Abstract
Background:
Automated blood glucose (BG) and insulin pump systems allow wireless transmission of all BG readings to a user’s pump. This study aimed to assess whether use of such a system, as compared with a manual BG entry insulin pump, resulted in higher mean daily frequency of BGs recorded after 6 months.
Methods:
A 12-month randomized crossover trial, comprising 2 phases, was conducted. All participants used insulin pump devices with automated vs manual BG entry for 6 months each; order of system use was randomly assigned. Device interactions were assessed from pump and glucometer downloads.
Results:
Thirty-five participants were enrolled; 9 withdrew during the study. Use of the automated insulin pump system resulted in higher mean daily BG recorded over 6 months of use when compared to a manual BG entry system (5.8 ± 1.7 vs 5.0 ± 1.9;
Conclusions:
Use of an automated glucometer/insulin pump resulted in higher number of BGs recorded over 6 months when compared to an insulin pump with manual BG entry. This may be especially beneficial for adolescent manual system users who enter <5 BGs per day into their pump.
Continuous subcutaneous insulin infusion (CSII) pumps have resulted in improved metabolic control and quality of life for motivated users, with reduced risk of severe hypoglycemic episodes.1,2 However, current devices cannot yet operate independently and require patient interaction regarding blood glucose level (BGL), amount and timing of carbohydrate consumption and expected levels of activity.
Maintenance of glycated hemoglobin (HbA1c) within defined targets is critical to reduce the risk of developing macro- and microvascular diabetes-associated complications, 3 however a significant proportion of youth do not attain this goal, particularly during adolescence.4-7 While CSII can provide increased meal and exercise flexibility relative to injectable insulin regimens, achievement of adequate glycemic control remains highly dependent on user behavior.8-10 Achievement of target HbA1c has repeatedly been shown to positively correlate with the number of BGLs performed per day, both in youth on injectable insulin regimens and in those utilizing CSII.8,11-17 Studies of this correlation have mainly relied on BGL data obtained from self-report or logbook, with few studies using objective BGL data extracted directly from glucometers.15-17 For BGLs to positively impact on glycemia with use of CSII, they need to be performed and also entered into the user’s pump, to enable correction insulin delivery when required. Until recently, this has required the user to manually enter BGLs into their pump device; adherence to this process is variable in children and adolescents. 18 Adjunct glucometers have recently been developed, which allow individuals’ capillary BGL to be automatically delivered to their pump using wireless technology, thus removing the necessity of this manual step.
The objective of this study was to assess whether use of an automated integrated blood glucose measurement and insulin pump device, as compared with standard insulin pump device with manual BGL input, resulted in higher mean daily frequency of blood glucose measurements recorded in a user’s pump after 6 months of use. We also aimed to assess whether use of the automated system impacted on frequency of other pump interactions, user satisfaction and glycemic control (as assessed by HbA1c) after 6 months of use.
Methods
Subjects
Participants were recruited for this randomized crossover trial from the outpatient department of a large single tertiary center. The trial was registered precommencement at the Australian New Zealand Clinical Trial Registry (ACTRN12611000142932). Ethics approval was obtained from the institutional Human and Research Ethics Committee and the study procedure conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants, with additional consent from a parent/guardian where a participant was aged <18 years old.
Inclusion criteria for participation were age <20 years old, established use of an insulin pump with manual BGL entry for >1 year, and a user’s current pump remaining within warranty for the trial duration. No HbA1c or baseline BGL monitoring criteria were applied. Individuals with a history of previous significant skin reaction to pump consumables or lack of English language comprehension were excluded.
