Abstract

Keywords
Reluctance to prescribe and/or intensify insulin therapy in patients with type 2 diabetes has been linked to clinicians’ perceptions regarding patient adherence and concerns about hypoglycemia.1,2 However, it is possible that the underlying reason may be a lack of understanding regarding the real and perceived differences between individuals with type 1 diabetes and those with type 2 diabetes who could benefit from intensified insulin therapy. Are the differences between these 2 populations clinically relevant? To explore this question, we compared the baseline characteristics and outcomes of individuals with type 1 and type 2 diabetes who were enrolled in the Automated Bolus Advisor Control and Usability Study (ABACUS) trial 3 in a post hoc analysis to identify potential differences and/or similarities that clinicians should consider when prescribing multiple daily insulin injection (MD) therapy for their type 2 diabetes patients.
Baseline and outcome data from matched type 1 diabetes (n = 16) and type 2 diabetes (n = 16) patients were used in this analysis, using ranked scores for gender, age, HbA1c, duration of diabetes, carbohydrate counting competency, and psychosocial measures. Use of the bolus advisor (intervention) and insulin rule sets (control) were also assessed.
Baseline data revealed no difference in HbA1c levels, carbohydrate counting competency, or psychosocial measures between participants with type 1 and type 2 diabetes (Table 1). Conversely, significant differences were seen in age and diabetes duration. Noticeable differences were seen in BMI, insulin dosages, frequency of daily insulin bolusing and frequency daily self-monitoring of blood glucose (SMBG). At study end, HbA1c levels for type 2 participants were similar to those with type 1 diabetes. Moreover, there were no between-group differences seen hypoglycemia, carbohydrate counting competency, or psychosocial measures.
Baseline Characteristics of Matched Type 1 and Type 2 Diabetes Patients Enrolled in the ABACUS Trial.
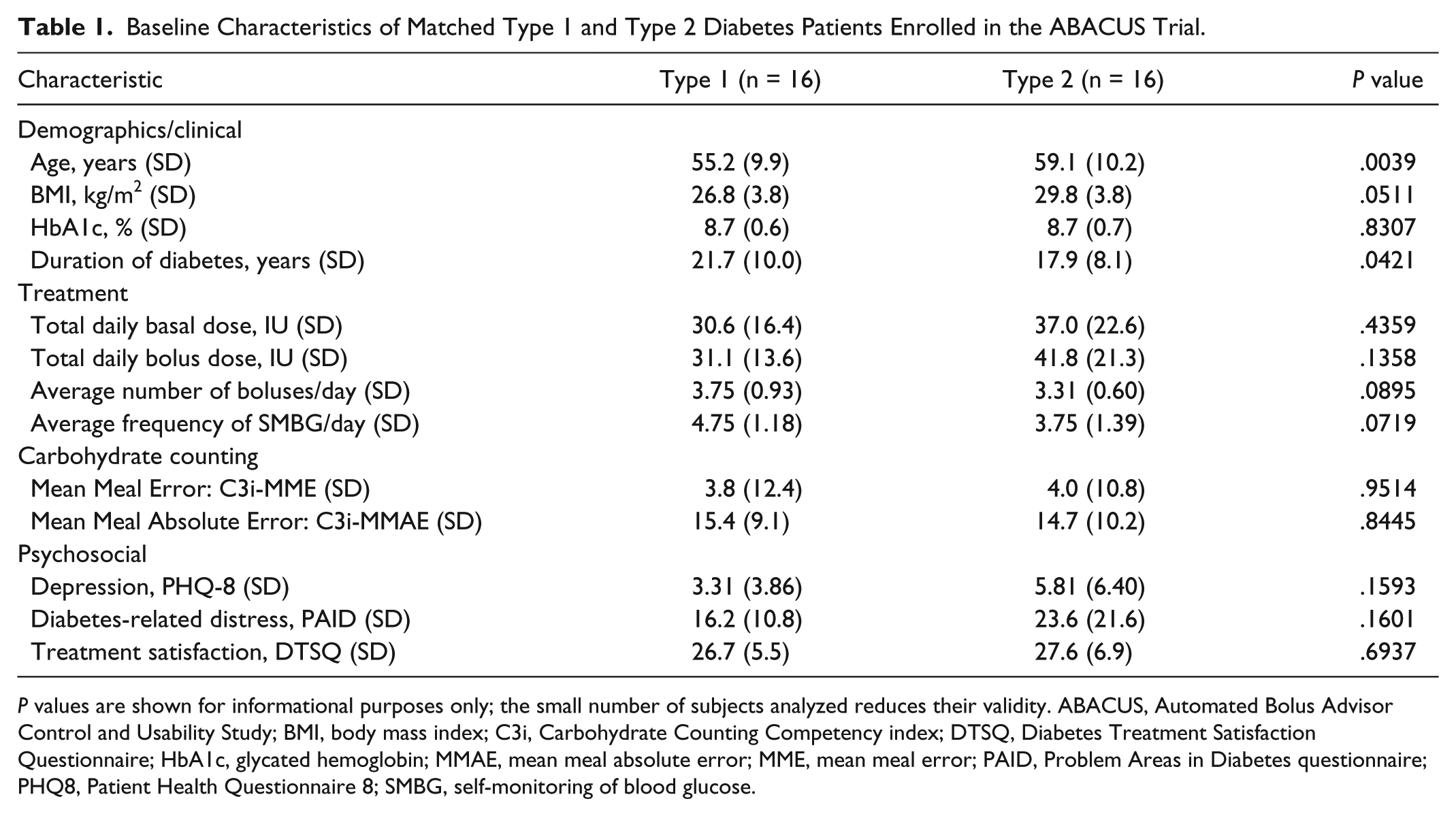
P values are shown for informational purposes only; the small number of subjects analyzed reduces their validity. ABACUS, Automated Bolus Advisor Control and Usability Study; BMI, body mass index; C3i, Carbohydrate Counting Competency index; DTSQ, Diabetes Treatment Satisfaction Questionnaire; HbA1c, glycated hemoglobin; MMAE, mean meal absolute error; MME, mean meal error; PAID, Problem Areas in Diabetes questionnaire; PHQ8, Patient Health Questionnaire 8; SMBG, self-monitoring of blood glucose.
