Abstract
Background:
Computerized insulin infusion protocols have facilitated more effective blood glucose (BG) control in intensive care units (ICUs). This is particularly important in light of the risks associated with hypoglycemia. End stage renal disease (ESRD) increases the risk of insulin-induced hypoglycemia.
Methods:
We evaluated BG control in 210 patients in 2 medical ICUs and in 2 surgical ICUs who were treated with a computerized insulin infusion program (CIIP). Our CIIP was programmed for a BG target of 140-180 mg/dL for medical ICU patients or 120-160 mg/dL for surgical ICU patients. In addition, we focused on BG control in the 11% of our patients with ESRD.
Results:
Mean BG was 147 ± 20 mg/dL for surgical ICU patients and 171 ± 26 mg/dL for medical ICU patients. Of both surgical and medical ICU patients, 17% had 1 or more BG 60-79 mg/dL, while 3% of surgical ICU and 8% of medical ICU patients had 1 or more BG < 60 mg/dL. Mean BG in ESRD patients was 147 ± 16 mg/dL similar to 152 ± 23 mg/dL in patients without ESRD. Of ESRD patients, 41% had 1 or more BG < 79 mg/dL as compared with 17.8% of non-ESRD patients (P < .01).
Conclusions:
A higher BG target for medical ICU patients as compared with surgical ICU patients yielded comparably low rates of moderate or severe hypoglycemia. However, hypoglycemia among ESRD patients was more common compared to non-ESRD patients, suggesting a need for a higher BG target specific to ESRD patients.
Keywords
The level of glycemic control needed to improve outcomes without increasing the risk of hypoglycemia in hospitalized patients with critical illness is controversial. Achieving reasonable control of blood glucose without causing an excess risk of hypoglycemia is especially challenging in patients with ESRD, a condition commonly encountered in the critically ill.
In 2001, Van den Berghe and colleague’s landmark study in a surgical intensive care unit (ICU) suggested that strict blood glucose (BG) control (mean 103 mg/dL) was associated with a mortality and morbidity benefit in comparison to standard therapy (mean 153 mg/dL). 1 A subsequent study of patients in the medical ICU however failed to show similar benefits and hypoglycemia was a frequent complication. 2 Meta-analysis of multiple studies shows an inconsistent mortality benefit with intensive glycemic control and highlights the increased risk of hypoglycemia. 3 More recently, the NICE-SUGAR study compared all-cause and cause-specific mortality and morbidity (new organ failure, bacteremia, red-cell transfusion, and volume resuscitation) in over 6100 surgical and medical ICU patients treated either with an intensive insulin regimen (target BG 81-108 mg/dL) or a standard insulin regimen (target BG less than 180 mg/dL). 4 This trial concluded that intensive BG control was associated with a significant increase in the incidence of both moderate hypoglycemia (BG 41-70 mg/dL), and severe hypoglycemia (BG ≤ 40 mg/dL). Both degrees of hypoglycemia were associated with increased all-cause mortality, and particularly cardiovascular mortality. 5
The cumulative effect of repeated hypoglycemia-related stress responses increases the risk of cardiac arrhythmias, neurological impairment, seizures, and death.5-6 In light of the negative outcomes associated with hypoglycemia higher BG targets have been suggested for patients in the ICU.
