Abstract
Real-time monitoring of physiological glucose transport is crucial for gaining new understanding of diabetes. Many techniques and equipment currently exist for measuring glucose, but these techniques are limited by complexity of the measurement, requirement of bulky equipment, and low temporal/spatial resolution. The development of various types of biosensors (eg, electrochemical, optical sensors) for laboratory and/or clinical applications will provide new insights into the cause(s) and possible treatments of diabetes. State-of-the-art biosensors are improved by incorporating catalytic nanomaterials such as carbon nanotubes, graphene, electrospun nanofibers, and quantum dots. These nanomaterials greatly enhance biosensor performance, namely sensitivity, response time, and limit of detection. A wide range of new biosensors that incorporate nanomaterials such as lab-on-chip and nanosensor devices are currently being developed for in vivo and in vitro glucose sensing. These real-time monitoring tools represent a powerful diagnostic and monitoring tool for measuring glucose in diabetes research and point of care diagnostics. However, concerns over the possible toxicity of some nanomaterials limit the application of these devices for in vivo sensing. This review provides a general overview of the state of the art in nanomaterial-mediated biosensors for in vivo and in vitro glucose sensing, and discusses some of the challenges associated with nanomaterial toxicity.
Glucose is involved in major adenosine triphosphate (ATP) producing pathways: glycolysis, oxidative phosphorylation, as well as numerous other metabolic and regulatory processes such as the glucose/insulin pathway. As such, maintenance and regulation of glucose metabolism and its downstream processes are critical for proper physiological function. The severity of metabolic diseases such as diabetes depends on the degree to which the glucose transport system is impaired. This review describes recently developed biosensor and nanosensor technologies for monitoring in vivo or in vitro glucose in diabetes research.
Commercially Available Glucose Biosensors
The current market for glucose biosensors, especially those for home glucose monitoring, is dominated by finger-prick type blood glucose sensors. Most popular are the FreeStyle Lite by Abbott, the Countor by Bayer, the OneTouch Ultra2 by LifeScan, and the Accu-Chek by Roche. These sensors detect H2O2 using amperometric detection, providing fast response times (~5 s) within a dynamic sensing range (10.8094 -599.9191 mg/dl) for measuring blood glucose levels. Other ex vivo blood glucose biosensors use a multiplexing approach, which allows for multianalyte monitoring on a point-of-care diagnostics platform. These include the Precision Xtra Advanced diabetes management system and the blood analyzer by i-STAT, which is now a part of Abbott. The i-STAT analyzer is a handheld analyzer which contains a microchip capable of simultaneously monitoring numerous clinically important biomarkers from blood samples, including glucose and lactate. 1 Recent research has been advancing electrochemical and optical glucose monitoring technologies in both sensitivity and accuracy compared to currently available commercial biosensors. The following sections review these emerging technologies.
Fundamental Working Mechanism for Glucose Biosensors
Currently, several techniques exist to study the transport of glucose including radiolabeling, microfluorometric assays, and spectrometric techniques, among others.2-4 Although each of these techniques are useful for measuring glucose concentration in small volumes, use of these devices for studying real-time glucose transport in living cells is a challenge. Radiolabeling, microfluorometric assays, and spectrometry have a relatively low temporal resolution (on the order of tens of minutes), and require bulky supporting equipment for measuring and recording signals. In addition, many protocols require destructive sampling procedures such as cell homogenization or addition of external reagents (eg, radiotracers). Thus, much research in the past decade has focused on the development of rapid, minimally invasive biosensors that do not depend on addition of external reagents.
Since the invention of the glucose enzyme electrodes by Clark and Lyons, 5 a wide variety of sensors and biosensors have been developed for measuring glucose in biological fluids (reviewed by Wang, 6 among others). Modern glucose biosensors facilitate real-time, continuous monitoring of glucose concentration in liquid samples as small as 10 nL by miniaturizing the sensors to the micro and nano scale. These microsensors and nanosensors have a high spatial resolution that allows measurement in single cells or isolated organelles.
Electrochemical Glucose Biosensors
Electrochemical glucose biosensors typically monitor oxidative current produced by glucose oxidase (GOx) (Figure 1). GOx catalyzes free glucose into gluconic acid and hydrogen peroxide (H2O2). H2O2 produced by GOx deprotonates to produce free protons, dissolved oxygen and 2 electrons under an external oxidative potential. The measured electrical signal is directly proportional to glucose concentration (Figure 1).