Recruitment, Randomization, and Design
Postal invitations to participate were sent to all eligible CSII users within our patient population. Further interested participants were then recruited from the outpatient clinic directly. The automated pump system studied was the Accu-Chek Spirit Combo (Roche™, Basel, Switzerland), which was the only system available at the time of recruitment which incorporated automated wireless BGL delivery. Remote control functionality from a handheld unit that includes the glucometer component of the system is also included in this device. This allows delivery of meal boluses and other pump setting adjustment, without the necessity for pump exposure. Bolus and correction insulin calculation (“bolus wizard”) is also integrated in this handset device and not accessible via the pump directly, although manual boluses can be delivered via the pump. As it was felt that removal of the handset and education regarding self-calculation of insulin dose would insert a large confounder to final analysis, participants used their own insulin pump during the manual phase of this study. All users were instructed to utilize the handset only for insulin dose calculation during the automated phase. This wireless functionality has since been incorporated into other insulin pump systems, delivered by Medtronic (Bayer® meter), Animas (One Touch® Ping® meter-remote), and has been intrinsic to the Omnipod system, although the latter is not available in Australia.
Participants were randomized either to stay on their own manual BGL entry pump system or to commence use of the automated version, using a randomized block randomization schedule to maintain balance between treatment arms and achieve allocation concealment. Sealed blank envelopes were selected sequentially by the principal investigator as participants were recruited. Participants used the device to which they were randomized for the first 6 months, then crossed over to use the alternative system for the next 6-month period (second phase).
At commencement of their 6-month automated system phase, all participants had a 3-hour standardized instructive session covering its use. Further support was provided as necessary via participant-initiated contact with hospital diabetes nurse specialists. All CSII users at our center are encouraged to regularly “download” their pump information to diabetes nurse specialists in between outpatient appointments, to assist with regular insulin titration according to BGL levels. Outpatient appointments are scheduled regularly at 3-month intervals with the participant’s treating physician, from where data for this trial were collected prospectively as set out below. When participants failed to attend a scheduled appointment with their physician, they were reviewed separately within 2 weeks by the principal investigator and their data were recorded.
Outcomes
Primary outcome examined was between group difference in the mean number of blood glucose measurements per day recorded by each pump, over the course of each 6-month period of use. Secondary outcomes were glycemic control, auxology, participant satisfaction, and other download parameters as detailed below.
For our primary outcome, the total number of BGLs as a mean from weeks 7 to 26 of each phase was documented and the means of the pooled automated system were compared with the means of the pooled standard manual system. The first 6 weeks of data from each phase were excluded from analysis to reduce the chances of a novelty effect or, in the second phase, carryover effect. BGLs recorded were obtained from pump download at baseline and 3, 6, 9, and 12 months. During the phase of pump and manual glucometer use, data from all glucometers used by participants were also downloaded, or manually extracted by investigators where electronic download was not possible.
HbA1c was recorded at baseline and 3, 6, 9, and 12 months using high-performance liquid chromatography using the Bio-Rad D-10TM Hemoglobin Testing System (Bio-Rad Laboratories Inc, Hercules, CA, USA). Normal reference range for persons without diabetes using this method is quoted at 4.5 to 5.7% (26-39 mmol/l).
As an objective measure of device satisfaction, participants independently completed the fully validated Insulin Delivery System Rating Questionnaire (IDSRQ) 19 at the end of each 6-month study phase. The IDSRQ is a Likert-type graded questionnaire composed of 6 subscales assessing patient perceptions of treatment satisfaction, impact on daily activities, treatment system efficacy, diabetes-related worries and treatment burden, psychological well-being, and overall treatment system preference. Responses were reverse coded where necessary, and means for each subscale were calculated, as per the original article. 19 Other pump download information was also assessed at 3-month intervals, namely: total daily dose per kilogram bodyweight, number of boluses per day, number of carbohydrate entries, and percentage of insulin delivered as basal versus bolus. Other than the scheduled investigations as outlined above, no additional contact above that of standard diabetes outpatient care was initiated by investigators.
Sample Size Estimation and Statistical Analysis
Audit of a sample of our patient population 2 years previously revealed a mean of 3.2 ± 1.7 BGLs per day being performed by those using a CSII regimen.
