Abstract
SUPER GL compact is a bench-top analyzer for glucose, lactate, and hemoglobin concentrations. Glucose measurements in the biosensor are based on an enzymatic-amperometric reaction of glucose with glucose oxidase.
In this study, trueness and precision were assessed with Standard Reference Material 965b (National Institute of Standards and Technology, Gaithersburg, MD) for 2 SUPER GL compact (S1 and S2) and 1 YSI 2300 STAT Plus (Y) device, using a protocol based on CLSI EP05-A3.
Precision was similar among S1, S2, and Y. S1 and S2 exhibited negative bias at low concentrations and positive bias at high concentrations, whereas Y showed negative bias that increased with higher concentrations. Overall, SUPER GL compact’s performance was comparable to that of YSI 2300 STAT Plus.
Introduction
Since the Diabetes Control and Complications Trial provided evidence that diabetes complications risk can be reduced by performing blood glucose (BG) self-measurements, 1 they have been a major factor in diabetes therapy for people with insulin-dependent diabetes. Nowadays, many BG monitoring systems as well as a variety of continuous glucose monitoring (CGM) systems are available.
The analytical performance of these systems is typically established in method comparisons using laboratory analyzers and/or, for CGM systems, BG monitoring systems. YSI 2300 STAT Plus (YSI Life Sciences, Yellow Springs, OH) was very widely used, being perceived as a quasi-standard in performance studies in which laboratory analyzers were used for comparative glucose concentration measurements. YSI announced end of support for that model for July 2021. 2 Initially, YSI did not seek the Food and Drug Administration (FDA) clearance as a comparator method for successor devices, raising the question of potential comparator devices in future performance studies.
In November 2019, the Diabetes Technology Society held the YSI 2300 analyzer replacement meeting. 3 During that meeting, different companies expressed their interest in getting FDA clearance and setting up supply chains to accommodate the increasing interest in laboratory analyzers.
Among these companies was Dr. Müller Gerätebau GmbH (Freital, Germany). This article describes one of their analyzers, SUPER GL compact, its measurement technology, and results from an evaluation of trueness and precision using Standard Reference Material (SRM) 965b (National Institute for Standards and Technology, Gaithersburg, MA). Limited performance data of SUPER GL compact were published in 2014 in an ISO 15197 system accuracy study. 4 That kind of study setting, however, does not allow for structured, separate assessment of trueness and precision, and it might not reflect potential improvements in the analyzers used. The objective of this evaluation was to obtain current performance data for SUPER GL compact and YSI 2300 STAT Plus. Since such data are often not published outside of user manuals, a small-scale pilot study was performed in a manufacturer-independent laboratory to get initial, but current results.
Description of the Device
SUPER GL Compact
SUPER GL compact (Figure 1, Table 1) is a bench-top biochemistry analyzer for the determination of glucose, lactate and hemoglobin, manufactured in Germany by Dr. Müller Gerätebau GmbH. Since the market introduction in 2011 SUPER GL compact has been subject to continuous enhancements.

Pictures of the SUPER GL compact device and the biosensor (not to scale).
Device Specifications for Glucose Measurement.
The SUPER GL compact was designed to measure single samples or small series of up to six samples in about 45 seconds per sample. Whole blood samples have to be hemolyzed, and plasma and serum samples have to be diluted using the same ratio (10/20-µl sample mixed with 500/1000 µl hemolysis solution). These samples are stable for 12 hours at room temperature. The manufacturer offers a closed system of ready-to-use consumables for an easy and safe workflow.
The instrument is equipped with an automatic one-point calibration function. The traceable standard is measured twice for the initial calibration. For the initial calibration, two measurements are performed. The mean value of this double measurement is used, if the second value differs by <5% from the first value.
The stability of the initial calibration is checked every hour or if the temperature of the biosensor changes by more than 2°C.
IFCC recommends that glucose concentrations should only be given as equivalent to the glucose concentration in plasma, regardless of the sample examined. 5 The SUPER GL compact is able to determine the hematocrit in whole blood samples during measurement based on a photometric hemoglobin concentration measurement to optionally convert glucose values to plasma-equivalent values if hematocrit is between 10% and 70%. Otherwise, no conversion is performed.