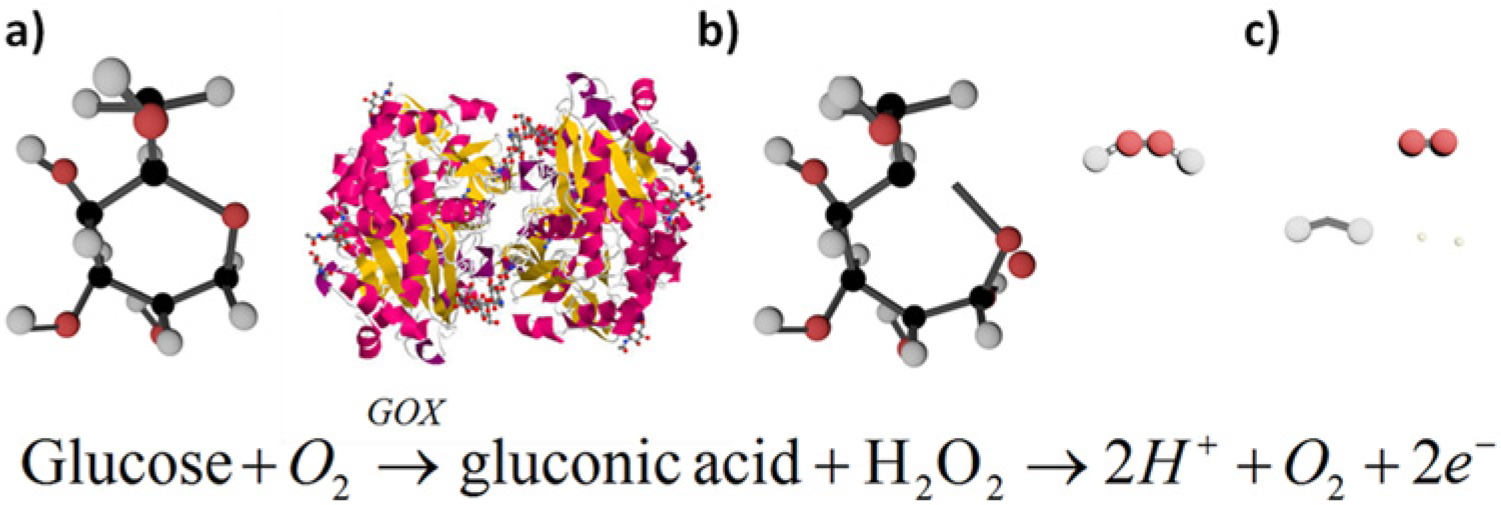
Basic working principle for glucose biosensors (molecules are not drawn to scale). (a) Glucose binds in the enzymatic binding pocket of glucose oxidase (GOX). (b) An applied potential catalyzes the oxidation of glucose to gluconic acid and hydrogen peroxide. (c) Hydrogen peroxide dissociates to O2, 2 H+, and 2 free electrons; electrons are measured using electrochemical or optical techniques.