8
A smaller sample of our CSII population 1 year previously revealed a mean of 4.4 ± 2.2 BGLs/day. Studies have consistently documented that clinically significant improvement in diabetes control is achieved when users perform ≥5 blood glucose measurements per day;11,13,20,21 we calculated a sample size of 24 would give 80% power to detect a meaningful difference (ie, detection of increase of 1.0-1.8 BGLs/day) in our population, using a crossover trial design. A
Data were recorded and described in accordance with published CONSORT guidelines for randomized trials. Data were entered into Microsoft Excel and Epidata, then exported to Stata© (version 11, StataCorp, College Station, TX, USA) for analysis. Paired
Results
Between June 2011 and February 2012, a total of 35 participants were recruited, 29 as a result of the original mail out and 6 directly from the outpatient clinic. Following randomization, a total of 9 participants withdrew from the trial (see Figure 1 for flowchart).
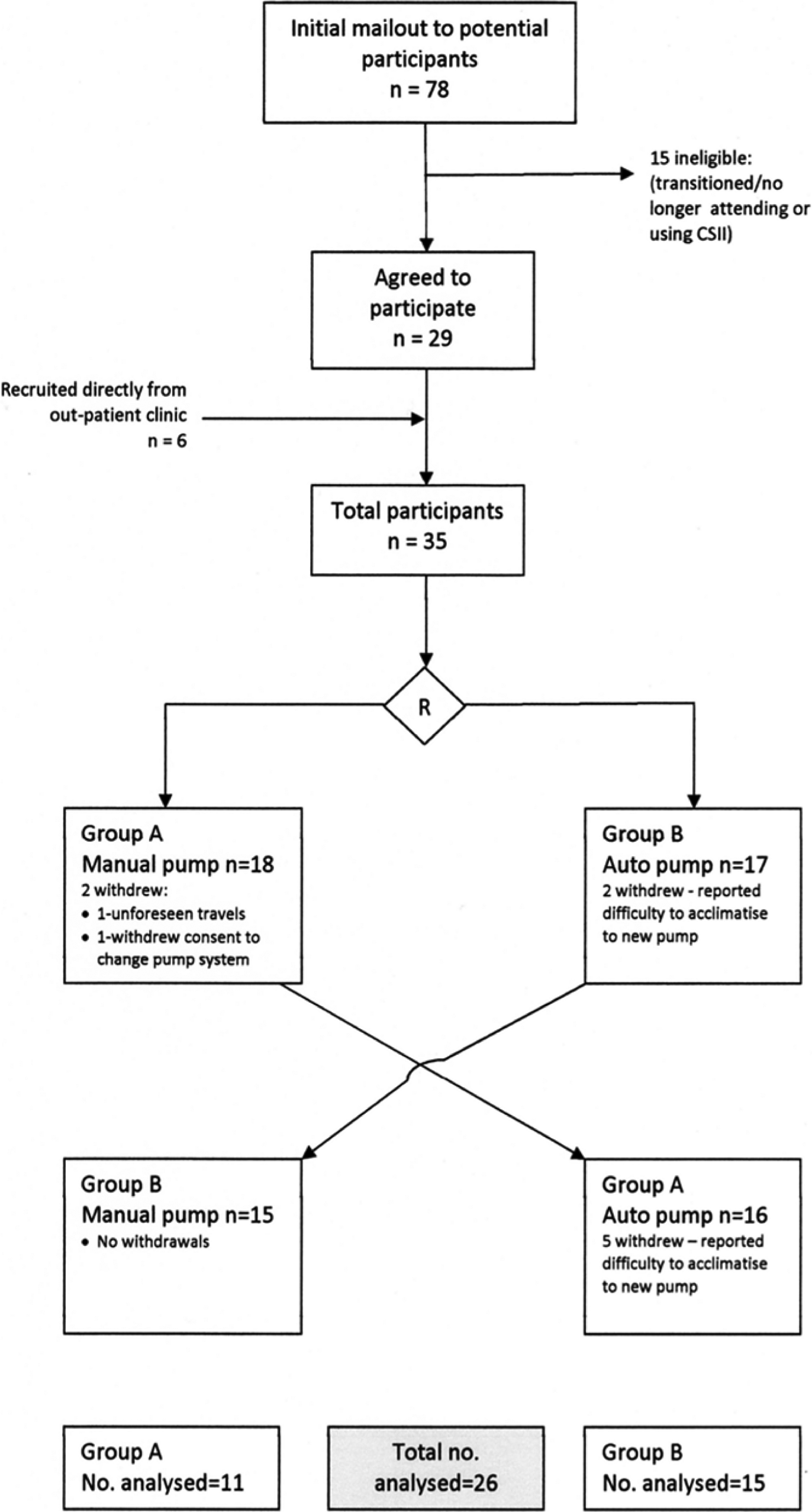
Participant flow diagram.