The availability and implementation of computerized infusion insulin programs (CIIPs) are increasingly being shown to be able to improve the stability of BG control in the ICU. In 1 CIIP implemented across multiple ICUs, there was a 20% absolute increase in number of BG in target range and a slightly lower frequency of BG < 50 mg/dL 3 months after transition from a paper-based protocol to a computerized protocol. 7 In a report using the Yale Insulin Infusion protocol, the incidence of BG < 60 mg/dL decreased from 23.1% of patients in the paper-based version to 9.5% of patients in the computerized version. 8 In the LOGIC–Insulin algorithm study, patients in the CIIP group experienced significantly less mild (BG < 70 mg/dL) moderate (BG < 60 mg/dL) and severe hypoglycemia (BG < 40 mg/dL) in comparison to patients treated with a paper-based insulin infusion protocol, while both groups achieved similar mean BG levels. 9 Comparable BG control with a reduction in the incidence of hypoglycemia was reported in other studies comparing computerized to paper-based insulin infusion protocols.10-12
In January 2013 we converted all 4 adult ICUs in our hospital (2 medical and 2 surgical) to a CIIP. In this report we describe the results achieved for mean BG and the incidence of moderate and severe hypoglycemia. In addition, we focused on BG control in the subpopulation of patients with chronic renal failure since our previous work has shown their increased risk of hypoglycemia during treatment with insulin. 13
Methods
Rush University (Chicago, IL) is an academic medical center housing 4 ICUs: medical (MICU), cardiac (CCU), surgical (SICU), and neurocritical care unit (NCCU). The SICU includes general surgical (SICU-Gen), cardiovascular (SICU-CV), and transplant (SICU-Tx) patients. ICU patients with 2 BG values above 160 mg/dL are started on the CIIP. The GlucoStabilizer™ program (Alere Informatics Solutions, Charlottesville, VA) is a CIIP that allows for titration of continuous insulin infusion to maintain BG within a target range specific to each ICU. The program runs on the hospital network and can be accessed from any bedside computer. Each run of CIIP data includes all BGs and insulin infusion rates. If the CIIP is restarted after being stopped (eg, for a procedure), the data collected after restarting are filed as a new run. For the purposes of data analysis we bundled together BG values from all runs for each individual patient. We began using the GlucoStabilizer program to manage all IV infusions in our ICUs in mid-December 2012. We collected data from all consecutive patients from January 1 to August 30, 2013, who were treated with IV insulin using the CIIP. Patients with diabetic ketoacidosis or nonketotic hyperosmolar syndrome were excluded from the study because they were treated with a paper protocol specific to them using a fixed rate of IV insulin.
All BG values were obtained via a bedside point-of-care device (Precision Xceed Pro™ Abbott Diabetes Care, Alameda, CA). The bedside nurse enters BG results into the CIIP and the program recalculates the next insulin infusion rate by using the formula (BG in mg/dL – 60) times a multiplier. The initial default multiplier is 0.02. The multiplier may change every hour, increasing if the current BG is above the target range or decreasing if the current BG is below the target range. The multiplier changed in increments of 0.01. The interval between BG checks may vary from 30 minutes to 120 minutes, depending on the rate of change of BG, but most of the time was every 60 minutes. We defined an undesirably low BG as being < 80 mg/dL. In the event of any BG < 80 mg/dL, the program calculates a corrective dose of 50% dextrose and schedules another BG check in 15 minutes. We set a target BG range of 120-160 mg/dL in the SICU and NCCU and a target BG range of 140-180 mg/dL in the MICU and CCU because our experience and published experience has noted a greater risk of hypoglycemia in the medical ICUs.1,2
IV insulin was used only for patients who were NPO or were receiving continuous enteral tube feeds, or continuous parenteral nutrition.
As soon as a patient was able to begin a PO diet, they were transitioned to a subcutaneous (SQ) basal-bolus insulin regimen. We used the mean IV insulin infusion rate for 4-6 hours prior to conversion. If a patient was converted to SQ glargine every 24 hours, the initial dose was 20 times the mean hourly IV insulin infusion rate. Glargine was used for patients who already took glargine prior to admission and for most postoperative patients. If the patient was converted to SQ neutral protamine hagedorn or isophane insulin (NPH) every 12 hours, the initial dose was 10 times the mean hourly IV insulin infusion rate. NPH was used for patients who already took NPH prior to admission, for patients receiving continuous enteral tube feeds and for patients after solid organ transplants who were typically receiving high doses of glucocorticoids. CIIP was continued for 3-4 hours, overlapping the first SQ dose of basal insulin.
