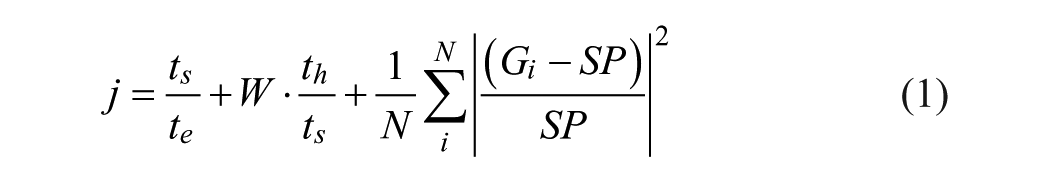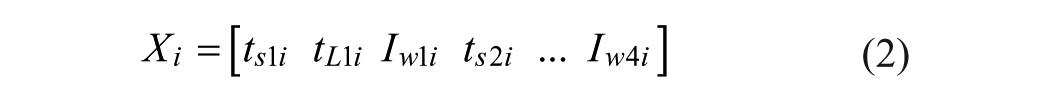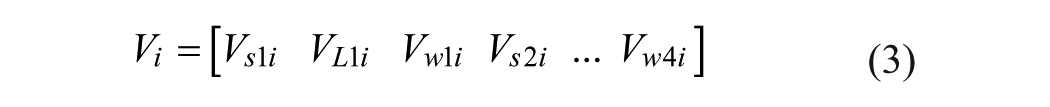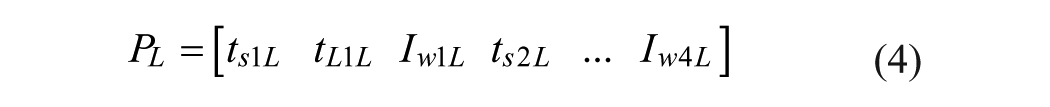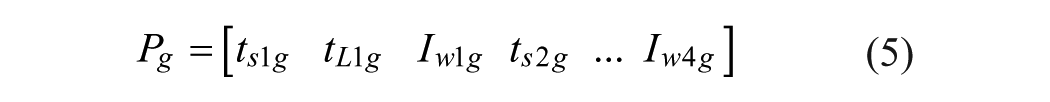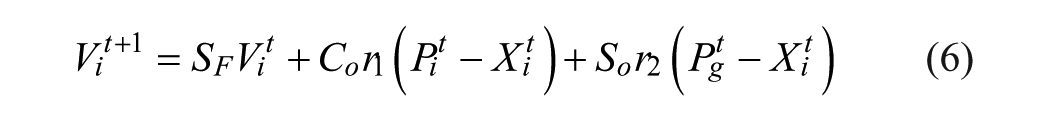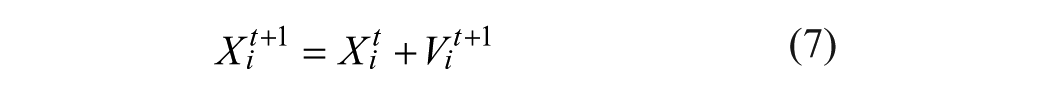Abstract
Background:
Maintaining euglycemia for people with type 1 diabetes is highly challenging, and variations in glucose absorption rates with meal composition require meal type specific insulin delivery profiles for optimal blood glucose control. Traditional basal/bolus therapy is not fully optimized for meals of varied fat contents. Thus, regimens for low- and high-fat meals were developed to improve current insulin pump therapy.
Method:
Simulations of meals with varied fat content demonstrably replicated published data. Subsequently, an insulin profile library with optimized delivery regimens under open and closed loop for various meal compositions was constructed using particle swarm optimization.
Results:
Calculations showed that the optimal basal bolus insulin profiles for low-fat meals comprise a normal bolus or a short wave. The preferred delivery for high-fat meals is typically biphasic, but can extend to multiple phases depending on meal characteristics. Results also revealed that patients that are highly sensitive to insulin could benefit from biphasic deliveries. Preliminary investigations of the optimal closed-loop regimens also display bi- or multiphasic patterns for high-fat meals.
Conclusions:
The novel insulin delivery profiles present new waveforms that provide better control of postprandial glucose excursions than existing schemes. Furthermore, the proposed novel regimens are also more or similarly robust to uncertainties in meal parameter estimates, with the closed-loop schemes demonstrating superior performance and robustness.
Keywords
Type 1 diabetes mellitus (T1DM) is a chronic condition caused by autoimmune destruction of pancreatic β-cells. 1 Without adequate insulin secretion, the body cannot metabolize blood glucose (BG). Since glucose is the main energy source for the body, maintaining euglycemia, or normal BG, is essential to avoid serious complications associated with hypo- and hyperglycemia. 2 In people with T1DM, rapid increases in BG from meals are difficult to control due to the lack of cephalic insulin delivery prior to consumption. Consequently, meal compensation is a main challenge in BG control.
T1DM is commonly treated with continuous subcutaneous insulin injections (CSIIs), 3 combined with user calculated insulin boluses prior to meals. Insulin pump therapy, a CSII system, performs automatic calculations of insulin dose required to cover ingested carbohydrates (CHO). 4 Automated closed-loop insulin delivery, referred to as artificial pancreas (AP), will ideally eliminate the need for user inputs. Several control algorithms for AP have been proposed, such as model predictive control (MPC),5-10 proportional-integral-derivative (PID) control,11-14 and fuzzy logic control.15,16 Limitations of current insulin therapy mean that novel delivery regimens to obtain optimal postprandial BG control that can compensate for intervarietal differences in meal absorption rates are needed.17-20
Following early efforts in mixed meal modeling, 21 compilations of different absorption patterns for meals with varying fat content were developed to cover wide disparities in insulin compensation requirements.22,23 Although combining such a meal library with a controller within the loop were explored, 23 meal compensation beyond basal bolus therapies has been limited. Insulin advisory software,4,24 called “bolus wizards,” 25 were tested under clinical conditions, but these merely facilitate the derivation of bolus amounts and still fundamentally depend on impulse deliveries. Current insulin delivery profiles are limited to single boluses (instantaneous insulin dose) and/or dual waves (instantaneous dose with a prolonged wave-like delivery). Single boluses work well for fast absorbing meals, but are not optimal for slower absorbing high-fat meals due to increases in risk of early hypoglycemia. While some studies suggest dual wave insulin delivery improves BG control following slow absorbing meals, 1 optimal insulin delivery regimens for these meals have not been verified. Figure 1 compares the simulated representative performances of various insulin regimens following a high-fat meal to illustrate the need of better control protocols.
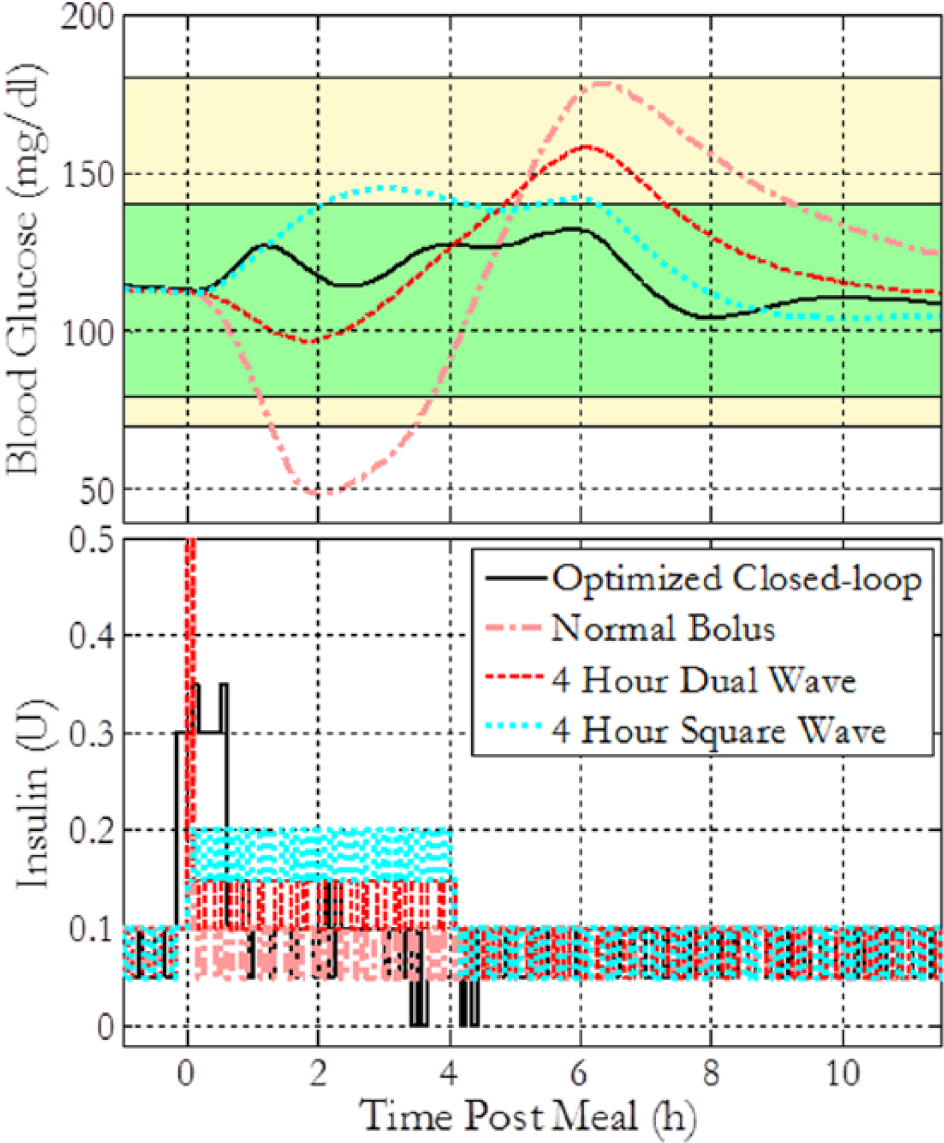
Simulated blood glucose trajectories and insulin profile comparison between a novel optimized insulin regimen under closed-loop control and optimized proportional-integral-derivative controller as referenced in the fifth section of this article, normal single bolus, dual wave (50% of the full bolus given initially, followed by 50% given as a remainder), and square wave regimens following a 100 g CHO high-fat pasta meal for 1 virtual subject. The green shaded region represents the 80-140 near-euglycemic zone, and the yellow region represents the zone between 70-180 hypo- and hyperglycemic thresholds. The insulin profile subplot has been zoomed to facilitate view.