Baseline data at trial commencement are shown in Table 1.
Baseline Characteristics of Participants by Randomization Group.
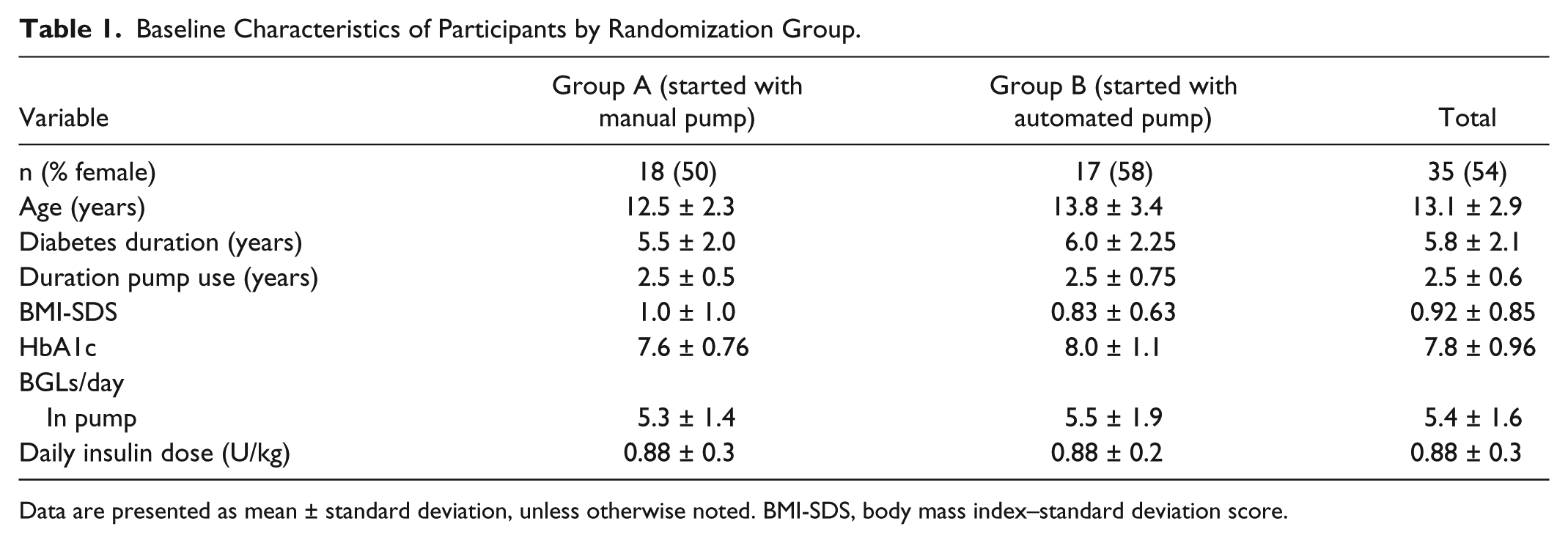
Data are presented as mean ± standard deviation, unless otherwise noted. BMI-SDS, body mass index–standard deviation score.