While electrochemical glucose biosensing continues to be the most widely used method of monitoring in vivo and ex vivo glucose concentrations, these biosensors can be restrictive. This is particularly true for in vivo glucose sensing, where endogenous electroactive species cause interference (ie, false positive signals or noise). Some electrodes have been shown to damage surrounding cells/tissue and sensitivity is limited to the amount of active immobilized enzyme.7,8
Optical Glucose Biosensors
Fluorescent-based glucose sensing is extremely sensitive, and is capable of detecting glucose down to the single-molecule level. 9 Fluorescence can be carried out through a variety of sensing modalities including those that use enzymes, 10 plant lectin (eg, Concanavalin A 11 ), bacteria, 12 or intrinsic cellular fluorescence. 13 Due to the small size of fluorescent probes such as dyes and quantum dots (< 100 nm), sensors can be biofunctionalized and loaded into cells by diffusion without endocytosis.14-16
Fluorescent probes can be interrogated remotely using an external UV excitation source that can penetrate several centimeters into tissues.17,18 Use of UV excitation can instigate distinct and programmable photoillumenscence (PL) responses in quantum dot/dye bioconjugates through Förster resonance energy transfer (FRET).17,19,20 Such PL measurements can be captured at distinct time-points to eliminate interference from light scattering in tissues and to correct for photobleaching or fluorophore loss through diffusion or degradation.17,21,22 FRET-based biosensors can also spatially resolve target analyte as PL emissions from acceptor/donor fluorophore decays as a sixth power function of the distance (1/R 6 ) between them. FRET signals attenuate rapidly when fluorophore-to-fluorphore distances reach above 10 nm. Thus fluorescent-based glucose biosensors are well suited for noninvasive, in vivo sensing and continuous glucose monitoring. Optical glucose sensors are also useful for monitoring oscillations in glucose flux, and their associated biochemical pathways such as the feedback cascade that involves glucose catabolism, ATP production, calcium/potassium transport, and insulin exocytosis.23,24 Medintz and Mattoussi provide a detailed review of FRET-based biosensors using quantum dots and their application in the life sciences (Figure 2). 25
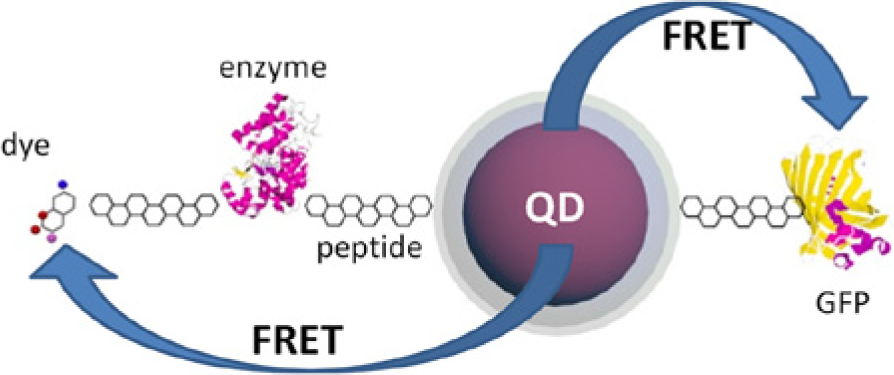
Conceptual schematic of a Förster resonance energy transfer (FRET) interaction between quantum dots (QD) and enzyme-dye conjugates (left) as well as QD-green fluorescent protein FRET pairing.
Nanomaterial-mediated Biosensors
Improving Biosensing Platforms
The major challenges with developing glucose biosensors are (1) efficient immobilization of GOx on a conductive metal and (2) low signal to noise ratio in biological fluids. Immobilization of GOx can be achieved by a number of techniques, including physical entrapment in polymers,26-28 self-assembled monolayer structures,29-31 and other structures based on covalent bonding of GOx. 32 As discussed in the following, signal to noise ratio can be improved by integrating catalytic nanomaterials such as carbon nanotubes (CNTs), graphene, or metal nanoparticles into the biosensor design.33-38 A wide variety of devices have been built for in vivo patient monitoring and theranostics, as well as for in vitro studies of cell/tissue physiology including glucose in blood, tears, and saliva. This review focuses on minimally invasive, real-time microsensors and nanosensors.
Nanomaterials (1-100 nm length scale) are widely used to enhance the performance of glucose biosensors. Unique spatial and charge properties of nanomaterials are associated with its high surface-to-volume ratio that promote superior electrical, optical, thermal, and catalytic properties when compared to the bulk-sized material. 39 Due to the rate of electron transfer being inversely proportional to the distance between the enzyme and the electrode, nanoparticles are much more capable of decreasing enzyme-to-electrode distances than bulk-sized materials. 40 Nanoparticles can also be easily absorbed into cells for in vivo / in vitro sensing applications. 41 Furthermore, noble metal nanoparticles have shown to exhibit enhanced localized surface plasmon resonance (SPR), and thus can be used for optical biosensing using techniques such as surface-enhanced Raman spectroscopy (SERS). 42 Thus, in recent years, research into nanostructured glucose biosensors has yielded previously unattainable sensitivity, lower detection limit, and a wide linear sensing range. In particular, devices utilizing CNTs, graphene, electrospun nanofibers, and nanoparticles have yielded some of the most promising results.