As can be seen in Figure 1, the single bolus strategy causes a significant hypoglycemic event for meals with high fat content. Four-hour dual and square waves perform significantly better in avoiding hypoglycemia, but still fall short of the optimal basal bolus scheme, discussed in the present work, in minimizing total BG excursion beyond the target of 110 mg/dl. Traditional basal bolus and closed-loop AP therapies with user announcements were studied in the present work. Novel insulin delivery profiles were identified through in silico trials for multiple meal compositions using particle swarm optimization (PSO). 26
Simulation of Meal Profiles
The present work was performed using the University of Virginia/Padova (UVA/Padova) metabolic simulator.27,28 This simulator reproduces the variations in insulin action, blood glucose dynamics, and meal absorption parameters seen in the general population, and allows for testing of a variety of scenarios utilizing subcutaneous insulin delivery using CSII pumps and sensing of BG with continuous glucose monitors (CGMs).
This study utilizes 10 in silico adult subjects included in the distributed version of the simulator for investigational purposes. Preliminary investigations using the given simulator have been accepted by the FDA in lieu of animal trials prior to multiple clinical trials. An auxiliary meal delivery rate was introduced to the simulator to enable simulation of slow absorbing high-fat meals. The auxiliary meal delivery rate was tuned to qualitatively replicate postprandial blood glucose profiles in literature. Simulated profiles for the 2 high-fat meals studied were generated under similar conditions as the published data, considering the time of the postprandial peak as the criterion for a good match. The mean blood glucose profiles of 10 subjects were compared with published data from Jones et al 29 and Normand et al. 17
Jones et al 29 investigated 26 adults with T1DM to determine the optimal insulin regimen for a pizza meal. The subjects consumed 3 pizza slices with approximately 90 g of CHO and 42 g of fat. Insulin was delivered as a bolus, a 4- or 8-hour dual wave. Normand et al 17 investigated 9 healthy women to determine the influence of dietary fat on postprandial glucose metabolism. One meal consisting 75 g of pasta with a fat content of 55% was chosen from this study for in silico evaluation.
Each reported scenario was simulated under subject specific optimal basal bolus (open loop) insulin therapy within the UVA/Padova simulator. The meal delivery rates were optimized based on least squares difference between simulated and literature reported time and value of the postprandial peak given meals with reported CHO and fat content for each scenario. The resulting optimized meal delivery rates are shown in Figure 2.
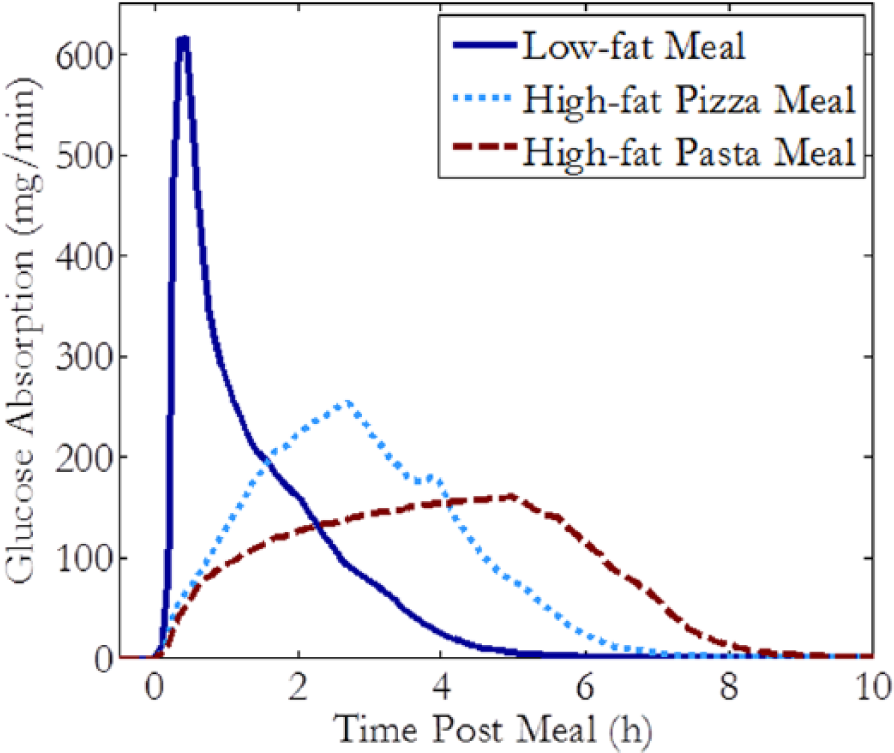
Ten subject average glucose absorption profiles for 3 different meal types, each containing 50 g of CHO.
The modified simulator with optimized meal delivery rates qualitatively replicate published clinical data. Figure 3 shows that altering the meal delivery rate made possible the replication of the time to postprandial peak for both medium-fat pizza meals and high-fat pasta meals. Although there is a qualitative mismatch in the magnitude of the glucose excursion for the high-fat pasta meal, the time of the peak for both meal types conform to published data and is of greater interest for insulin regimen optimization as this can determine the optimal timing of the insulin deliveries.
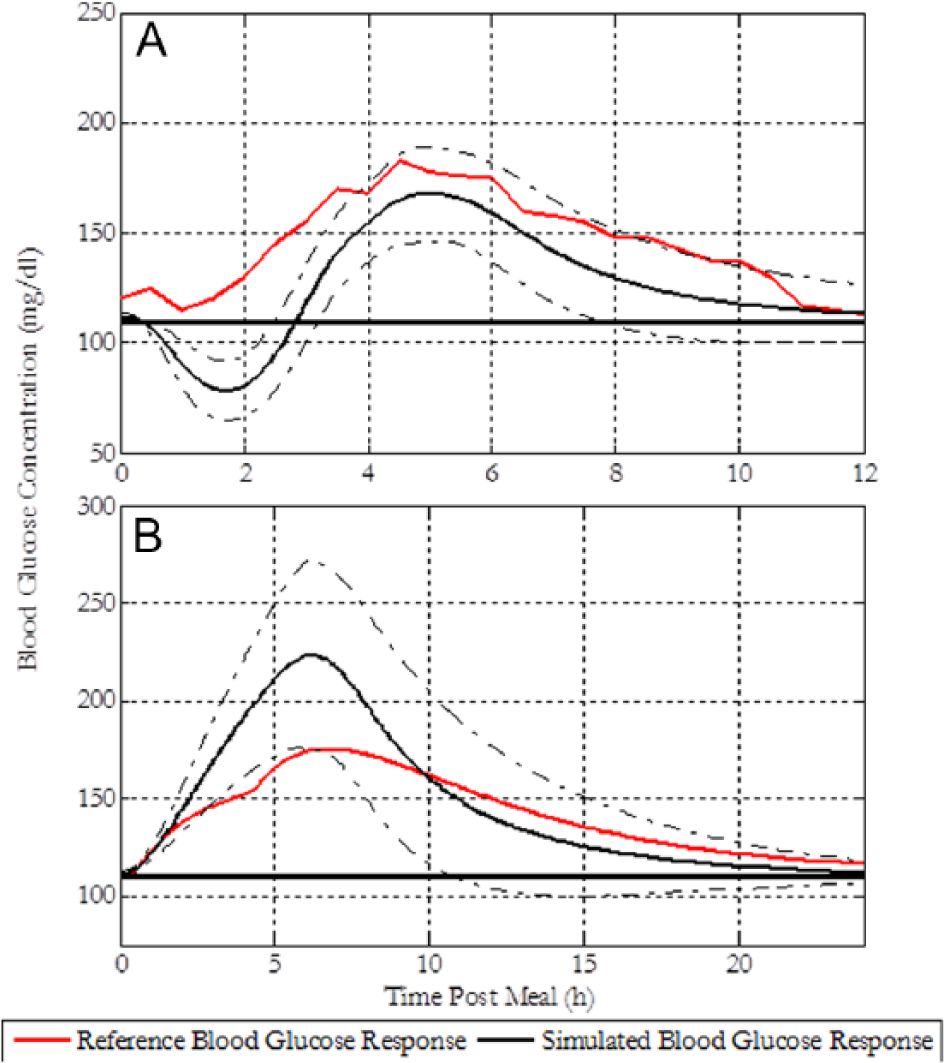
(A) Comparison of simulated 10 subject average blood glucose responses to a 90 g CHO pizza meal and published data from Jones et al 29 given a full insulin bolus at the time of the meal. (B) Comparison of simulated 10 subject average blood glucose response profiles for a 63.4 g CHO high-fat pasta meal and a simulated reference profile based on in silico data from Herrero et al. 22 The dashed lines represent min-max profiles, and the solid line at 110 represents the blood glucose concentration at the start of meal. The scenarios were simulated under basal insulin therapy only.