Within the experimental group, the percentage of bolus advisor use by participants with type 1 (n = 7) and type 2 (n = 7) diabetes for all bolus possible bolus dosage calculations was similar, as were the average number of daily advices sought and accepted during the course of the study. Among type 2 (n = 9) and type 1 (n = 9) participants within the control group, we saw no apparent differences in the number of daily boluses in which the correct insulin-to-carbohydrate (I:CHO) ratios and or insulin sensitivity factor (ISF) settings were used.
Our analysis of this matched cohort of participants with type 1 and type 2 diabetes suggests that the clinical needs of these 2 populations are mostly similar. Although, this post hoc analysis of the ABACUS data was not powered to obtain definitive conclusions regarding differences between individuals with type 1 and type 2 diabetes, our findings suggest that both populations behave in a similar manner and have similar outcomes. Moreover, these findings suggest that both populations benefit from intensive insulin therapy when the appropriate tools, clinical guidance and ongoing support are provided. Although larger studies are needed to further elucidate the similarities and differences between groups, it would be prudent at this time for health care providers and payers to provide similar health care and education to all patients who would benefit from intensive insulin therapy.
Footnotes
Abbreviations
ABACUS, Automated Bolus Advisor Control and Usability Study; BMI, body mass index; C3i, Carbohydrate Counting Competency index; DTSQ, Diabetes Treatment Satisfaction Questionnaire; HbA1c, glycated hemoglobin; I:CHO, insulin-to-carbohydrate; MDI, multiple daily insulin injections; MMAE, mean meal absolute error; MME, mean meal error; PAID, Problem Areas in Diabetes questionnaire; PHQ-8, Patient Health Questionnaire 8; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CGP and WK received consulting fees for their involvement in the study design, study implementation, and preparation of the manuscript. CGP has received consulting fees from Roche Diagnostics and Dexcom, Inc (San Diego, CA). RSW is an employee of Roche Diagnostics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study was provided by Roche Diagnostics, Indianapolis, Indiana, USA.