Recorded Blood Glucose Measurements in Pump
Use of the automated insulin pump and meter resulted in a higher number of blood glucose levels per day entered into the pump after 6 months of use when compared to use of the manual glucometer/pump device (5.8 ± 1.7 BGLs per day vs 5.0 ± 1.9 BGLs per day;
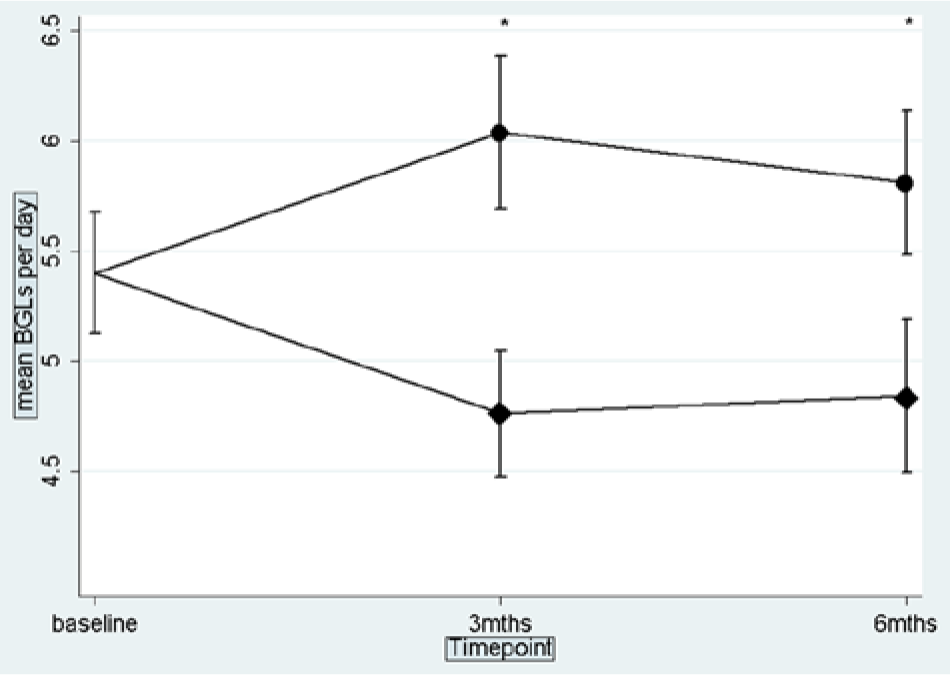
Graph of mean and confidence intervals of number of BGLs per day at each trial time point. Automated pump group represented by circles, manual glucometer/pump by diamonds.
Download of all glucometers used by participants during the manual phase demonstrated a significant difference between the number of BGLs performed per day and the number that were successfully manually entered into the user’s pump over 6 months (5.7 ± 2.4 per day in glucometer vs 4.8 ± 1.8 per day entered into pump;
Glycemic Control
No difference in overall glycemic control was demonstrated between groups at any point in the study period. Mean HbA1c after 6 months of automated pump use was 8.0% ± 1.3 (64 mmol/mol) vs 7.7% ± 0.9 (61 mmol/mol) after 6 months of manual glucometer/pump use (
On subgroup analysis, participants at trial commencement who were entering a suboptimal number (<5) of BGLs per day into their pump (n = 13) experienced significantly improved glycemic control after 6 months of automated pump use (mean ΔHbA1c: –0.9% [<5 BGLs/day] vs +0.5% [≥5 BGLs/day at baseline];
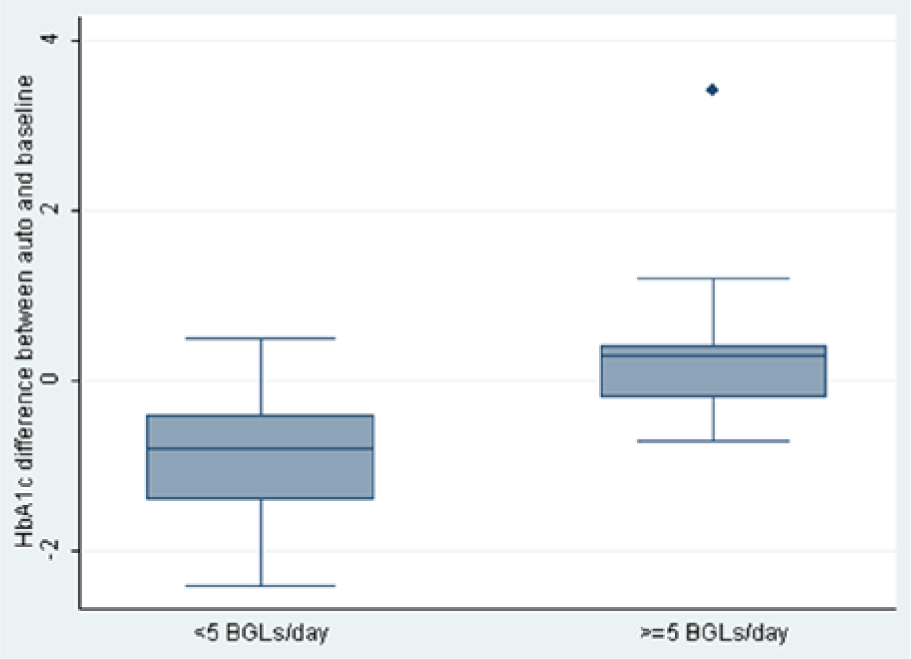
Box plot of change in HbA1c by baseline BGL/day.