Measurement Principle
Determination of glucose and lactate with the SUPER GL compact is based on an enzymatic-amperometric electrochemical measuring principle with a biosensor. 6 The measurement principle for glucose is based on an electrode coated with the enzyme glucose oxidase (GOD). The electrode is separated from the flow of liquid by barrier membrane layers. The glucose in the sample is oxidized upon contact with the enzyme. The resulting hydrogen peroxide (H2O2) is oxidized on the platinum anode and generates an electrical current flow between the platinum anode and silver electrode. The electrical current is directly proportional to the concentration of glucose in the sample.
The coated electrode, together with the measuring chamber, forms an easy to use, maintenance-free biosensor with a typical operation time of 3 months (up to 6 months maximum). Since this biosensor is designed as a disposable item, the enzyme membrane does not have to be changed. The replacement of the biosensor as a whole does not require any special knowledge and only takes a few minutes.
Methods
Measurements with SRM 965b were performed on 20 days, spread over 10 weeks from March 2020 to May 2020. Measurements were planned to be performed on 20 consecutive working days, but had to be interrupted for approximately 1.5 months due to governmental restrictions in the course of the Covid-19 pandemic. The study design and analysis were based on the Clinical and Laboratory Standards Institute’s guideline EP05-A3, section 3 (single-site precision evaluation study). 7 On each day, 2 runs with 2 measurements each were performed on 2 different SUPER GL compact devices (systems S1 and S2, provided by the manufacturer), and on one YSI 2300 STAT Plus device (system Y, provided by the study site). All devices were maintained and operated according to the respective manufacturer’s recommendations by three different trained medical technologists. Samples were prepared following the specific system’s instructions for use. For each run, a new aliquot of each level of SRM 965b standards were used. The samples were stored at −80°C and measured immediately after thawing.
Precision was estimated by repeatability (within-run precision) and by within-laboratory (within-device) precision; 95% confidence intervals were calculated for each estimate. Measurement data were checked for data integrity using Grubbs’ test. 8 To estimate how much the factors day and run contribute to the within-laboratory precision, two-way nested ANOVAs 9 were conducted for each level of SRM 965b and each device separately, as described in EP05-A3, section 3.6.3.
Trueness was estimated by calculation of mean relative difference between individual measurements and the SRMs’ certified target values (determined with isotope dilution-gas chromatography-mass spectrometry); 95% confidence intervals are provided for mean relative differences.
Results
Results for systems S1, S2, and Y are presented in Tables 2–4, respectively.
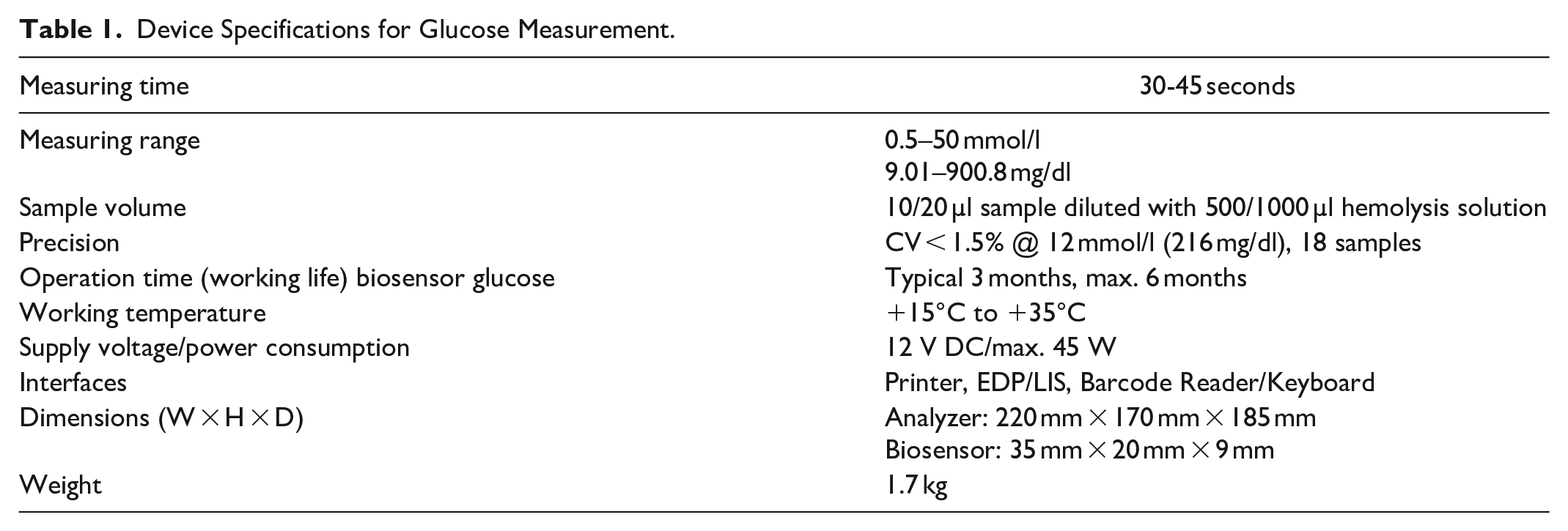
Measurement Results for Device S1 with 4 Levels of NIST SRM 965b. Certified Glucose Concentrations are given in Brackets.