Global Optimization Approach
Novel insulin delivery profiles are needed to improve postprandial BG control and provide appropriate bolus recommendations for a wide variety of meals. Delivery options are currently limited to normal, dual wave, and square wave boluses. As meal absorption rates are influenced by its composition, insulin regimens must be altered accordingly for optimal control. Novel delivery profiles for different meals were formulated as an optimization problem.
The problem was designed as a multiple delivery scheme with 3 optimization variables for each insulin delivery: the time of delivery, the delivery duration, and the fraction of the full dose. The regimen’s performance was characterized by the cost function in Equation 1. te and th are time in near-euglycemia (80-140 mg/dl) and hypoglycemia (below 70 mg/dl), respectively. ts represents total simulation time and Gi denotes ith recorded average BG concentration. N represents the number of sample points in simulated response (12 data points per hour, 12 hours for a total value of 144), chosen to normalize the summed square of deviations of blood glucose from the set point to an order 1.
The cost function is designed to maximize time in euglycemia, minimize time in hypoglycemia, and reduce deviations from the baseline SP of 110 mg/dl, each scaled to be unitless and of order 1. Hypoglycemia can have life-threatening short-term consequences,
2
and was weighted 5 times the other terms (ie,
Xi represents current position of the particle in parameter space, Vi is the current velocity of the particle in each dimension, PL is the locally best found position, and Pg is the globally best found position. The position of each particle was then updated according to Equations 6 and 7,
where
The PSO algorithm was implemented in this manner to find the best individualized/generalized insulin delivery regimens for meals of different CHO and fat content during open and closed loop. Prior to the identification of delivery regimens under closed loop, tunable parameters for the simple controller utilized during the closed loop were also identified based on these settings.
Optimized Basal Bolus Regimens
General optimal insulin regimens (GORs) for 10 in silico subjects were found using PSO for 9 different scenarios. A total meal CHO of 50, 75, or 100 g and low, medium, or high fat content was specified for each scenario. These scenarios were designed to cover the range of variability in meal content seen in the daily life. In the context of this study, a GOR is a recommended set of insulin deliveries based on any subject’s I:C ratio—for instance, the insulin regimen for typical meal compensation can be described as a GOR where a bolus amounting to 100% of the quantity as calculated by the I:C is given at the time of meal. Three different meal types with varying absorption rates were considered for the optimization, and each type was simulated with 50, 75, and 100 g carbohydrate content. For each meal scenario, a GOR was acquired through a combined optimization for 10 in silico subjects. Similarly, individual optimal regimens (IORs) were also determined for each scenario. They are, unlike GORs, individually tailored regimens obtained through separate optimizations for each of the 10 subjects, utilized to compare the validity of each GOR. A summary of the GOR results is presented in Figure 4.
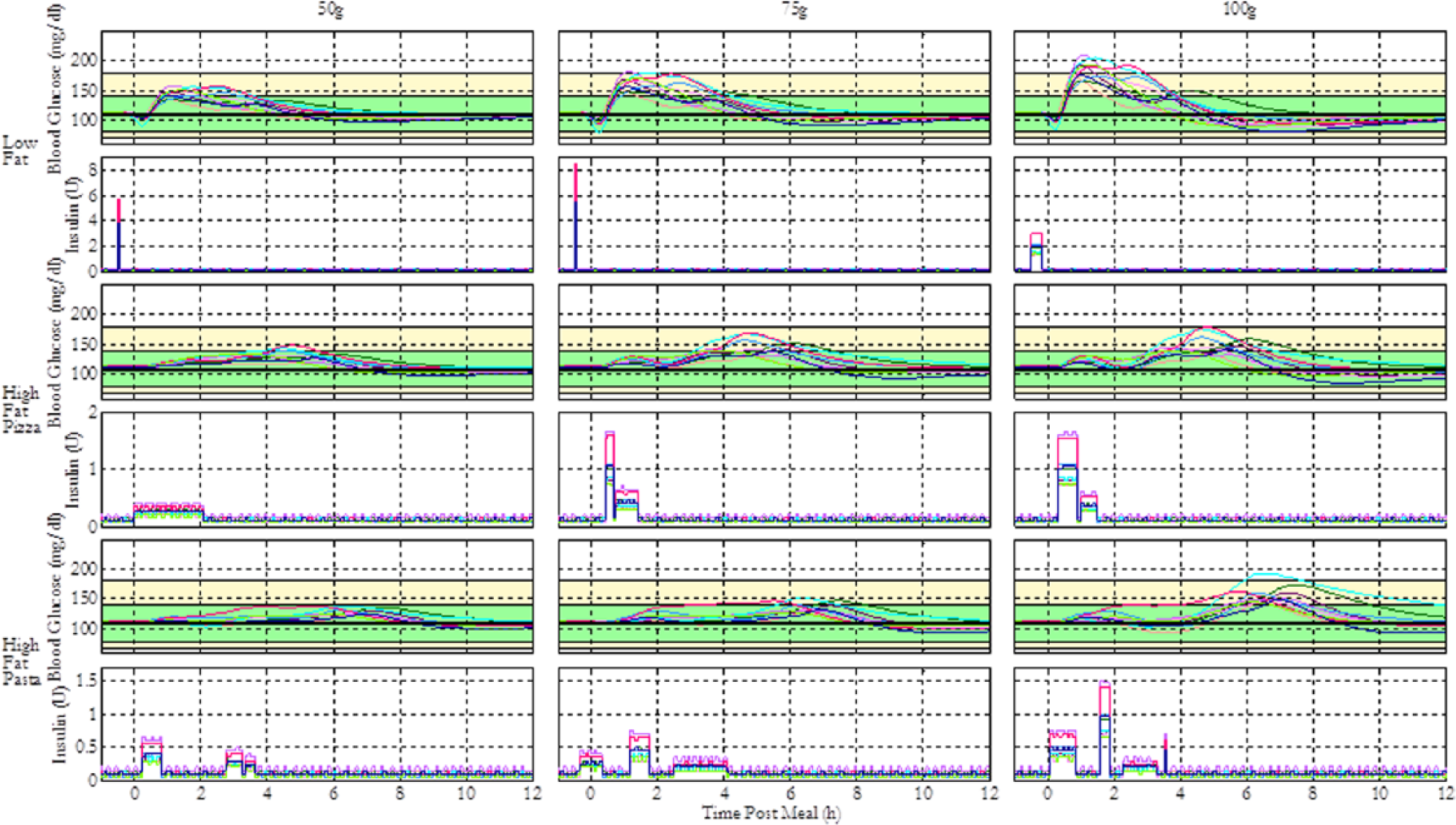
Summary of the performance of general optimal insulin regimens (GORs) for 3 different meal types of various sizes. The figure shows the blood glucose response profiles and insulin delivery patterns for 10 in silico subjects given 9 different meal scenarios. The green shaded region represents the 80-140 near-euglycemic zone, and the yellow region represents the zone between 70-180 hypo- and hyperglycemic thresholds.
Figure 4 shows that the optimal insulin delivery profile for low-fat meals with 50 and 75 g of CHO is a normal bolus administered 30 minutes prior to the meal, whereas the optimal profile for 100 g low-fat meals is a 15-minute square wave delivered starting at the same time. This is feasible considering the increased risk of hypoglycemia that follows normal bolus compensation for larger meals. The novel regimens maintain most subjects within the safe BG range of 70-180 mg/dl. The scheduling of preprandial insulin deliveries is influenced by the constraint for the earliest dose, set to be 30 minutes before the meal in consideration of the practical difficulties of meal planning. The fast digestion of low-fat meals causes rapid glucose absorption that is faster than insulin absorption and action given subcutaneous delivery and thus needs preprandial insulin to avoid severe hyperglycemia.
In contrast, the GOR for medium-fat pizza meals with 50 g CHO is a 2-hour square wave, and the optimal profiles for 75 and 100 g CHO meals are biphasic with the insulin still delivered within a 2-hour postprandial period. The regimens maintain the BG within 70-180 mg/dl for most subjects. Since medium-fat pizza meals exhibit slow glucose absorption due to the longer digestion process, 20 the optimized regimens do not include preprandial insulin (see Figure 4).