Similar analysis of the same subgroups of users during the phase of manual glucometer/pump use showed no significant difference in ΔHbA1c (–0.4% [<5 BGLs] vs +0.12 [≥5 BGLs];
Insulin Dose, Bolus Behavior and BMI
The total daily insulin dose (TDD; units/kg/day) was similar for the 2 groups at the start of the study. Those using the automated pump had an increased TDD after 6 months compared to 6 months using the manual pump (0.93 ± 0.2 automated pump vs 0.87 ± 0.1 manual glucometer/pump;
There was also no difference in the number of boluses per day performed by users in either study group (6.5 ± 0.5 automated pump vs 6.9 ± 0.5 manual pump;
User Satisfaction
Contacts with diabetes nurse specialists were similar between the 2 groups during each phase of pump use (4 contacts [interquartile range (IQR) 3-5.5] during automated phase vs 3.5 contacts [IQR 1 – 6] during manual phase;
Withdrawals and Adverse Events
In total, 9 out of the 35 participants withdrew from the trial (6 female, 3 male), 7 during the phase of automated pump use and 2 from the manual phase, just prior to crossover. Participants who withdrew from the trial protocol had a shorter duration of diabetes (54 months duration vs 75 months;
Ability to perform intention to treat analysis was limited by the absence of final BGL outcome data in the 9 participants who withdrew during the trial. Methods of data modeling were felt to be inappropriate given that participants reverted to using the comparator pump system and that only 3 of the 9 withdrawals persisted with the automated pump for >3 months. However, intention to treat analysis was performed using participants’ final HbA1c data and this showed no difference between groups (7.8% ± 1.3 [62 mmol/mol] automated pump vs 7.7% ± 1.0 [61 mmol/mol] manual pump;
No severe hypoglycemic events or episodes of diabetic ketoacidosis were recorded. No difference in minor adverse events was noted between users in either study group.
At trial conclusion, participants were offered the option of continuing to use the automated pump on an ongoing basis. Nine (25.7% of overall) participants (6 female, 3 male) chose this option. This group were more likely to be adolescent, with longer duration of diabetes (87 months vs 63 months;
Discussion
The technologies incorporated into insulin pump devices are continually evolving; however as new features emerge, it is important to determine whether they are beneficial to users. This randomized crossover study demonstrates that wireless BGL entry can result in increased numbers of BGLs recorded in users’ pumps; however no extra boluses and no overall change in HbA1c occurred as a result. Akin to other work,15,16 this study is one of the few to utilize objective BGL data extracted directly from users’ glucometer(s) and insulin pump systems, but extends this further by (1) randomization of participants to manual versus wireless, (2) inclusion of user satisfaction survey, and (3) data collected prospectively at multiple time points.