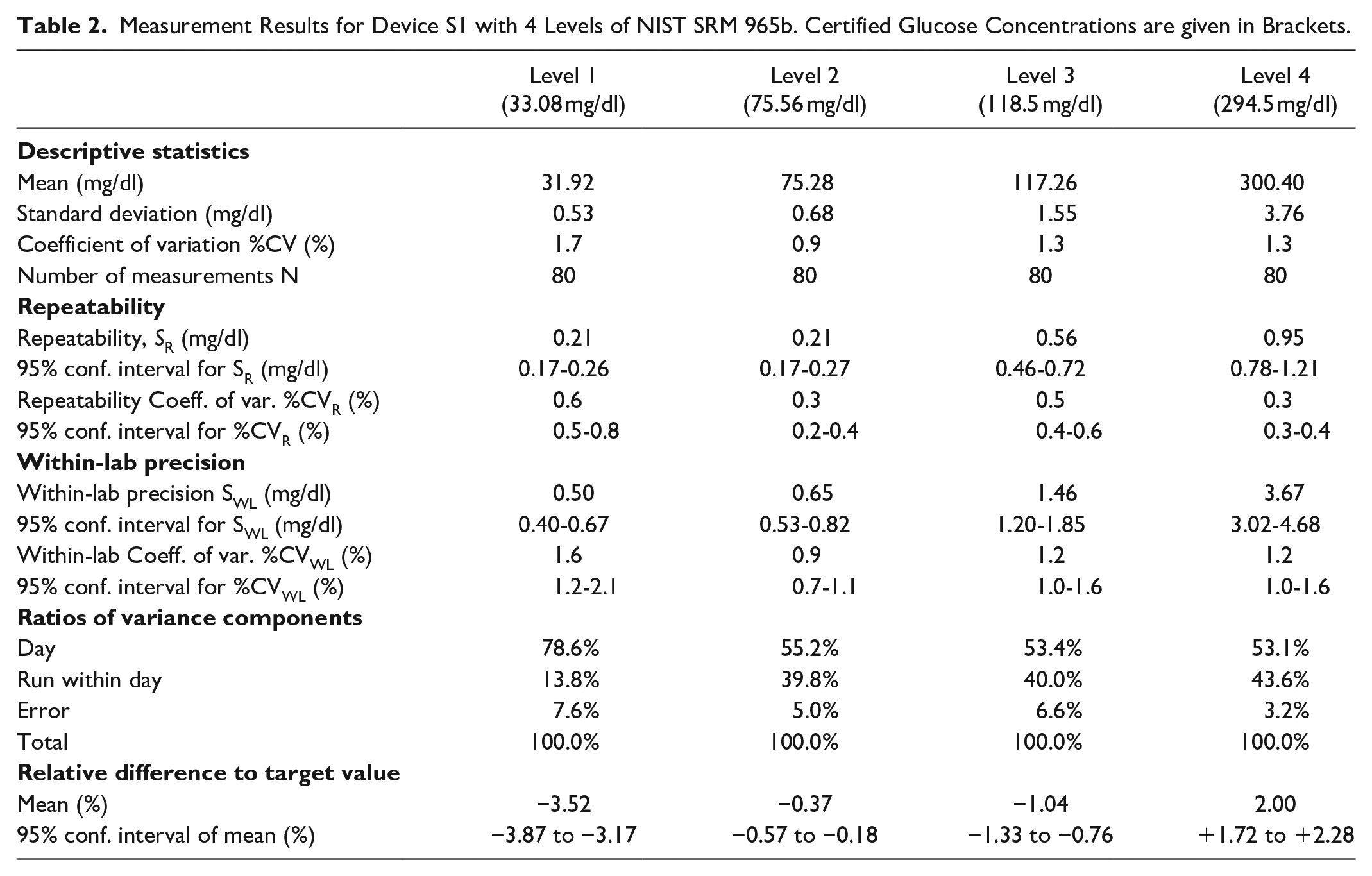
Measurement Results for Device S2 with Four Levels of NIST SRM 965b (Certified Glucose Concentrations are Given in Brackets)