Statistical Analysis
Data from all CIIP runs were initially entered into Excel. Statistical analysis was performed using SPSS 11.0 (SPSS Inc, Chicago, IL) and Microsoft Excel. Normally distributed continuous variables were reported as mean and SD and compared using t tests. Statistical significance was defined with P value < .05, and all results were 2-tailed.
The study was performed with the approval of the Rush University human subjects investigational review board.
Results
Between December 2012 and August 2013, we prospectively collected BG data from all 210 patients, admitted to the 4 ICUs who were treated with IV insulin using the CIIP. A total of 267 CIIP runs were recorded. Baseline patient characteristics are presented in Tables 1 and 2. Patients in the 2 medical units were more likely than patients in the 2 surgical units to have a past history of diabetes, a higher admission HBA1C, and a lower mean estimated glomerular filtration rate (eGFR). Patients with ESRD were more likely to have a past history of diabetes, but mean HBA1C was similar to patients without ESRD. Patients with ESRD had a significantly longer ICU and hospital length of stay (data not shown).
Patient Characteristics.

Patient Characteristics Medical Versus Surgical Units.
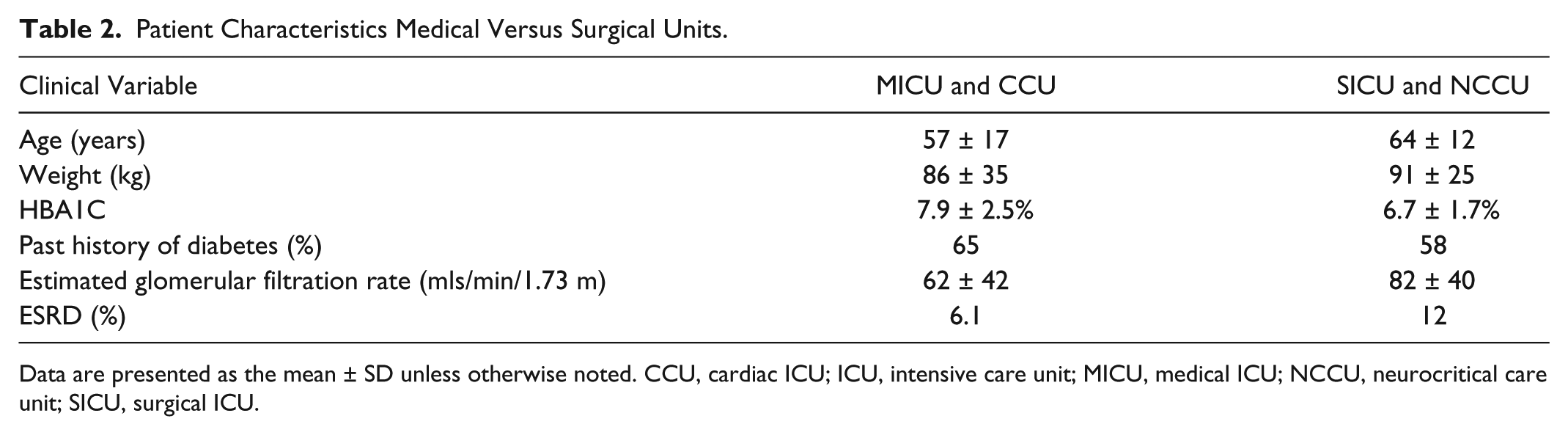
Data are presented as the mean ± SD unless otherwise noted. CCU, cardiac ICU; ICU, intensive care unit; MICU, medical ICU; NCCU, neurocritical care unit; SICU, surgical ICU.
BG Control
Patients in the surgical ICUs reached goal BG range faster than those in medical ICUs (2.64 ± 3.4 hours vs 4.39 ± 3.9 hours, P < .001), as shown in Table 3. Surgical ICU patients spent more time on CIIP (63 hours) than medical ICU patients (53 hours), and SICU-CV patients spent fewer hours on CIIP in comparison to other SICU patients.