Analysis of this entire cohort’s data during the phase of manual glucometer/pump use revealed that on average, users were neglecting to enter 1 blood glucose value per day into their pump. It is possible that this value was within euglycemic range and therefore recognized by participants to be unlikely to change any insulin dose delivered, as has recently been elegantly demonstrated. 18 However we could not retrospectively test this hypothesis as a significant proportion of glucometers had manual extraction of the frequency of BGLs/day performed, without precise values being recorded. Previous research has established glycemic benefits with entry of ≥5 BGLs per day to an individual’s pump.12,20,22 The lack of appreciable impact of the increased BGL data on overall HbA1c in this cohort may also relate to the fact that participants were already entering a mean of 5.4 BGLs per day at baseline. It is possible that recruitment was biased by a more motivated cohort volunteering to partake in this study. which was more likely to display higher levels of diabetes self-care.
Given this unexpected baseline finding, we felt it appropriate to perform subgroup analyses of our cohort, by those who were and were not recording an optimal number (≥5) of BGLs per day at baseline. Of note, these analyses were post hoc and unplanned at the study’s outset. The subgroup of older adolescents who were not routinely performing this recommended number of BGLs per day at baseline demonstrated a marked improvement in glycemic control during the phase of automated pump use. Akin to other studies documenting a Hawthorne effect on HbA1c, this group exhibited a 0.4% improvement while using their own pump,23,24 but experienced a further mean improvement of 0.5% in addition to this while using the automated system. Whereas the overall group recorded an increased number of BGLs during the automated pump phase, our data indicate the automated system may be especially advantageous for individuals who are having difficulty manually transferring more than 5 BGLs per day into their pump.
Nine participants did not complete the study protocol, the majority dropping out during the phase of automated pump use, or just prior. This may represent a limitation of the crossover study design in this instance, as participants had a comparator with which they were already very familiar. However, the required number of participants still completed the study so we believe that the dropout rate has not skewed the final population analyzed and that this had no effect on the overall power of the trial.
Although the majority of participants were less likely to change to the automated system at the end of the study, 9 individuals chose to remain using the automated pump after trial completion. That this group was older may represent an increased comfort level with technology operation amongst adolescents, in comparison to older adults, who are more likely to assume responsibility for daily pump operation on behalf of younger children. This study was also limited by the inability to reliably document the regularity and appropriateness of insulin basal rate or bolus ratio titration by users in response to identified glycemic patterns. Passivity regarding setting adjustment to achieve target BGLs has recently been recognized as a significant barrier to control 10 and may also explain why glycemic control did not improve overall, despite increased BGL data with automated system use. It is also interesting that users increased their TDD during the automated pump phase, but neither an increase in number of boluses per day, nor change to their bolus to basal ratios were observed. It may be that increased interaction with a novel technology resulted in users augmenting their basal insulin delivery rates themselves, while the increased number of BGL data resulted in an increase in the proportion of bolus insulin delivered as a ‘correction’, as has been noticed previously with the bolus calculation algorithm used by this automated device; 25 unfortunately, this is not possible to elucidate from pump download data.
It remains the case that despite increased allied health support and continued enhancement of diabetes technologies, the majority of youth with type 1 diabetes still do not achieve target HbA1c. 4 This is the primary incentive driving closed-loop technology development but until this is a clinical reality, supported BGL monitoring remains paramount. Future research direction in this area could ideally focus on augmentation of BGL frequency in those individuals displaying suboptimal testing. Results of the above post hoc subgroup analysis may merit further prospective study, ideally in a cohort with a wide range of baseline metabolic control. The challenges of participant withdrawal should be noted in future trial design, especially in a less engaged cohort.
Footnotes
Acknowledgements
We gratefully acknowledge all participating children and adolescents and their parents and guardians for their collaboration. All automated pump equipment was supplied by Roche for use in this study. This company did not have any further input into study design, analysis, or reporting of findings. We are also grateful to M. Peyrot and R. Rubin (Loyola College, Maryland, USA), who granted permission for use of the IDSRQ in this study, and to the CEBU department of Murdoch Childrens Research Institute, who gave assistance with trial design and statistical analysis of results.
Abbreviations
BGL, blood glucose level; CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin; IDSRQ, Insulin Device Satisfaction Rating Questionnaire; IQR, interquartile range; SD, standard deviation; TDD, total daily dose; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