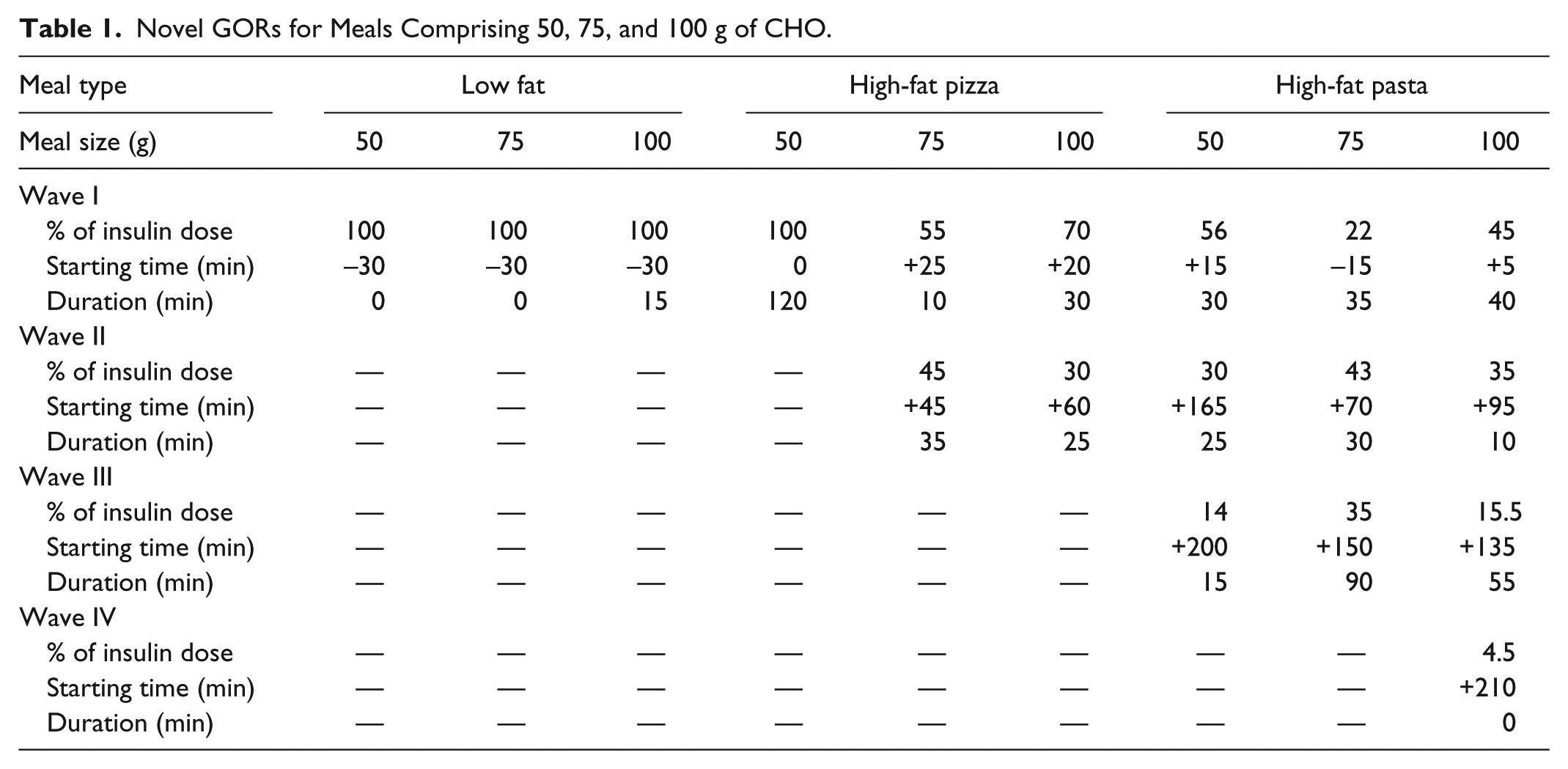
Similarly, the optimal profiles for 50, 75, and 100 g CHO high-fat pasta meals are biphasic, multiphasic, and multiphasic over 4 hours, respectively. Increasing number of waves with meal size is realistic since a greater, steady glucose excursion would require multiple frequent insulin deliveries to avoid hyperglycemia. The novel regimens maintain all but 1 subject within 70-180 mg/dl. A summary of the novel basal bolus regimens can be found in Table 1.
Novel GORs for Meals Comprising 50, 75, and 100 g of CHO.
Large meals and interpatient variations present great challenges in BG control and contribute to the difficulty of prescribing general recommendations. Hence, IORs were next determined to confirm the general regimens, and to find potential trends that may be useful in prescribing regimens for certain groups of patients. The IORs and GORs for 100 g low-fat, high-fat pizza, and high-fat pasta meals are presented in Figure 5.
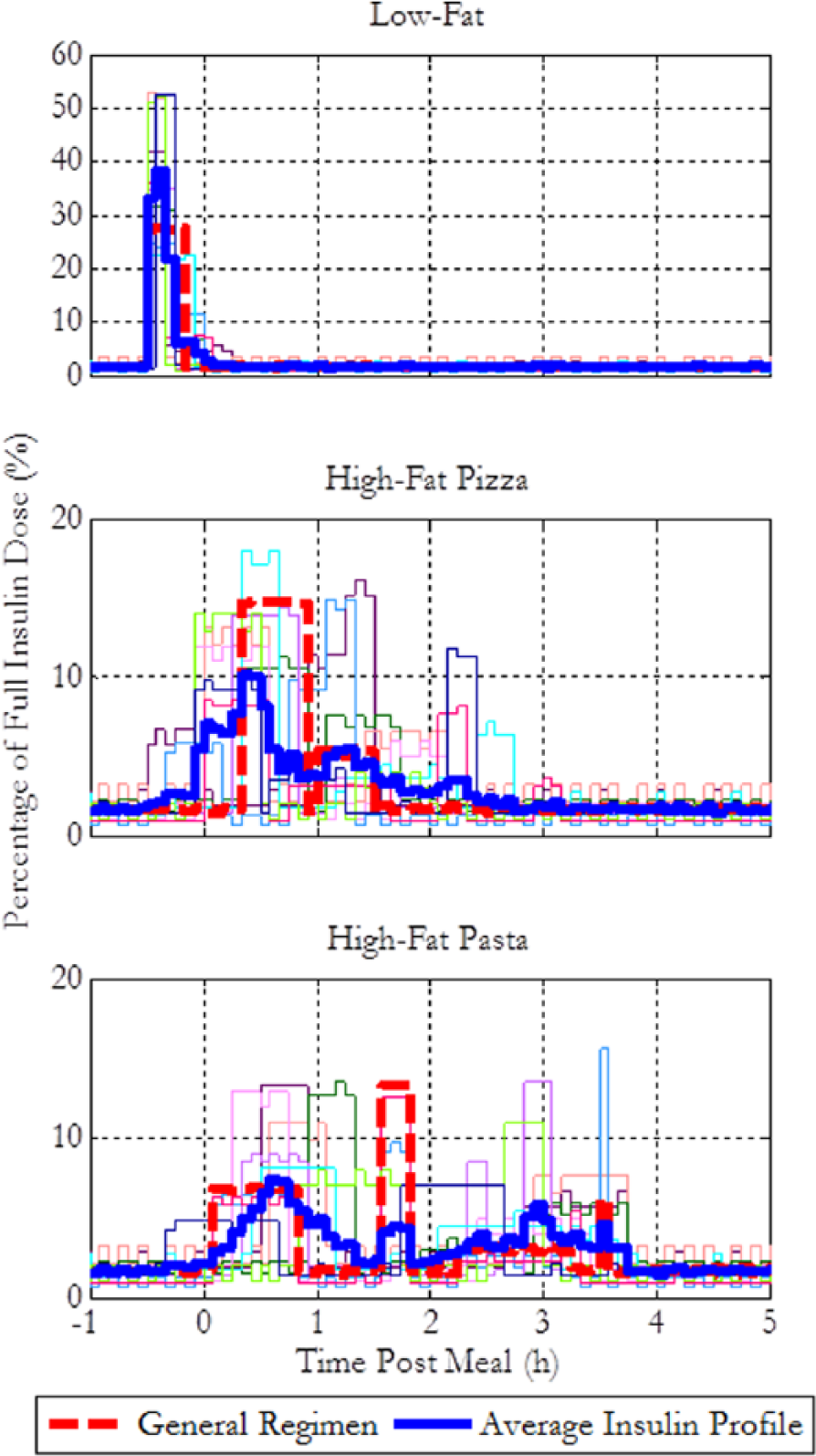
Comparison of individual optimal regimens (IORs), the average individual profile, and the general optimal insulin regimen (GOR) for 10 in silico subjects when given 100 g CHO meals of different fat content.
Figure 5 shows that highly insulin sensitive subjects (high I:C ratios) display a consistent trend of short wave patterns, whereas the less sensitive subjects benefit from extended waves. Figure 6, showing the IORs of in silico subjects for a high-fat pizza meal, grouped by their insulin sensitivities, also qualitatively demonstrates this trend, suggesting that patients with low insulin sensitivity require a longer delivery period due to their relatively slow insulin action.
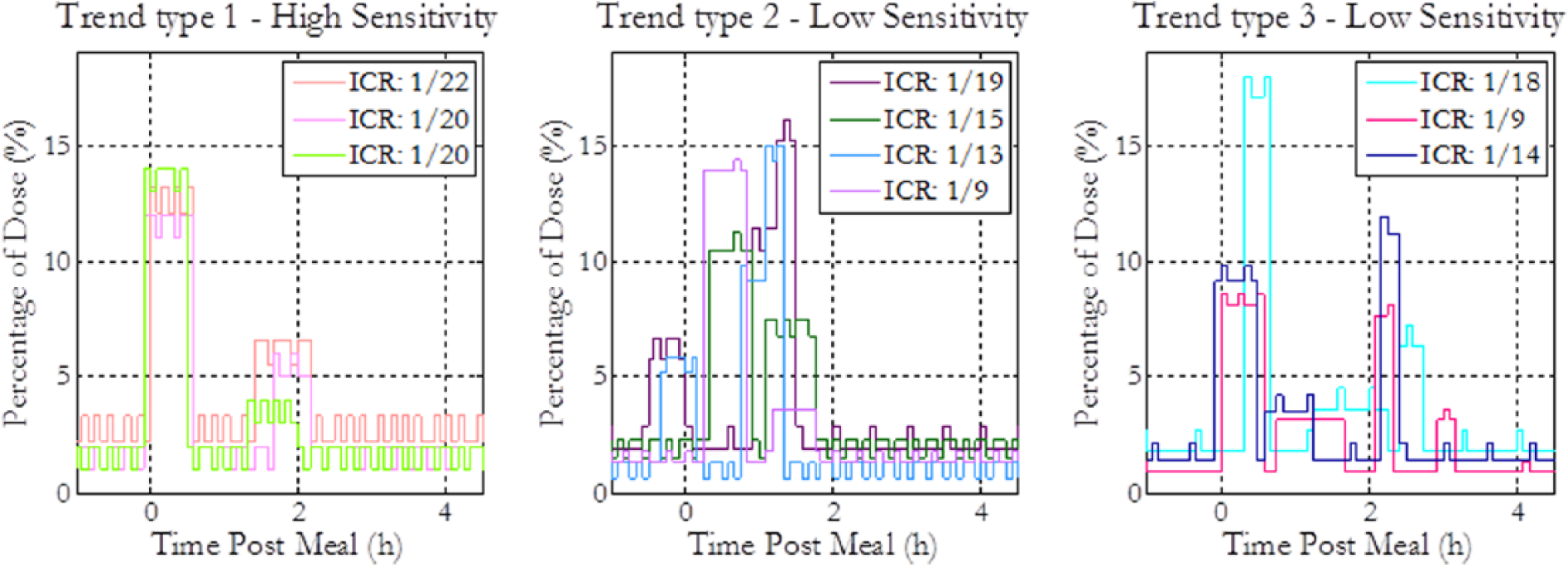
Observed trends in individually optimized regimens for 10 in silico subjects in the case of high-fat pizza meals. The left plot shows the group of in silico subjects with the highest insulin sensitivities and whose individual optimal regimens (IORs) are biphasic. The middle plot presents another group of subjects that have biphasic insulin delivery patterns, and the right plot shows the subjects whose insulin regimens are multiphasic.