Time to Target Blood Glucose and Total Time on Insulin Infusion.
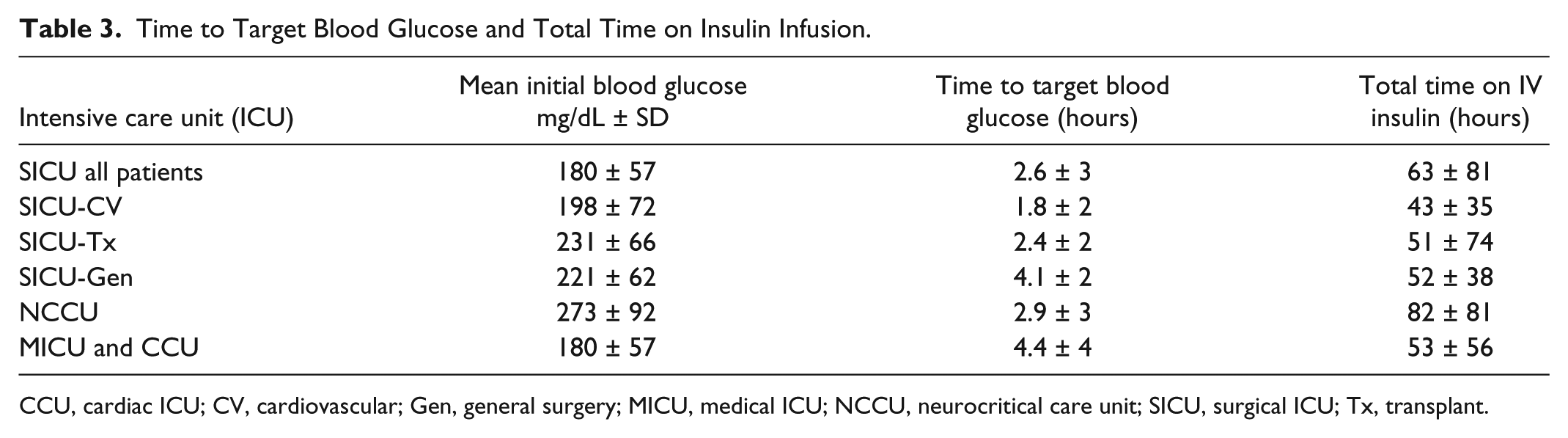
CCU, cardiac ICU; CV, cardiovascular; Gen, general surgery; MICU, medical ICU; NCCU, neurocritical care unit; SICU, surgical ICU; Tx, transplant.
As expected given their different target ranges, mean BG for surgical ICU patients was significantly lower than that of medical ICU patients (147 ± 20 vs 171 ± 26 mg/dL), as shown in Table 4. There was no significant difference in mean BG between MICU and CCU patients. Among SICU patients, SICU-CV patients had a mean BG (141 ± 7 mg/dL) that was significantly lower than those in SICU-Gen and NCCU (150 ± 15 and 150 ± 26 mg/dL respectively; P = .005 and .001). Mean BG in patients without a prior diagnosis of diabetes was significantly lower than in patients with type 2 diabetes (146 ± 14 vs 155 ± 26 mg/dL, respectively; P = .0002). Patients with ESRD had a similar mean BG as compared to non-ESRD patients (147 ± 15 vs 152 ± 23; P = .18).
Mean Blood Glucose by Hospital Unit, Renal Function, and Type of Diabetes Mellitus.
