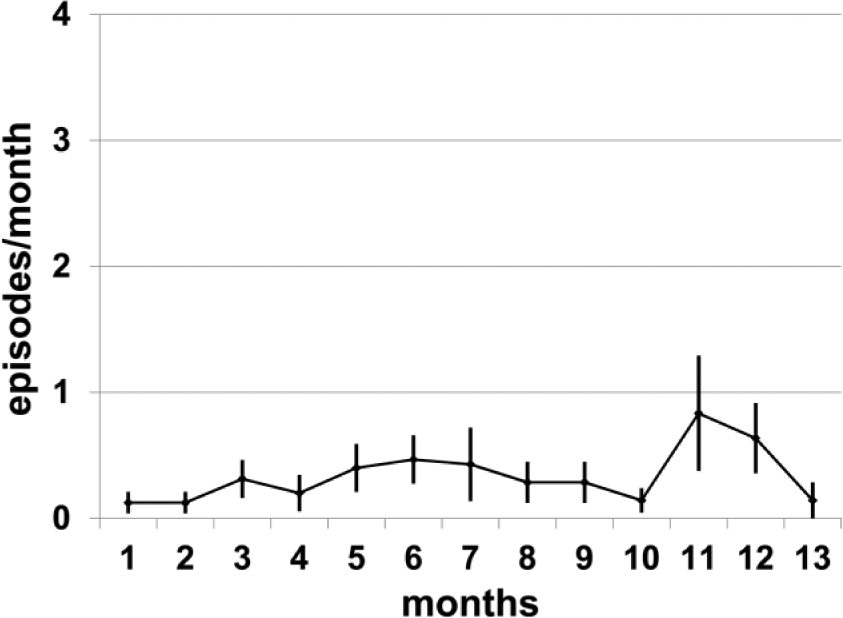
Hypoglycemia (recorded home glucose monitoring values and/or hypoglycemic symptoms) in adherent subjects whose charts were reviewed to ascertain details about hypoglycemia (n = 16). Mean ± SEM.
Discussion
We evaluated a decision support program designed (1) to incorporate 4 strategies which have been shown to be effective in improving glycemic control—health care team changes, case management, patient education, and facilitated relay of patient data to clinicians 3 and (2) to overcome limitations which often hinder translation of diabetes management guidelines—lack of specificity in the guidelines, inadequate use of glucose monitoring to guide treatment, analysis of monitoring data being labor intensive, lack of algorithms designed for widely used, inexpensive drugs, and difficulty in matching pharmacotherapy with clinical need. Prior to implementation of the program, 113 patients managed in primary care had A1c levels averaging 8.18%. With decision support, utilizing mainly metformin, non-long-acting glipizide, and glargine insulin, A1c fell to 7.16% after 6 months, and rose slightly to 7.54% at 12 months. In 42 adherent patients, A1c fell from 8.12% to 6.98% at 6 months, and remained near this level for 12 months. Hypoglycemia (symptoms and/or glucose <70 mg/dl averaged less than one episode per patient per month in 16 patients whose charts were reviewed. These findings demonstrate that this decision support program can improve A1c levels with little hypoglycemia.
There has been extensive promulgation of diabetes management guidelines 27 and use of hard-copy and computerized decision support in attempts to improve diabetes management.28-41 Involvement of patients without decision support may not lower A1c levels,28,29 but decision support with personalization (based on individual patients’ clinical markers and medications)34,35,37 may be more effective than that without personalization;30-33,36 recent reviews found effectiveness to be aided by inclusion of feedback or case management, 40 or “forcing” providers to justify overriding recommendations. 41 However, improving diabetes management has been difficult. As reviewed recently, 42 National Health and Nutrition Examinations Surveys (NHANES) from 2003-2006 and 2007-2010 found slight improvements in the percentage of US adults with diagnosed diabetes who had A1c levels >9% (decreasing from 13.0% to 12.6%) and those who had A1c levels <8% (increasing from 78.0% to 79.1%), but there was no improvement in those with A1c <7.0% (decreasing from 56.8% to 52.2%). A more recent analysis 43 of NHANES 2007-2012 data, with individualized A1c goals based on considerations of age and the presence or absence of complications, found that 63.7% of US adults with diagnosed diabetes met A1c goals. Problems in management appear to include both clinical inertia 16 and health disparities. 44 Although diabetes management in the VA has been reported to be better than in conventional managed care, 45 we found in veterans in the southeastern United States that the diagnosis was usually not guideline-based, 46 and that medications were started late—particularly in black veterans. 44 In a recent study, the mean A1c when insulin was initiated was over 8.0% and higher in 2010 than 2005. 47
While most health care providers know that diabetes management is important, they may be uncertain as to exactly what to do for individual patients, they may be appropriately concerned that initiating and intensifying therapy with sulfonylureas and insulin can lead to problems with hypoglycemia, 22 and they are very busy. As a result, guideline recommendations and computer programs may fail if they require a provider to choose among different options, and/or to have to invest considerable time in using the system. This may be why despite the availability of guidelines, management algorithms, computer programs, and reports showing the efficacy of different strategies for improving diabetes management, there has been no general adoption into practice. Such considerations prompted design of our program to incorporate features not found in most existing guidelines, or computer/decision support programs. Our program is aimed at health care teams rather than providers, it provides individualized recommendations (drugs, dosages, and times of day) and rationales for the recommendations, and it adjusts for hypoglycemia and glucose variability. (Increased A1c variability was associated with increased mortality in the Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation [ADVANCE] trial, 48 and may have increased mortality and led to early termination in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study. 49 ) Our program also promotes a relatively new use for an old drug; the underlying algorithms use metformin first, start insulin early, and use small dosage increments of non-long-acting glipizide before meals and at bedtime. While sulfonylureas are generally used only once or twice a day, our approach appears to be effective and safe—with minimal risk of the hypoglycemia typically associated with these drugs. 2