Carbon-nanotube-based Biosensors
CNTs have been used in biosensing applications to improve sensor response time, sensitivity, and detection limit.43,44 CNTs have large aspect ratios that increase the surface area of the electrode for enhanced heterogeneous charge transport and increased docking points for biorecognition agents such as GOx.45,46 CNTs also have exceptional electrical conductivity properties and electrochemical charge transport. 47 The enhancement in electrochemical reactivity occurs at CNT defect sites where breaks in C-C bonds exist,48,49 and oxygenated species such as carboxylic acids, alcohols, and quinines form.50,51 Likewise metal impurities found within the CNTs may also bolster their electrocatalytic properties during electrochemical biosensing. 8
Glucose biosensing has benefited immensely from CNT-based electrochemical biosensors. For example, highly sensitive nanoelectrode arrays of vertically aligned single-walled CNTs, that is, SWCNTs (diameter approximately 1-3 nm) grown from a silicon substrate for glucose sensing have been developed. 52 GOx was covalently linked to carboxyl-tipped nanotubes and used to sense glucose at concentrations as low as 1.4412 mg/dl. Furthermore, the CNT array electrode was able to operate at a negative working voltage (–0.2V), eliminating electrochemical interference from common blood interferents (ie, uric acid, ascorbic acid, and acetaminophen) that oxidize at positive working potentials. Likewise, multiwalled CNT (MWCNT) arrays with a much thicker density were grown on a silicon substrate from a nickel catalyst for glucose sensing. 53 Functionalization of the CNT surface was less complex in this structure, as GOx could be directly absorbed onto the surface of the MWCNTs instead of needing to use covalent linking schemes with the SWCNT design. This enzyme immobilization scheme produced promising results as the enzyme retained 86.7% of initial enzyme activity after a 4-month period.
Perhaps the best performing CNT-based glucose biosensors are those that combine CNTs with metallic nanoparticles. A CNT-based biosensor modified with palladium (Pd) nanoparticles and Nafion (anion repellant) was capable of detecting glucose within a linear range from 2.7023 mg/dl to 216.1871 mg/dl while minimizing interference from uric and ascorbic acid. 54 Yang and coworkers show how CNTs modified with cobalt nanoparticles could reach an extremely low detection limit of 9.01 × 10−2 mg/dl. 55 In terms of glucose detection limit and sensing range, one of the top performing CNT/metal nanoparticle hybrid biosensors 33 was created by consecutively electrodepositing Pd and then gold (Au) to form Au coated Pd nanocubes on SWCNTs arrays that were grown from a porous anodic alumina template.34,35,56 A very low glucose detection limit of 2.34 × 10−2 mg/dl and a wide linear sensing range of (1.80 x10−2−900.7795 mg/dl) were achieved with this Au/Pd-SWCNT. Another slightly more sensitive (glucose detection limit of 6.85 × 10−3 mg/dl) version of this biosensor was created using platinum (Pt) nanospheres.35,36 Such advanced CNT-metal nanoparticle hybrid biosensors are capable of detecting blood glucose levels in blood where the physiological range for blood glucose of healthy and diabetic patients can range from 64.8561 mg/dl to 135.1169 mg/dl and 19.8171 mg/dl to 374.7242 mg/dl, respectively. 57 In addition, these nanomaterial-mediated electrochemical biosensors open the door to noninvasive glucose monitoring through saliva and tears where glucose level concentrations can be in the micromolar concentration range.58,59
Graphene-based Biosensors
Graphene-based glucose biosensing is a burgeoning field of research. Graphene is essentially an unrolled SWCNT consisting of a monatomic 2-dimensional sheet of sp 2 bonded carbon. 60 As with CNTs, graphene displays outstanding material properties including high thermal and electrical conductivity as well as excellent tensile strength. 60 Likewise, defect sites within the C-C lattice are conducive to heterogeneous charge transport; graphene defect sites are well suited for subsequent electrochemical metal decoration and immobilization of biorecognition agents such as GOx.60,61 Recent reports suggest that graphene displays single-electron Nernstian behavior which correlates to rapid electron transfer in electrochemical sensing. 62 Perhaps one of the more important inherent properties for potentially measuring glucose is the environment graphene produces for cell immobilization. Chen et al demonstrated the successful growth of mouse fibroblast cells (L-929) cells on graphene paper and recorded adhesion and proliferation rates similar to L-929 cells cultured on polystyrene tissue. 63 The use of these cell types was a great indicator of biocompatibility as mouse fibroblast cells (L-929) are typically used in analyzing the cytotoxicity of materials. A similar cell culture study was performed on graphene oxide. 64 In this report ARPE-19 cells cultured on graphene oxide (GO) displayed excellent adhesion and differentiation after a 72-hour culture time as verified through fluorescence microscopy. Thus, potential immobilization of cells onto graphene or graphene oxide for monitoring glucose is indeed feasible, and the biocompatibility of grapheme indicates that future implantable devices are on the horizon.