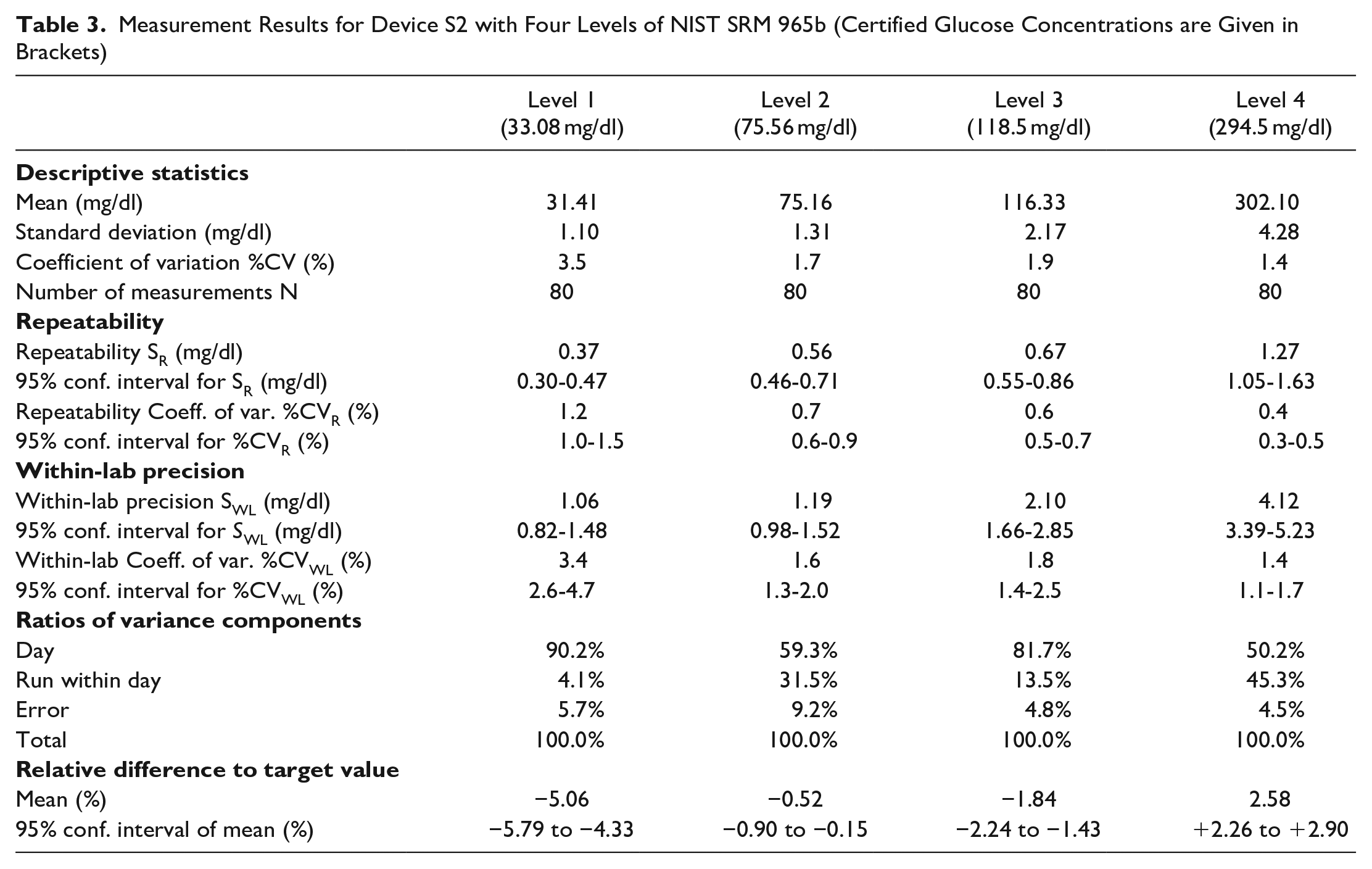
Measurement Results for device Y with Four Levels of NIST SRM 965b. Certified Glucose Concentrations are given in Brackets.