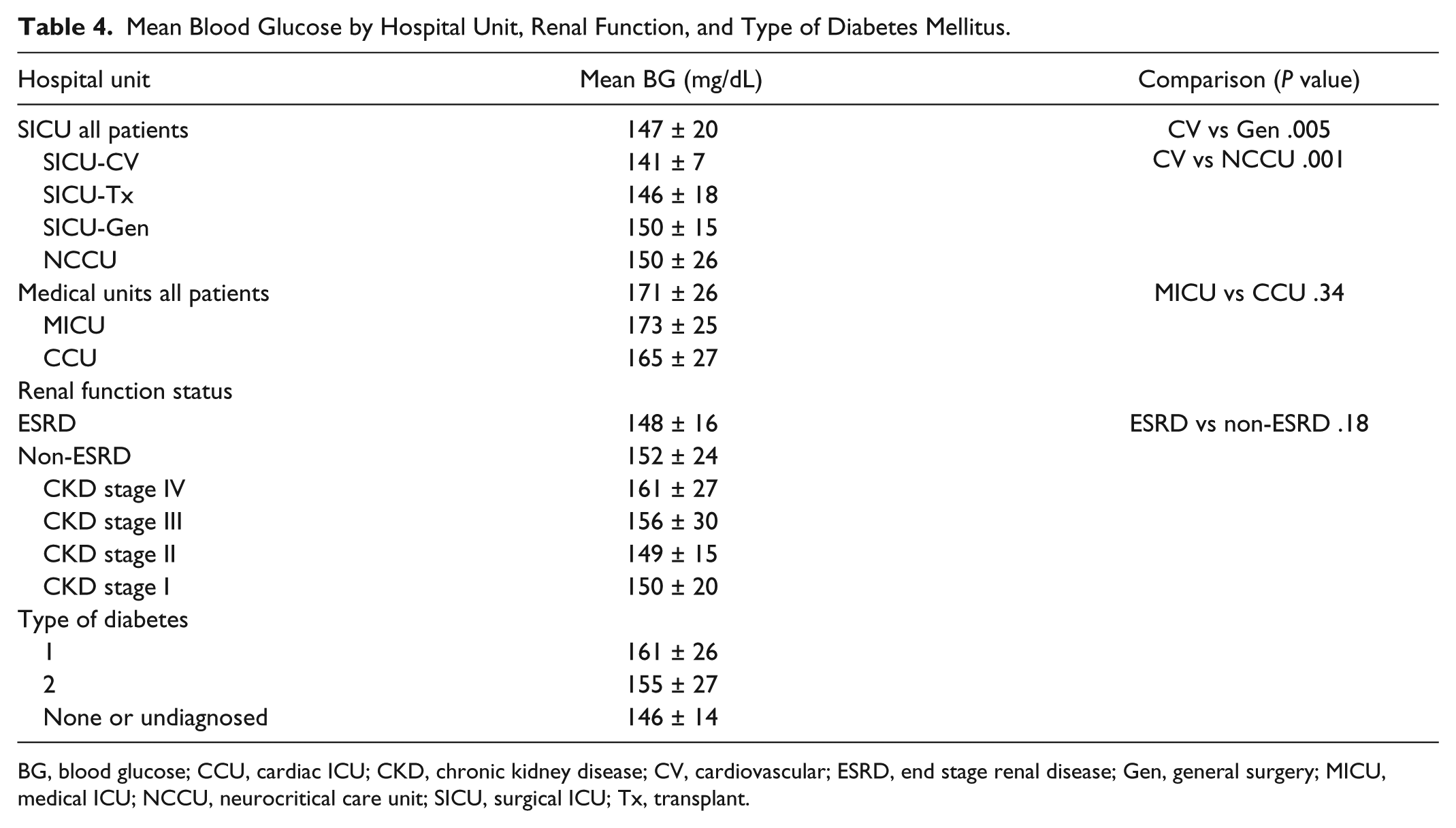
BG, blood glucose; CCU, cardiac ICU; CKD, chronic kidney disease; CV, cardiovascular; ESRD, end stage renal disease; Gen, general surgery; MICU, medical ICU; NCCU, neurocritical care unit; SICU, surgical ICU; Tx, transplant.