Glucose biosensing with graphene has produced promising results that are comparable to, or better than, CNT-based GOx biosensors. Kang et al created a graphene-based glucose biosensor by sonicating graphene (etched from graphite sulfuric acid, nitric acid, and potassium chlorate) with chitosan and drop-coated the mixture onto a glassy carbon electrode. 65 This graphene-based biosensor was able to measure glucose with a detection limit of 0.3603 mg/dl and a linear sensing range of 1.4412 mg/dl to 216.1871 mg/dl. Another graphene-based glucose biosensors utilized the conductive polymer polypyrrole (Ppy) to encapsulate and entrap graphene and GOx onto a glassy carbon electrode. 66 This PPy-graphene biosensor was used to sense glucose with a detection limit of 5.4 × 10−2 mg/dl and a linear sensing range of 3.60 × 10−2−0.7206 mg/dl.
As with the CNT-biosensors, graphene biosensors fabricated with metal nanoparticles yielded some of the most promising results. For example, exfoliated graphite nanoplatlets (xGnPs) were dispersed in ethylene glycol with a Pt precursor, sonicated, and centrifuged to form xGnPs decorated with Pt nanoparticles. Nafion was then used to stabilize the nanoplates together GOx. 67 This biosensor showed high glucose sensitivity with a glucose detection limit of 1.80 × 10−2 mg/dl and a linear sensing range of 18.0156-360.3118 mg/dl. Recently, chemical vapor deposition was used to grow multilayered graphene petal nanosheets (MGPNs) on a silicon-based surface for use in glucose biosensing (Figure 3). 