Maximum values for coefficients of variation (%CV) were 1.7% for S1, 3.5% for S2, and 2.4% for Y. %CV were largest, as expected, for the lowest concentration.
Repeatability coefficients of variation (%CVR) were ≤0.6% for S1, ≤1.2% for S2, and ≤0.5% for Y. The devices exhibited within-lab coefficients of variation (%CVWL) of ≤1.6% (S1), ≤3.4% (S2) and ≤2.4% (Y). For all devices and all levels of SRM 965b, measurement day was the largest contributor to variance (50.2–90.2%); less than 10% of variance could not be attributed to day or run.
Systems S1 and S2 underestimated low concentrations (bias −3.52% and −5.06%, respectively) and over-estimated high concentrations (bias: +2.00% and +2.58%, respectively). System Y results indicate a small negative bias at lowest concentration (−0.68%) and a slightly more negative bias at highest concentration (−1.97%). Of the three systems, Y exhibited the smallest variation in bias between the 4 levels of SRM.
Summary
The bench-top analyzer SUPER GL compact can determine glucose, lactate, and hemoglobin concentrations in hemolyzed whole blood, plasma, and serum samples. Glucose measurements are based on an enzymatic-amperometric reaction using glucose oxidase.
In this study, reference materials (NIST SRM 965b) were used to assess precision and trueness of SUPER GL compact (systems S1 and S2) and YSI 2300 STAT Plus (system Y). A potential limitation of this study was the comparably small scale. With additional devices and/or combinations of reagent lots, device-to-device and lot-to-lot variability could have been estimated for both types of analyzers.
Regarding overall coefficients of variation, systems S1 and S2 showed precision results similar to those of system Y, with largest values found for the lowest glucose concentration. The measurement day was the biggest contributor to variance for all glucose concentrations and all devices.
Bias (mean relative difference to the assigned target value), showed the smallest variation for system Y, which exhibited a negative bias that increased with higher concentrations. Bias for systems S1 and S2 was negative at low concentrations and positive at high concentrations.
Due to the small scale of this evaluation, a definite conclusion regarding adequacy of the SUPER GL compact as comparator device is not possible. Overall, SUPER GL compact showed satisfactory results similar to those of YSI 2300 STAT Plus when used for measurements of glucose concentrations in reference materials.
Footnotes
Acknowledgements
The authors would like to thank IfDT study staff for performing the study.
Abbreviations
%CVR, repeatability coefficient of variation; %CVWL, within-laboratory coefficient of variation; BG, blood glucose; CGM, continuous glucose monitoring; SRM, Standard Reference Material.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is general manager and medical director of the IfDT (Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies on the evaluation of BG meters, with CGM systems and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IfDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Dexcom, i-SENS, LifeScan, Menarini Diagnostics, Metronom Health, Novo Nordisk, PharmaSense, Roche, Sanofi, Sensile and Ypsomed. AB and SP are employees of IfDT. MH is managing director/CEO of Dr. Müller Gerätebau GmbH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study and medical writing was supported by Dr. Müller Gerätebau GmbH.