Although our study demonstrates both efficacy and safety, there are also limitations. Since the study utilized a before-and-after design and was not a randomized, controlled trial with a comparator group with a more traditional approach to medication adjustment, we cannot be sure that the patients’ glycemic control would not have improved if they had continued to be managed in primary care. However, the lack of any trend for such improvement during a prior year of primary care makes such an alternative hypothesis unlikely. The primary patient contacts were research staff rather than health care team staff, but the research staff involved included coordinators and a graduate student as well as a nurse, and all had little prior experience in diabetes management. The research staff did have more time to devote to patient education than might be available in typical health care settings, which might tend to reduce generalizability; strategies to promote efficient use of time might be needed to facilitate broad use in clinical practice. The study was conducted in a VA setting and largely involved male veterans, but the few female veterans who participated appeared to respond as well as the men. Since the managing clinician in this study was an endocrinologist, we cannot be sure that PCPs will be comfortable in working with their health care team to manage their patients’ diabetes, and further studies will be required to determine how best to implement use of the program. Adherence was also less than uniform in this pragmatic study, but the improvement in A1c levels in all 113 patients was still both statistically and clinically significant. Some of the patients would have received long-acting exenatide (the primary study is still ongoing, double-blind), which may have contributed to improvement in A1c levels. However, the improvement in A1c was relatively uniform across study participants, greater A1c lowering in the adherent patients is consistent with an impact of decision support (Figure 3 vs Figure 2), and safe addition of either exenatide or insulin would have required decreasing the dose of sulfonylureas as shown in Figure 1, demonstrating the need for decision support. (Since the intervention was multifactorial, we cannot assess the contributions of decision support per se.)
Finally, while hypoglycemia was infrequent, we were only able to evaluate hypoglycemia in 16 subjects, and we cannot be sure that hypoglycemia will not be more of a problem with use of sulfonylureas and insulin in larger and longer studies. While use of non-long-acting glipizide and long-acting insulin carries a risk of hypoglycemia, episodes in our study were all mild and easily corrected with reductions in dosage of the medication; it is possible that increased early morning insulin resistance due to the “dawn phenomenon” 50 helps to decrease the tendency of these medications to produce nocturnal hypoglycemia. After the study was completed, one subject taking glargine insulin twice daily did experience severe hypoglycemia prelunch when the morning dose was not reduced as recommended despite an anticipated increase in physical activity—demonstrating the importance of moderating the A1c goals in such patients, as recently emphasized by Cryer. 22
Conclusions
We demonstrate that a novel computer-assisted decision support program which incorporates features shown to be effective in improving metabolic control—health care team changes, case management, patient education, and facilitated relay of patient data to clinicians—can reduce patients’ glucose levels with little risk of hypoglycemia. While additional research is needed to establish the utility of this approach in different health care settings, it seems likely that this program should be widely applicable—helping health care teams to keep their patients with diabetes well controlled in primary care, and possibly reducing the need for specialist referrals as well.
Footnotes
Acknowledgements
The authors gratefully acknowledge the contributions of Gordana Pajkovic, MS, and Mari Hart, RN, MSN, CDE, research staff who initially had little experience in diabetes management, but interacted with study patients to obtain glucose data and implement plans for management, used the decision support system, and through their many recommendations improved both the decision support system and their patients’ care. LSP (Atlanta VA Medical Center and Emory University School of Medicine) had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. This work was presented in part at the American Diabetes Association meeting in June 2014.
Abbreviations
ACCORD, Action to Control Cardiovascular Risk in Diabetes study; ADVANCE, Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation trial; A1c, hemoglobin A1c; CPRS, Computerized Patient Record System; NHANES, National Health and Nutrition Examination Surveys; PCP, primary care provider; RN, registered nurse; VA, Veterans Administration
Declaration of Conflicting Interests
The author(s) declare that there is potential duality of interest associated with this manuscript: With regard to potential conflicts of interest, within the past several years, LSP has served on Scientific Advisory Boards for Boehringer Ingelheim and Janssen, and has or had research support from Merck, Amylin, Eli Lilly, Novo Nordisk, Sanofi, PhaseBio, Roche, Abbvie, Vascular Pharmaceuticals, and the Cystic Fibrosis Foundation. In the past, he was a speaker for Novartis and Merck, but not for the past several years. DEO has research support from Novo Nordisk and Amylin, and QL receives support from NIH, PCORI, and the Cystic Fibrosis Foundation and has served as a consultant for Eisai. SLJ received support from Amylin. These activities involve diabetes, but have nothing to do with this manuscript. After the study reported here was completed, 2 of the authors (LSP and CY) founded a company, Diasyst LLC, which aims to develop and commercialize diabetes management programs. Other authors have no potential conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by FDA award RO1FD003527 (LSP), VA award HSR&D IIR 07-138 (LSP, SLJ), NIH awards DK066204 (LSP), U01 DK091958 (LSP and MKR), U01 DK098246 (LSP and DEO), R21 DK099716 (LSP, QL, and SLJ), and a Cystic Fibrosis Foundation award PHILLI12A0 (LSP). It is also supported in part by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR000454. The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript. MKR, DB, LSP, DEO, and AMT are supported in part by the VA. This work is not intended to reflect the official opinion of the VA or the US government.