68 Electrochemical deposition of Pt nanoparticles deposited on the 3D graphene petals were followed by electrochemical deposition of the conductive polymer poly(3,4-ethylenedioxythiophene)-poly(styrenesulfonate) (ie, PEDOT:PSS) doped with GOx. The size, density, and morphology of the Pt nanoparticles were altered by changing the magnitude of the current pulse used to deposit the nanoparticles. This Pt-MGPN biosensor obtained a lower glucose detection limit (5.40 × 10−3 mg/dl) and wider linear sensing range (0.1801-900.7795 mg/dl) than comparable nanostructured biosensors. Such a broad linear glucose sensing range enables glucose monitoring in blood, 57 saliva, 58 tears, 59 and urine, 69 which would permit new noninvasive sensing protocols where glucose from numerous serum samples could be monitored simultaneously. 68
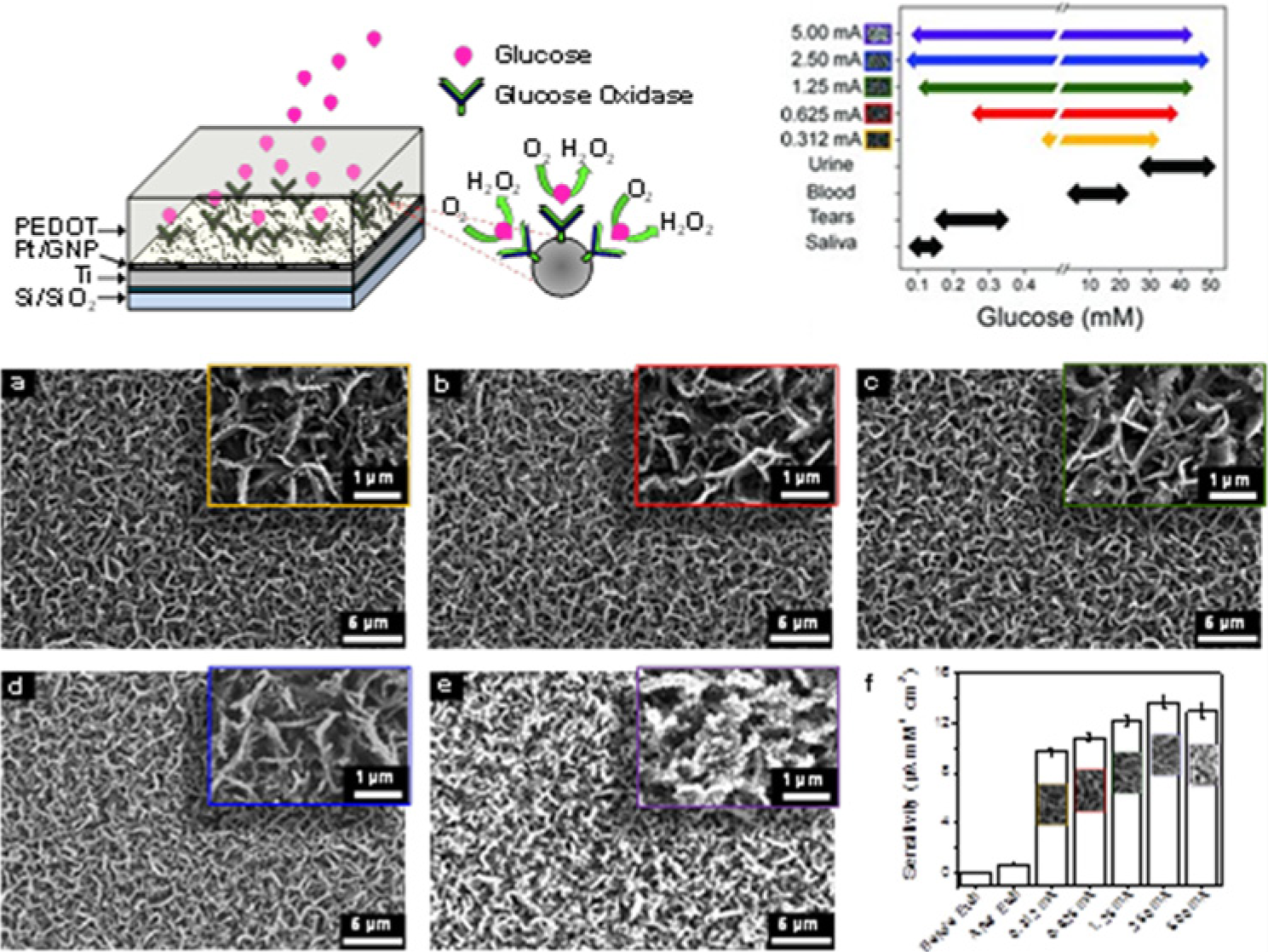
(Top Left) Tilted cross-sectional schematic illustrating the GOx/PEDOT biofunctionalized PtNP-MGPN glucose biosensor with adjacent magnified view portrayal of GOx immobilized on a single PtNP. Glucose binds within the GOx enzymatic pocket producing H2O2 while consuming O2. (a-e) Field emission scanning electron microscopy (FESEM) micrographs of PtNPs electrodeposited on MGPNs. Current pulses (500 ms) of (a) 312 µA, (b) 625 µA, (c) 1.25 mA, (d) 2.5 mA, and (e) 5.0 mA were used to electrodeposit Pt nanoparticles of distinct size and density onto the MGPNs. (f) Bar graph displaying the H2O2 sensitivity of the MGPN electrode (before and after the oxygen plasma etch) and the PtNP-MGPN electrodes. Errors bars show standard deviation for 3 different experiments. (Top Right). Glucose sensing ranges of the Pt-MGPN glucose biosensors (Pt electrodeposition current pulses of 312 µA, 625 µA, 1.25 mA, 2.5 mA, and 5.0 mA) as compared to glucose levels found in urine, blood, tears, and saliva. Reprinted with permission from Advanced Functional Materials. 68 Copyright 2012 Wiley-VCH.
