Abstract

Keywords
The new long-acting insulin Degludec and its new prefilled pen device, Flex Touch® (Novo Nordisk A/S, Bagsvaerd, Denmark), have been used since March 2013 in Japan. We conducted a multicenter, observational study involving private and public diabetes clinics in the west Tokyo area and recruited diabetes patients on intensive insulin therapy, basal bolus therapy with basal insulin injections once or twice a day (using the previous basal insulin). We used a questionnaire comprising 15 questions about usability of the device (eg, needle attachment, ergonomics, unit setting, injection, click sound, and appearance, etc) rating on a scale of 1 (very bad) to 6 (very good), and 1 question on the pain experienced rated by the visual analog scale from 1 (most painful) to 6 (not at all painful). Subjects were also asked to give the devices a score out of 100. They were handed the questionnaires before, and 1 month after, switching basal insulin, and asked to mail them back to our research office. Among 107 subjects, 92 (86.0%) answered the first questionnaire asking about previous devices, and 86 (80.4%) answered about the new device by the second questionnaire. In all, 67% had type 1 diabetes. Their average age was 58.8 (standard deviation 16.6) years old. Average duration of illness was 18.6 (12.3) years. Their HbA1c at the start was 8.1% (1.1). In the comparison between prior to and after switching to the new device (Table 1), it was better evaluated overall (75.2 vs 79.7; paired
Comparison of Scores Pre– Versus Post–Switching to the New Device.
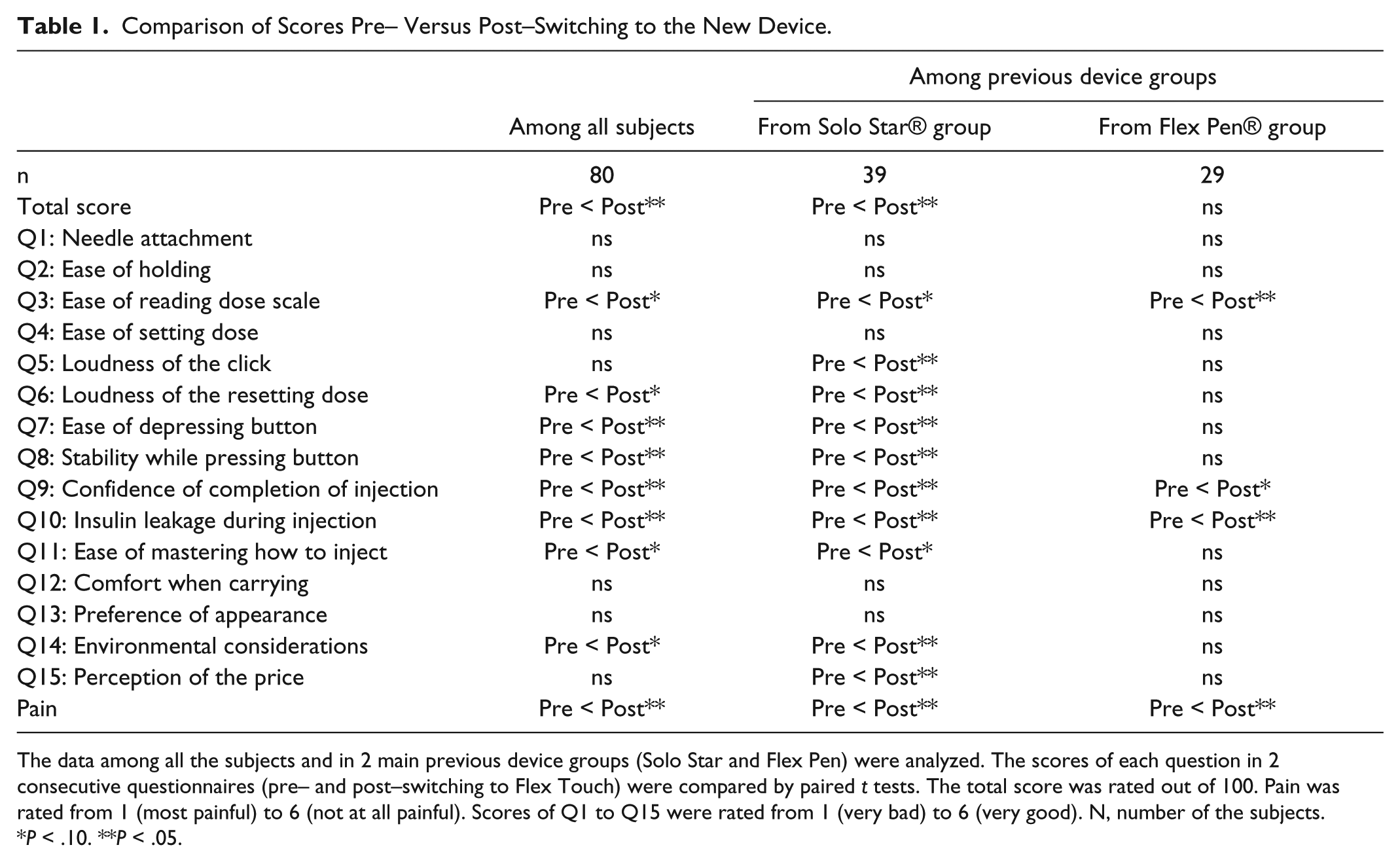
The data among all the subjects and in 2 main previous device groups (Solo Star and Flex Pen) were analyzed. The scores of each question in 2 consecutive questionnaires (pre– and post–switching to Flex Touch) were compared by paired
Flex Touch is a newly designed device and has recently begun to be used in Japan, one of the first countries in the world where it is used. Recent prefilled devices have already been well developed in terms of usability; the evaluation of existing pen devices was already high enough with around 80 out of 100 as a total score. Still, the new prefilled device was evaluated higher in total. The indicated improvements were in perception of pain, completion of the injection, and leakage. It was developed to require no extension of stroke, 1 to require less force to depress even when injecting a higher dose, 2 and to make a clear and relatively loud clicking sound, targeting the elderly and patients who have reduced manual dexterity. 3
Overall, the new prefilled pen device received high evaluation scores from patients in real-world clinical practice (on intensive insulin therapy). The reported improvements included less pain, less insulin leakage, and more confidence in completion of the injection. Preferences related to appearance, click sound, and cost were variables depending on individual responders.
Footnotes
Acknowledgements
We thank our patients and the diabetologists who participated in our multicenter observational studies, the TRAIN Study. This study was accomplished through the cooperation of Dr Mitsutoshi and Ms Noriko Kato (Kato Clinic), Dr Kazuo Kanno (Kanno Clinic), Dr Eitaro Kodani (Nippon Medical School Tama-Nagayama Hospital), Dr Hiroko Kondo, Dr Nobuyuki Yoshida, Dr Michiko Morita (Kondo Clinic), Dr Ryuji Sato (Kunitachi Clinic), Dr Harumi Daikoku (Showa General Hospital), Dr Hiroshi Takamura, Dr Akio Ueki (Takamura Clinic), and Dr Masaro Takesue (Takesue Clinic).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