Overall, IOR of most subjects for the low-fat meals is a short wave, validating the GOR. Similarities between the GOR and the average individual profile indicate providing a general recommendation to patients is a reasonable approach for this meal type. This was also confirmed by statistical paired t tests with an alpha level of .05, revealing no significant difference in performance between the optimized general and individual regimens. A comparison of the average performance of the GORs and IORs for a 100 g meal with different fat contents can be found in Table 2.
Comparison of the Mean Performances of the GOR and IOR for Meals With 100 g CHO and Varying Fat Content.
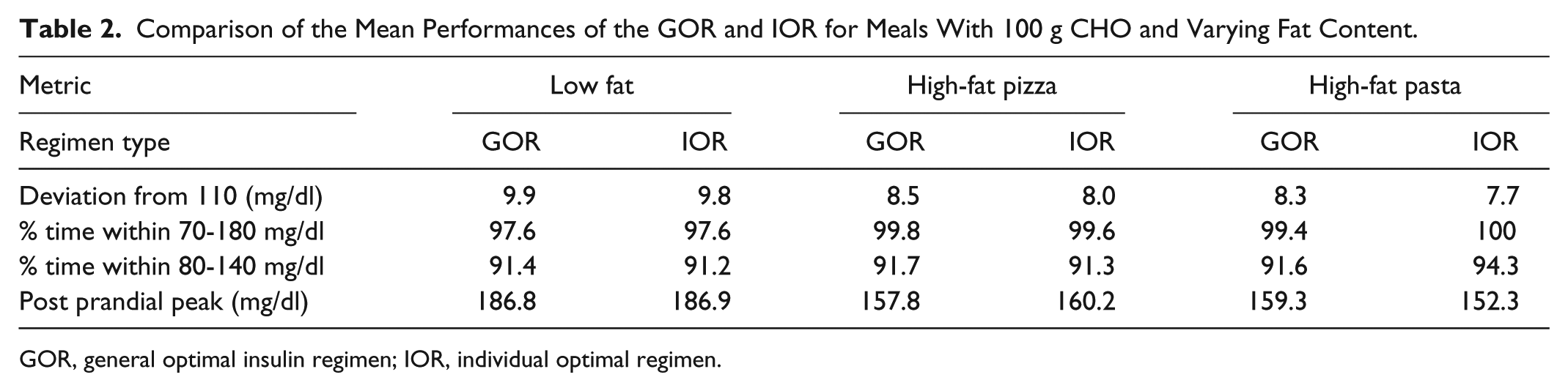
GOR, general optimal insulin regimen; IOR, individual optimal regimen.
The results for the high-fat pizza meal indicate notable variations between the individual regimens, mainly determined by insulin sensitivity. Three distinguishable trends can be observed: biphasic pattern with specific time of deliveries, biphasic pattern with varying time of deliveries, and multiphasic patterns. Extremely sensitive subjects display the most consistent trend and receive optimal control from the first trend type. The GOR is similar to the average individual profile despite significant variations between the individual regimens. Furthermore, statistical tests show insignificant difference in performance between the regimens.
Similarly, the IORs for the high-fat pasta meals display bi- and multiphasic patterns. IORs of highly sensitive subjects are biphasic, whereas those of less sensitive subjects are multiphasic. This is reasonable since multiple waves can cause increased fluctuations in BG for sensitive subjects. The GOR can maintain all but 1 subject within the safe BG range, similar to the IORs, showing the relevance and applicability of a GOR.
Optimized Closed-Loop Regimens
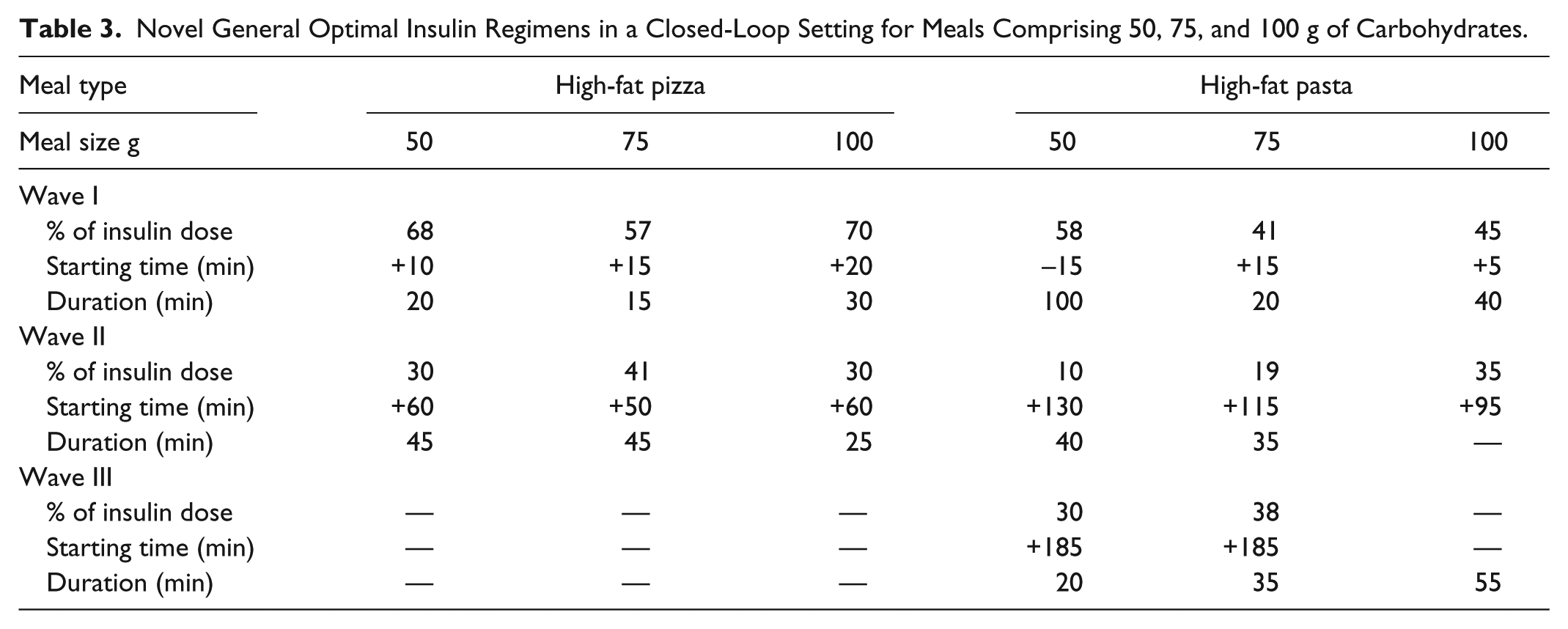
An AP with premeal boluses can benefit from combining controller action with a fixed supplementary insulin regimen specific to each meal. A controller regulates insulin infusion based on feedback of calculated error between the measured and desired BG. A PID controller calculates control action based on current error, summation of past errors, and rate of change of current error. The tuning of 3 parameters that define a simple, fixed parameter PID controller (Kc, τI, and τD) was optimized to minimize the cost function as in Equation 1 simultaneously for 10 virtual subjects within the UVA/Padova simulator when given a meal with 50 g of fast-absorbing CHO under closed-loop control. A PSO algorithm using the same settings as in section III was utilized to find the optimal values. Optimization of GORs of 9 different meal scenarios was performed with PID control, using CGM values as its input. A summary of results is shown in Figure 7.