Nanoparticle and Nanoparticle Array-based Glucose Biosensors
Nanoparticles are well suited toward biosensing due to their enhanced catalytic properties, electron transfer, and their ability to be used in biomolecule labeling and adsorption. 41 The small size of nanoparticles improves electrochemical, enzymatic biosensor performance by increasing electron transfer rates by shortening enzyme-to-electrode distances. 40 Noble metal nanoparticles can also enhance localized SPR and accordingly can improve optical biosensors based on SERS. 42
SERS-based glucose biosensors have been a widely studied optical-based approach to glucose biosensing. Shafer-Peltier et al demonstrated the first systematic study of directly monitoring glucose concentrations via SERS with silver nanoparticles. 70 This study demonstrated that SERS could detect glucose concentrations over a wide (0-4503.8975 mg/dl) and clinically relevant (0-450.38 mg/dl) concentration range. A SERS-based approach was developed for glucose sensing by monitoring the aggregation (via concanavalin A (Con A) and dissociation of dextran coated 20-nm gold particles and monitoring changes in plasmon absorption and dissociation. 71 By modifying the amount of dextran or Con A used in nanoparticle fabrication, the glucose sensing range could be tuned to detect μM to mM concentrations that correspond with varying glucose concentrations found in physiological fluids (eg, tears, blood, and urine).
Solution suspensions of nanoparticles have also been used to sense glucose via other electrochemical and optical methods. Research groups have immobilized the enzymes on magnetic nanoparticles due to the ability of the nanoparticles to be easily delivered and recovered in biomedical applications through magnetic attraction. For example, Rossi et al immobilized GOx on magnetite (Fe3O4) nanoparticles 20 nm in diameter that were capable of detecting glucose concentrations up to 360.3118 mg/dl for 3 months when stored at 4°C. 72 Quantum dots composed of manganese-doped zinc sulfide (ZnS), functionalized with GOx, and suspended in solution was able to sense detection limit of 0.0540 mg/dl and 2 linear ranges from 0.1802 to 1.8016 mg/dl and from 1.8016 to 18.0156 mg/dl via a phosphorescent detection mode. 73
Nanoparticles immobilized on electrode surfaces have been used in a wide variety of glucose biosensors with metallic nanoparticles and quantum dots.54,74,75 By separating the nanoparticles/nanowires between nonconductive insulating material (eg, polycarbonate, alumina) in an ordered/semiordered arrangement, arrays of individual nanoelectrodes can be created on a single electrode surface. 76 These nanoelectrode arrays experience improved signal-to-noise ratios, 77 enhanced mass transport, 76 and improved detection limits 78 as compared to traditional macroelectrodes.
Yang et al developed an array of platinum nanowires (250 nm in diameter) grown in polycarbonate membrane via an electrodeposition method that was able to detect glucose with a wide linear range (0.018 to 540.477 mg/dl) when functionalized with glucose oxidase. 79 Furthermore, these Pt nanoelectrode arrays were also able to detect glucose in actual blood samples. Pt nanoparticles have also been inserted into arrays of CNTs and functionalized with the enzyme glucose oxidase by Wen et al. This Pt-CNT biosensor was capable of sensing glucose with a wide range (0.0288-207.1793 mg/dl) and low detection limit (0.9909 mg/dl). 80 Furthermore, other studies have demonstrated how nanoparticles can be electrodeposited on arrays of horizontally aligned CNTs grown from a porous anodic alumina template.34,56 Pt nanoparticles were electrodeposited on these CNT arrays with distinct current densities to alter the density of the nanoparticles while GOx was covalently linked to the particles to create enzymatic glucose biosensors. 35 By increasing the relative density of Pt nanoparticles deposited on the CNTs, the linear glucose sensing range could be increased from a 5.4047- 270.2334 mg/dl range to a 1.8016-360.3118 mg/dl range. Similarly, the detection limit showed an improvement from 1.3331 mg/dl to 0.1045 mg/dl (S/N = 3). Gold nanowires were grown in a similar porous anodica alumina template via an electrodeposition method. 81 The nanowires were chemically etched back even with the template so only tips of the nanowires or circular tips were exposed during electrochemical sensing. By covalently immobilizing GOx via thiol linkage, the gold nanoelectrode arrays were able to sense glucose with detection limit of 1.8016 mg/dl and with a linear sensing range up to 378.3274 mg/dl.