Incidence of Hypoglycemia
Hypoglycemia was subdivided into 4 categories: BG 60-79 mg/dL, BG < 70 mg/dL, BG 40-59 mg/dL, and BG < 40 mg/dl. The incidence of BG 60-79 mg/dL did not differ between surgical ICUs and medical ICUs; both were 17% of patients. Of all patients, 9.8% had a BG < 70 mg/dL, higher in the medical as compared with the surgical ICU patients. The incidence of BG 60-79 mg/dL was significantly higher among ESRD patients as compared to non-ESRD patients (34% vs 14.5%). Similarly 17% of ESRD patients had a BG < 70 mg/dL as compared with 3.3% of non-ESRD patients. The incidence of a BG < 40 mg/dL was very low, only 1 patient in the entire data set, and only 3.3% of all patients has a BG 40-59 mg/dl (Table 5).
The Incidence of Hypoglycemia by Type of ICU and Renal Function.
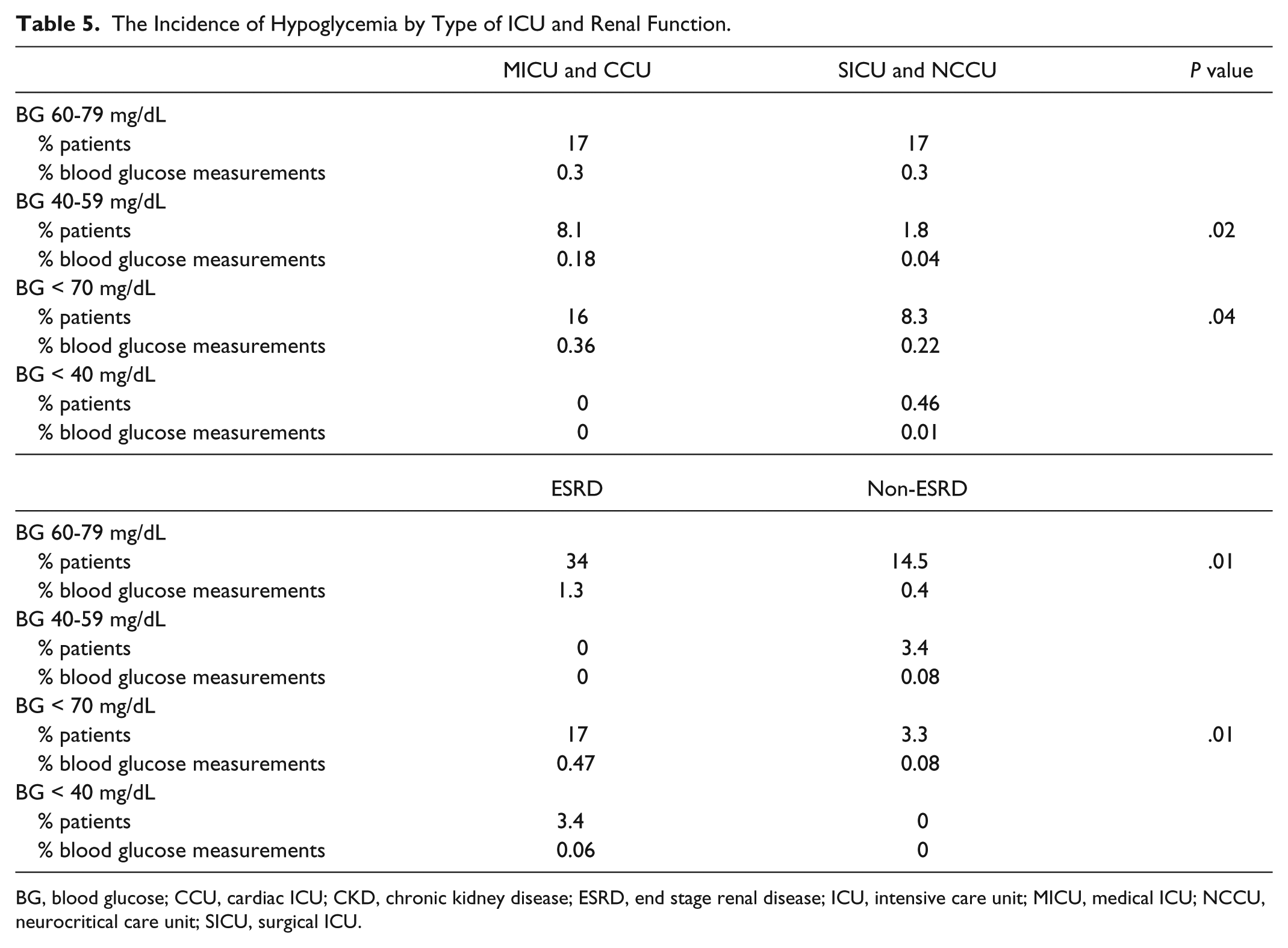
BG, blood glucose; CCU, cardiac ICU; CKD, chronic kidney disease; ESRD, end stage renal disease; ICU, intensive care unit; MICU, medical ICU; NCCU, neurocritical care unit; SICU, surgical ICU.