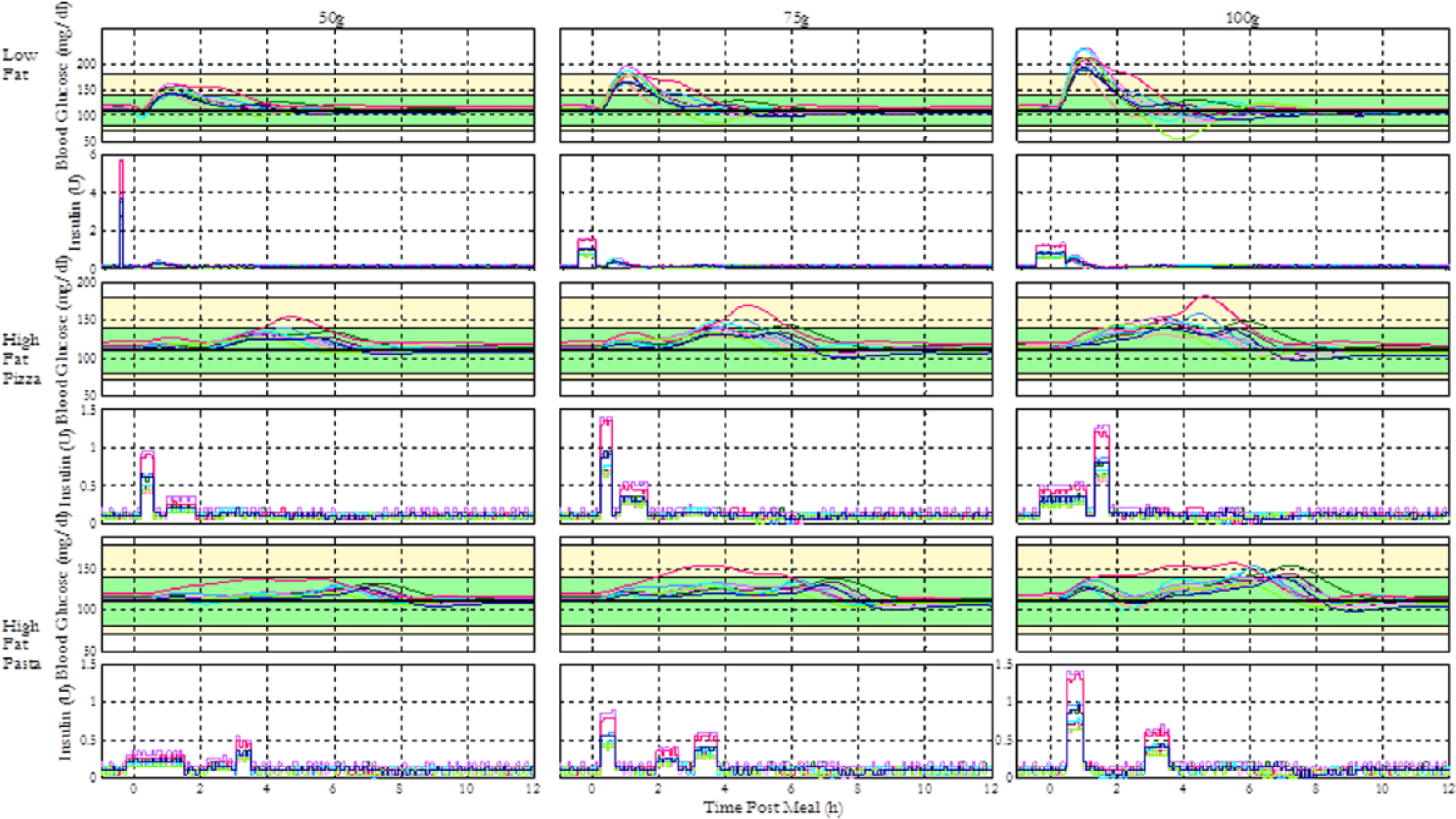
Summary of closed-loop general optimal insulin regimens (GORs) for 3 different meal types of various sizes. The figure shows the blood glucose response profiles and insulin delivery patterns for 10 in silico subjects given 9 different meal scenarios. The green shaded region represents the 80-140 near-euglycemic zone, and the yellow region represents the zone between 70-180 hypo- and hyperglycemic thresholds.
Optimized low-fat closed-loop regimens presented in Figure 7 are nearly identical to results obtained in basal bolus setting. The proposed high-fat pizza meal GORs are biphasic with a 2-hour postprandial insulin infusion compared to the basal bolus regimen, which consists of 1 long wave. This difference can be attributed to limited control action due to slow glucose excursion, explaining the initial short wave in the closed-loop regimen for the 50 g meal to address the need for fast insulin infusion. Likewise, the dissimilarity for 100 g meals can be attributed to increased initial derivative control action as a result of the fast glucose excursion that follows large meals. Therefore, the closed-loop regimen for this meal is initialized by slower insulin infusion than the smaller meals. The novel closed-loop regimens maintain all subjects within 70-180 mg/dl, even for the largest meal size. A statistical comparison to the basal bolus regimens shows significant improvement in performance of the closed-loop approach.
The PSO algorithm was also used to determine the optimal closed-loop GORs for high-fat pasta meals. The 50 and 75 g meal regimens are multiphasic, whereas the 100 g meal regimen is biphasic with insulin being infused over a 4-hour postprandial period. This can be attributed to the faster rise in BG concentration caused by large meals, resulting in higher derivative action and reducing the need for frequent insulin waves. The optimized regimens for high-fat pasta meals maintain all subjects well within the safe BG range. A summary of the regimen designs can be found in Table 3.
Novel General Optimal Insulin Regimens in a Closed-Loop Setting for Meals Comprising 50, 75, and 100 g of Carbohydrates.
These promising results indicate that controller action in combination with GORs can provide very effective control of postprandial glucose excursions.
Discussion
The insulin regimen for a standard meal in conventional basal bolus therapy is a normal bolus, 33 and the performances of our novel open- and closed-loop regimens were compared to this scheme in Figure 8, showing that the novel regimens perform significantly better in terms of the postprandial peak, time in euglycemia, and deviations from the baseline concentration for all meal scenarios. The normal bolus performs adequately for low-fat meals, but is not optimal for slower absorbing meals. This confirms published data from recent clinical trials,29,34,35 which suggest dual wave insulin delivery as optimal for high-fat meals. Traditional normal bolus and square wave boluses have also been considered for such meals. However, these regimens have not been as successful at attenuating the postprandial BG peak as the dual wave. 34 The results obtained in this study also show that square waves, biphasic, and multiphasic regimens better attenuate postprandial glucose excursions following high-fat meals. Although the dual wave bolus is currently considered optimal for high-fat meals, novel open- and closed-loop GORs generated in this study show superior postprandial control in in silico trials.
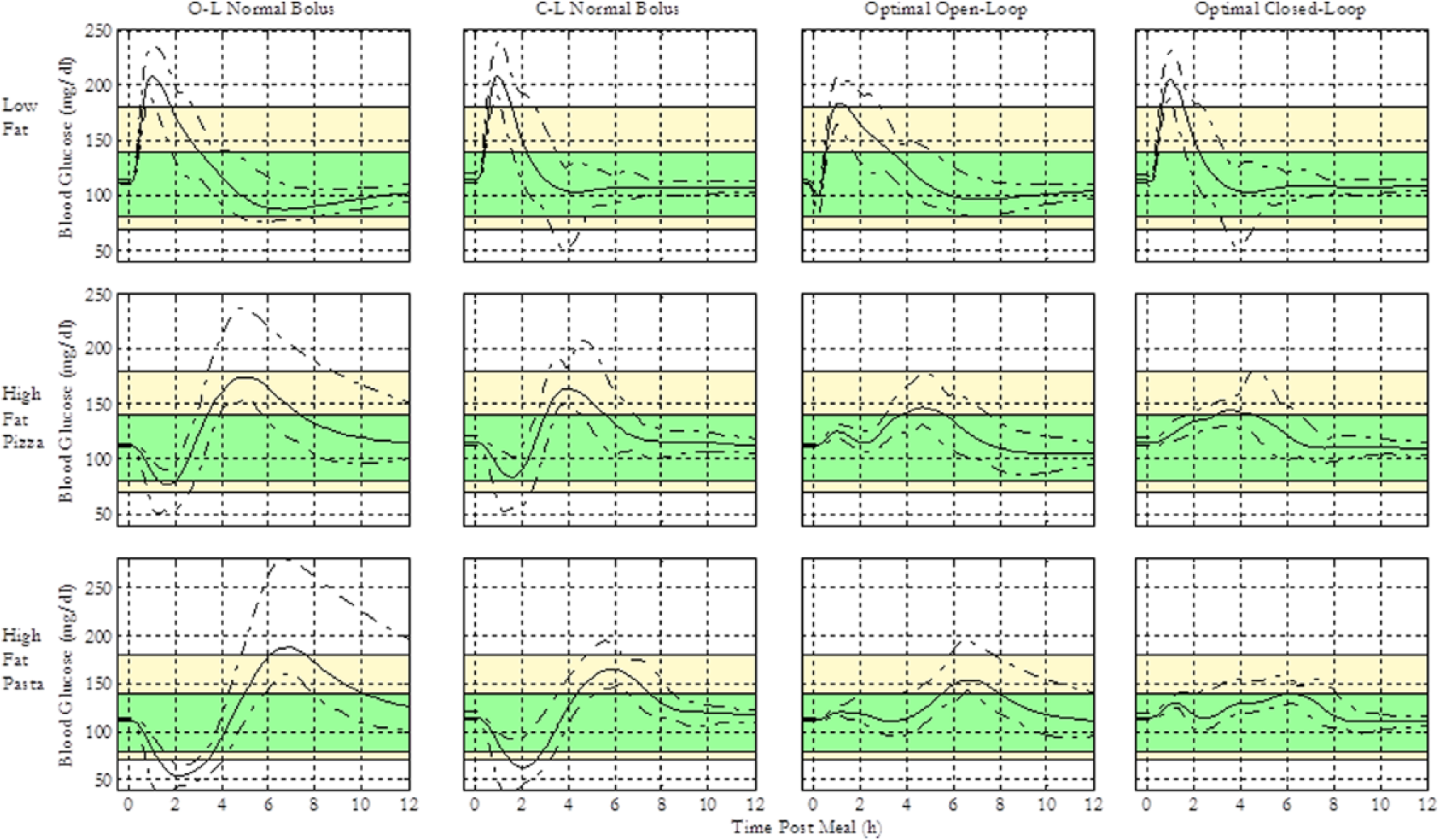
Comparison of insulin regimen performance for 3 different meal types, each comprising 100 g of CHOs. The solid curves represent the average blood glucose response profiles for 10 in silico subjects, and the area between the dashed lines represents the min-max profiles. The green shaded region represents the 80-140 near-euglycemic zone, and the yellow region represents the zone between 70-180 hypo- and hyperglycemic thresholds.