Risks of Real-time Biosensors
As with any new technology, there are risks associated with nanomaterial-mediated biosensors. Implantable devices suffer from some challenges including postimplantation complications and insufficient miniaturization to reduce trauma from implantation. 82 To reduce traumatic effects on the implant patient, indirect glucose monitoring through subcutaneous implantation has been generally favored over the direct method of vascular bed implantation. 83 Hydrogel platforms have also been shown to be more biocompatible and reduce protein adsorption which subsequently reduces postimplantational effects.84,85
The cytotoxicity of carbon nanomaterials such as single-walled CNTs, MWCNTs, and graphene is still unclear.86,87 This is a particular concern for implantable devices that depend on the catalytic action for highly sensitive detection of glucose. Sohaebuddin et al. have investigated the effects of various nanometal oxides and MWCNTs on physiologically different cell types and concluded that the nanomaterial toxicity was highly dependent on the type of nanomateriral, size, concentration and the function of the target cell. 88 However, this study exposed cell lines to a constant dosage of free nanoparticles which differ from most electrochemical sensing applications where the nanomaterials are immobilized on an electrode (i.e., nanoparticle leaching is expected to be minimized) and the expected concentration exposure of cells is expected to be much less. Au nanoparticles have shown cytotoxicity in some studies of mammals, but in other studies the nanoparticles are benign and are excreted through urine. 89 The toxicity of quantum dots is well known, and is major deterrent against mainstream use for fabricating implantable devices.90-92
There have been many concerns regarding the cytotoxicity of quantum dots in biological systems. This is due to many quantum dots being composed of heavy metal ions such as cadmium. The release of these heavy metal ions can potentially cause cytotoxic effects. There is some ambiguity to the toxic effects of quantum dots due to the varying types of quantum dots and the variability in the toxic threshold for certain cell lines and types. 93 It has been shown that one of the keys to reducing the cytotoxic effects is to provide a shell coating for the quantum dots that has a high colloidal stability and high water solubility which would prevent particle aggregation and facilitate excretion of the quantum dot particles reducing toxicity. 94 Specifically, polyethylene glycol terminated microcapsules aid in preventing the cellular uptake of particles.94,95
Despite the challenges of minimizing associated health risks, nanomaterials continue to be widely studied for various glucose sensing paradigms. The high sensitivities achieved with nanostructured glucose biosensors offer the potential for minimally invasive or noninvasive glucose sensing for diabetic patients as well as the potential for monitoring minute glucose oscillations during cellular metabolism. Such strides in glucose sensing technologies have the potential to revolutionize the treatment and prognosis of myriad diseases including diabetes.
Conclusion
This study aimed to review the current and developing state of technology concerning nanomaterial-based glucose sensors and nanosensors for diabetes monitoring and research. Emerging technologies have allowed for a considerable improvement to optical glucose biosensing, but electrochemical techniques still remain the most prevalent. Though some concerns of toxicity and risks remain, implementation of nanomaterials, in particular CNTs and graphene, have improved the sensing range and sensitivity of glucose biosensors expanding their use to more systems where the need to achieve a reliable and consistent signal is paramount. These nano-inspired glucose biosensors hold tremendous promise at overcoming the low temporal/spatial resolution and large size (which prohibits in vitro/in vivo glucose sensing) of conventional glucose biosensors.
Footnotes
Abbreviations
ATP, adenosine triphosphate; Au, gold; CNT, carbon nanotube; FRET, Förster resonance energy transfer; GO, graphene oxide; GOx, glucose oxidase; H2O2, hydrogen peroxide; MGNP, multilayered graphene petal nanosheets; MWCNT, multiwalled carbon nanotube; Pd, palladium; PEDOT:PSS, poly(3,4-ethylenedioxythiophene)-poly(styrenesulfonate); PL, photoilluminescence; Ppy, polymer polypyrrole; Pt, platinum; SERS, surface-enhanced Raman spectroscopy; SWCNT, single-walled carbon nanotube; xGnP, exfoliated graphene nanoplatelets; ZnS, zinc sulfide.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the Agricultural and Biological Engineering Department at UF and the UF Early Career Award as well as the US Naval Research Laboratory and George Mason University for financial support.