Transition From CIIP to SQ Basal Insulin
Of patients, 19% received no SQ basal insulin when CIIP was discontinued, usually because their insulin requirement was less than 1 unit/hour. Of patients, 44% received NPH insulin (mean dose 25 ± 14 units), while 37% received glargine insulin (mean dose 28 ± 17 units).
Discussion
We describe the implementation of a computerized protocol for controlling IV insulin infusion in a group of 4 adult medical and surgical ICUs in a single institution. The impetus to switch from a common paper protocol to a computerized protocol was 2-fold. First, the paper protocol required hourly calculations at the bedside. Late or missed BG determinations and protocol misunderstandings contributed to excessive glucose variability and an increased frequency of hypoglycemia. Second, maintaining consistently excellent and safe BG control in multiple ICUs staffed by hundreds of nurses and resident physicians is a challenging task given nursing staff turnover and frequent rotation of resident physicians. The continued success of the paper protocol depended on constant monitoring and supervision of the effort by the inpatient diabetes management team consisting of endocrinology fellows and faculty. By switching to a computerized protocol we are able to automate and standardize all aspects of IV insulin titration and greatly simplify training needed for the bedside caregivers, as well as much of the need for endocrinology oversight.
The use of IV insulin in the ICU setting has matured over the past decade, and computerization of the process was a natural step in quality improvement. As we identify optimal BG targets for different patient types, it is straightforward to create specific parameters within the program for IV insulin infusion that promote greater success and uniformity at meeting these BG targets with the lowest risk of hypoglycemia. By setting a slightly higher BG target in our medical ICUs (140-180 mg/dL) as compared with our surgical ICUs (120-160 mg/dL), we are able to achieve a comparably low incidence of BGs less than 80 mg/dL (0.35-0.5% of total BG measurements). This success is very similar to that achieved in other studies and underscores the predictability and uniformity which can result from computerization of the process.7-12 The recent analysis of the NICE-SUGAR study found that overall 48.7% of patients had a BG value less than 71 mg/dL as compared with our results where 9.8 % of patients had a BG value < 70 mg/dL. 5 In the recent publication of results from computerization of the Yale IV insulin protocol, 17% of their patients had a BG less than 70 mg/dL, somewhat more than our results. 8 The LOGIC-1 trial of a computerized IV insulin protocol found that 32.2% of patients had a BG value less than 70 mg/dL, however their BG target was 80-110 mg/dL, lower than our targets or the Yale protocol target of 100-140 mg/dl. These comparisons illustrate how the closely the frequency of hypoglycemia will associate with which particular BG target range is chosen. 9
The NICE-SUGAR study reported that BG levels between 41-70 mg/dL were associated with an increased mortality, 5 while Egi et al reported that even higher BG levels, 72-81 mg/dL, were associated with an increased mortality as compared with patients whose BG levels were always greater than 81 mg/dL. 14 These results suggest that the ideal BG target for these patients is one that keeps the frequency of a BG < 70-80 mg/dL to a minimum.