In addition, further investigations of GORs were conducted to test for wider applicability to patients beyond the training population. The full UVA/Padova simulator allows simulation of 100 in silico subjects that thoroughly cover interindividual variability in insulin-glucose dynamics. Figure 9 shows performances of the 6 optimized open- and closed-loop GORs for meals with varied fat content for this larger population, which validate the improvements in blood glucose control using the proposed scheme in the 100 subject testing population.
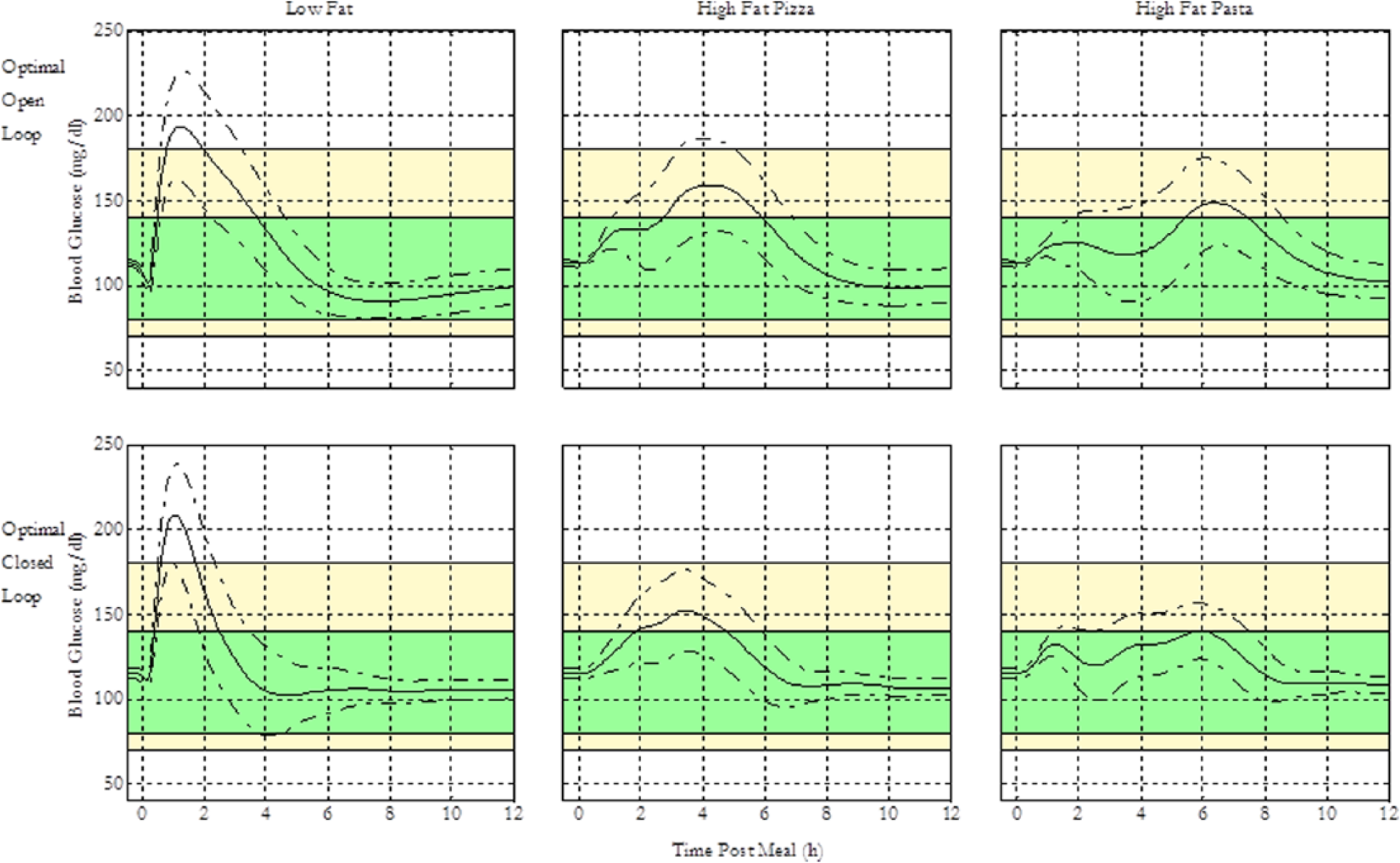
Summary of open- and closed-loop general optimal insulin regimen (GOR) performance under the validation scenario for 3 meal types with different fat content and 100 g CHO, under the 100 in silico subjects for the UVA/Padova simulator. The figure shows the blood glucose response profiles for 100 in silico subjects in the full version of the UVA/Padova simulator given 3 different meal scenarios. The green shaded region represents the 80-140 near-euglycemic zone, and the yellow region represents the zone between 70-180 hypo- and hyperglycemic thresholds.
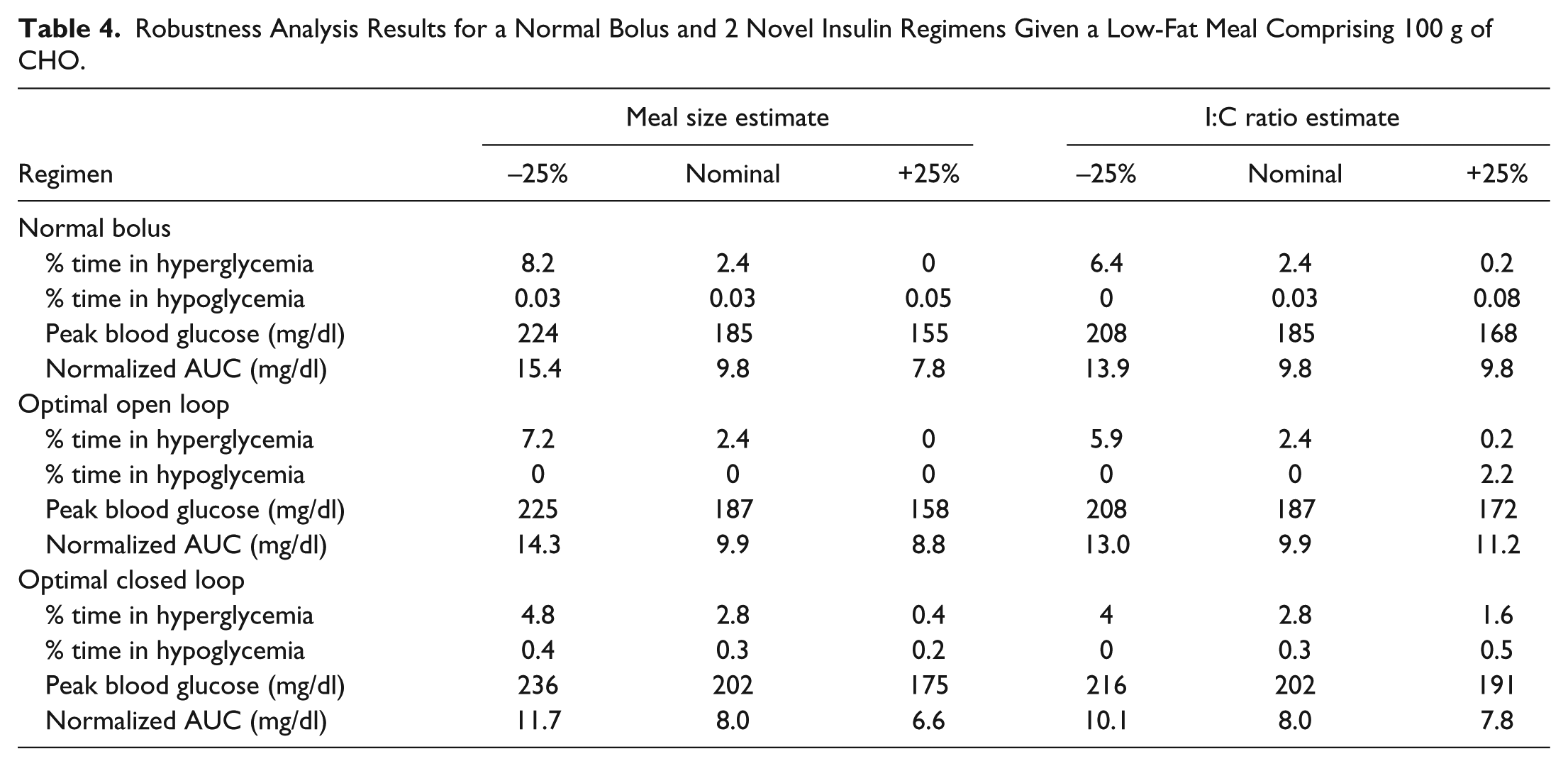
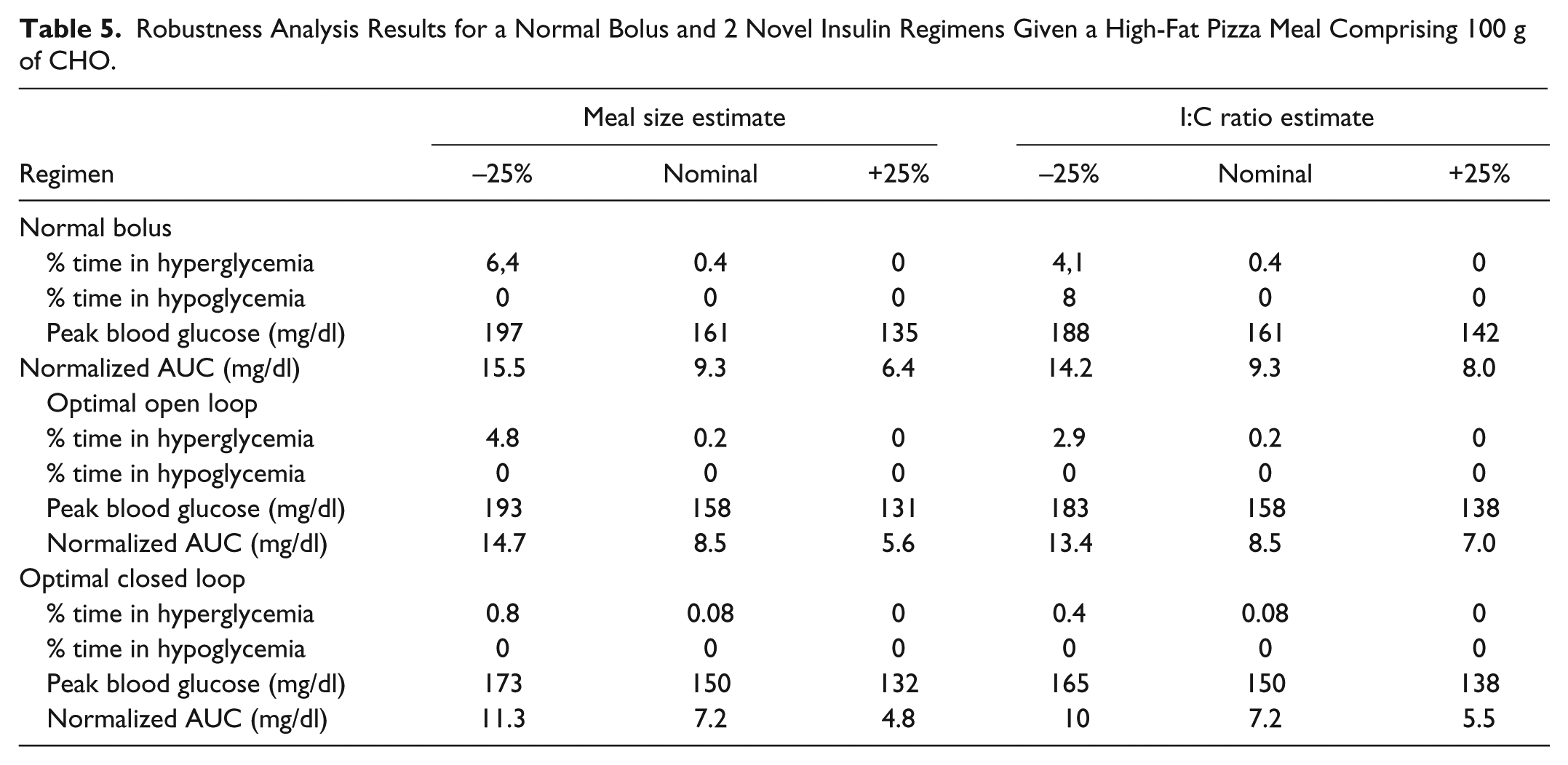
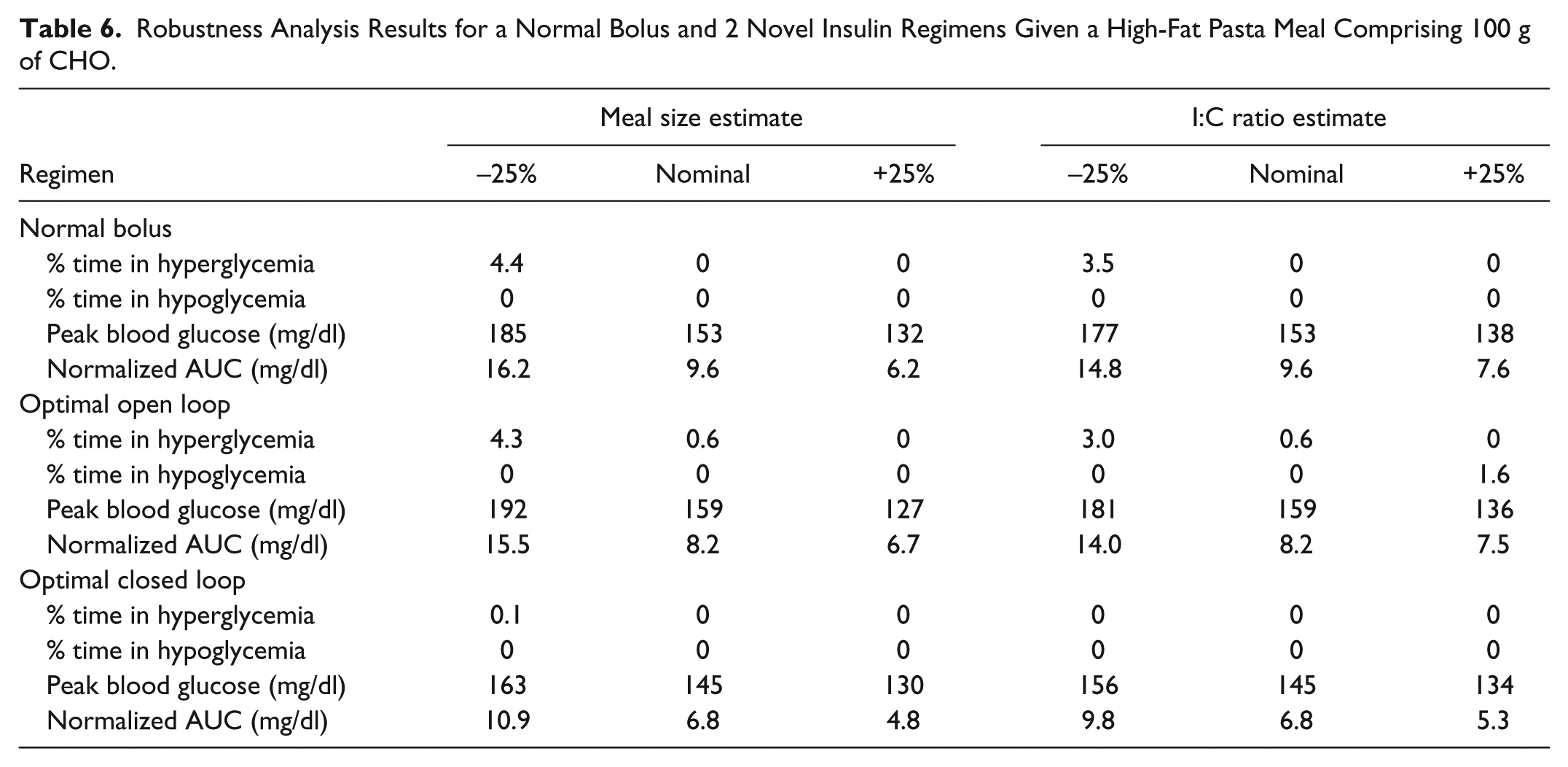
Finally, the results of this study are more robust to uncertainties in meal size and insulin-to-CHO ratio estimates. Tables 4 to 6 show the effect of 25% over- and underestimations of 2 parameters that effect insulin delivery to compensate meals—meal size estimate and I:C ratio estimate—on each bolus delivery regimens and for low, medium, and high-fat 100 g CHO meals. Overall results from this analysis show more robustness of the novel closed-loop schemes versus existing schemes. The data also indicate that closed-loop regimens combining a PID controller with a supplementary GOR have smaller variation in performance over the different scenarios.
Robustness Analysis Results for a Normal Bolus and 2 Novel Insulin Regimens Given a Low-Fat Meal Comprising 100 g of CHO.
Robustness Analysis Results for a Normal Bolus and 2 Novel Insulin Regimens Given a High-Fat Pizza Meal Comprising 100 g of CHO.
Robustness Analysis Results for a Normal Bolus and 2 Novel Insulin Regimens Given a High-Fat Pasta Meal Comprising 100 g of CHO.
The proposed scheme in this study does not rely on optimal basal therapy and is thus a realistic strategy with significant potential real-life applications in AP. A clinical trial of the GOR is necessary to confirm results before practical applications of the proposed regimens.
Conclusion
Qualitative replications of published data of high-fat meal absorption were conducted in silico, and a library of novel meal compensation strategies for a variety of meal compositions was constructed using PSO methodology. Optimization yielded novel open- and closed-loop insulin regimens that provide better control of postprandial glucose excursions than existing schemes. Novel delivery patterns propose unique waveforms not available in existing insulin pump wizards. GORs for low-fat meal are a normal bolus or a short square wave delivered at least 30 minutes prior to the meal. GORs for high-fat meals are typically biphasic, but can extend to multiple phases depending on meal composition. Preliminary investigations of the GORs under varied fat content also display bi- or multiphasic patterns.
Closed-loop schemes with optimized supplementary delivery profiles are significantly more robust to uncertainties compared to traditional schemes despite wide variations in individual subject insulin sensitivities. Since the proposed schemes do not rely on individual optimizations, it is a more realistic strategy with significant potential for practical applications in an AP.
Footnotes
Acknowledgements
The full population UVA/Padova simulator was received from Dr Claudio Cobelli and Dr Boris Kovatchev for research purposes.
Abbreviations
AP, artificial pancreas; BG, blood glucose; CGM, continuous glucose monitor; CHO, carbohydrate; CSII, continuous subcutaneous insulin injection; GA, genetic algorithm; GOR, general optimal insulin regimen; IOR, individual optimal regimen; MPC, model predictive control; NIH, National Institutes of Health; PID, proportional-integral-derivative; PSO, particle swarm optimization; T1DM, type 1 diabetes mellitus; UVA/Padova, University of Virginia/Padova.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health grant DP3DK094331.