The metabolism of insulin is decreased in patients with ESRD, and the uremic milieu impairs gluconeogenesis, an important defense from hypoglycemia. 15 As patients with type 2 diabetes evolve from chronic renal failure to ESRD, insulin requirements decrease and may disappear completely, a phenomenon known as “burnt out” diabetes. 16 While having diabetes or chronic renal failure independently raises the chance of hypoglycemia, this risk is highest when both conditions are present simultaneously. In a retrospective study of over 200 000 veterans, those with chronic kidney disease and type 2 diabetes had an adjusted rate of hypoglycemia that was 2 times higher than those with diabetes alone (10.72 vs 5.33 per 100 patient-months, P < .001). Furthermore, each hypoglycemic event was associated with increased 1-day mortality, and the degree of risk was directly related to the severity of hypoglycemia. 17
Treatment of diabetic patients with chronic renal failure and ESRD inevitably involves balancing glycemic control with prevention of hypoglycemia. In a previous study we randomized hospitalized patients with type 2 diabetes and chronic renal failure (mean eGFR 30 mL/min) to glargine/glulisine insulin (total daily dose 0.5 units/kg) versus a reduced dose regimen (0.25 units/kg). The mean BG seen during the hospital stay was similar between the 2 groups, however the incidence of hypoglycemia was reduced by 50% in the reduced dose group. 14 Studies of outpatients with diabetes and ESRD have shown, using HBA1C, that extremes of glycemia (HgA1C <6% or above 8%) are associated with increased all-cause mortality. 18 Thus, a target HgA1C in the 7-8% range has been recommended for patients with diabetes and ESRD. 19 Since this HBA1C range translates to an estimated mean glucose of 150-180 mg/dl, we suggest that this BG range is also reasonable for these patients when they are hospitalized. To our knowledge no prior studies of IV insulin infusion in the ICU setting, either controlled by a paper protocol or a computer program, have focused on the subset of patients with ESRD.
The results from the current study support the concept that patients with ESRD being treated with an IV insulin infusion in critical care units will benefit from a specifically modified protocol to minimize the consequences of excess hypoglycemia. A study of setting the BG target to 160-200 mg/dL in such patients is currently underway.
The study has several limitations. First, all data were collected from a single medical center, although 4 different ICUs caring for diverse types of patients are included. Second, data were not available to compare the BG control in our ICU patients using our paper IV insulin protocol versus using our CIIP.
Conclusions
We continue to make strides to improve glycemic control in patients needing intensive care while minimizing their risk of hypoglycemia. The implementation of a CIIP for all adults in our medical center has improved our ability to provide safe and stable BG control. We have identified target ranges for BG control specific to different patient types that have enabled us to achieve a comparably low incidence of hypoglycemia in all patients. We have identified that critically ill patients with ESRD are at a greater risk to develop hypoglycemia. Our findings suggest that a higher BG target, such as 160-200 mg/dL, may reduce the risk of hypoglycemia in critically ill patients with ESRD.
Footnotes
Acknowledgements
The authors acknowledge Melissa Browning, Carol Squires, Connie Weissman, and Marsha Mulbarger.
Abbreviations
BG, blood glucose; CCU, cardiac intensive care unit; CIIP, computerized insulin infusion program; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; ESRD, end stage renal disease; HBA1C, hemoglobin A1C; ICU, intensive care unit; MICU, medical intensive care unit; NCCU, neurocritical care unit; NPH, neutral protamine hagedorn or isophane insulin; SICU, surgical intensive care unit; SQ, subcutaneous; Tx, transplant.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Baldwin is the principal investigator of a research grant to Rush University from NovoNordisk.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The implementation of the GlucoStabilizer™ program at Rush University was supported by a generous gift from Richard Seidel.
