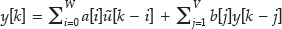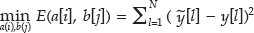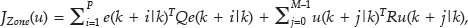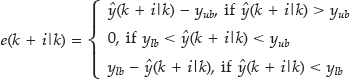Abstract
Background:
Because of the slow pharmacokinetics of subcutaneous (SC) insulin, avoiding postprandial hyperglycemia has been a major challenge for an artificial pancreas (AP) using SC insulin without a meal announcement.
Methods:
A semiautomated AP with Technosphere® Insulin (TI; MannKind Corporation, Valencia, CA) was designed to combine pulmonary and SC insulin. Manual inhalation of 10 U ultrafast-absorbing TI at mealtime delivers the first, or cephalic, phase of insulin, and an SC insulin pump controlled by zone model predictive controller delivers second-phase and basal insulin. This AP design was evaluated on 100 in silico subjects from the University of Virginia/Padova metabolic simulator using a protocol of two 50 g carbohydrate (CHO) meals and two 15 g CHO snacks.
Results:
Simulation analysis shows that the semiautomated AP with TI provides 32% and 16% more time in the controller target zone (80–140 mg/dl) during the 4 h postprandial period, with 39 and 20 mg/dl lower postprandial blood glucose peak on average than the pure feedback AP and the AP with manual feed-forward SC bolus, respectively. No severe hypoglycemia (<50 mg/dl) was observed in any cases.
Conclusions:
The semiautomated AP with TI provides maximum time in the clinically accepted region when compared with pure feedback AP and AP with manual feed-forward SC bolus. Furthermore, the semiautomated AP with TI provides a flexible operation (optional TI inhalation) with minimal user interaction, where the controller design can be tailored to specific user needs and abilities to interact with the device.
Introduction
Type 1 diabetes mellitus (T1DM) is a metabolic disorder in which pancreatic β cells are destroyed by autoimmune attack and cannot produce a sufficient amount of insulin to regulate blood glucose (BG) concentration. Without proper treatment, people with T1DM may develop neuropathy, nephropathy, retinopathy, and cardiovascular and cerebrovascular diseases due to chronic hyperglycemia (BG > 180 mg/dl). Also, ketoacidosis caused by severe hyperglycemia may result in a coma or even death. Thus people with T1DM must receive insulin therapy to regulate their BG.1–4
Regulating BG within a clinically accepted region (70–180 mg/dl) 5 with either multiple daily insulin injections or a continuous subcutaneous insulin infusion (CSII) pump is a burdensome task. This regimen requires multiple daily BG measurements and estimates of insulin dosages based on meal size, body weight, and other clinical factors. Furthermore, the insulin sensitivity of an individual changes over time. Thus the insulin dosage estimation procedure should be carefully adjusted continually. Otherwise, an overdelivery of insulin may cause hypoglycemia (BG < 70 mg/dl), which may result in seizure, coma, or death.1–4
The effort to develop an artificial pancreas (AP) started in the late 1970s with the Biostator®, which measured BG and delivered insulin and dextrose. 6 However, because of its invasiveness and large size, the intravenous (IV) closed-loop system was not tested outside of a hospital setting. Increasing academic and industrial effort has focused on minimally invasive subcutaneous (SC) closed-loop glucose control using a continuous glucose monitor (CGM) and CSII. To date, several studies have reported clinical and in silico results for closed-loop glucose control using a SC CGM and CSII.7–28
Despite the current focus of treatment using the SC route, because of the slow pharmacokinetics and long residence time (pharmacokinetic peak time of 50–60 min and residence time of 6–8 h) of the SC insulin, an AP design using the SC route has a number of limitations (e.g., prolonged postprandial hyperglycemia or late postprandial hypoglycemia) when this design encounters meal challenges.29–31 Recently, pulmonary insulin delivery has received significant attention as a complementary route because of the large surface area of the alveolar region (∼100 m2) and permeable membranes. Technosphere® Insulin (TI) is an inhalation powder
In this work, a novel semiautomated AP that uses TI to improve BG regulation at mealtime is presented. The semiautomated AP with TI combines two insulin delivery methods: ultra-rapid-acting TI and CSII controlled by zone model predictive control (zone- MPC). 32 To evaluate the potential of the semiautomated AP with TI, an autoregressive with exogenous input (ARX) pharmacokinetic model of TI was developed to simulate TI delivery. The performance and robustness of the semiautomated AP with TI was evaluated on 100 in silico subjects from the U.S. Food and Drug Administration (FDA)-accepted University of Virginia/Padova metabolic simulator. The semiautomated AP with TI was compared with both a pure feedback AP and an AP with manual feed-forward SC bolus treatments.
Methods
Autoregressive with Exogenous Input Model and Least-Square Method
The ARX model assumes that the current value of the time variable (output) is a function of its past values and exogenous inputs as shown in
where y[k], ũ[k], a[i], b[j], W, V, i, and j are the system output signal, input signal, coefficients of input terms, coefficients of output terms, number of input signal terms, number of output signal terms, ith term of the input signal, and jth term of the output signal, respectively.
33
The complexity of the ARX model presented in this work (W = 1 and V = 5) was determined based on several factors: the number of data points, the quality of data points, and the desired accuracy of the model. Then, using the fminunc() function in MATLAB (MathWorks, Natick, MA), the coefficients of the equation were determined by the least squares method:
where E(a[i], b[j]), y˜[l], y[l], N, and l are the error of the model, the measured data points from an experiment, the predicted outputs by the ARX model, the number of data points, and the lth term of data points, respectively. 33
Semiautomated Artificial Pancreas with Technosphere Insulin
Normal physiological insulin secretion at mealtime consists of two phases: the rapid rising cephalic first phase of insulin secretion and the prolonged second phase of insulin secretion.
34
Thus, to reproduce the physiological insulin secretion pattern, an ideal AP needs to capture both phases. As depicted in
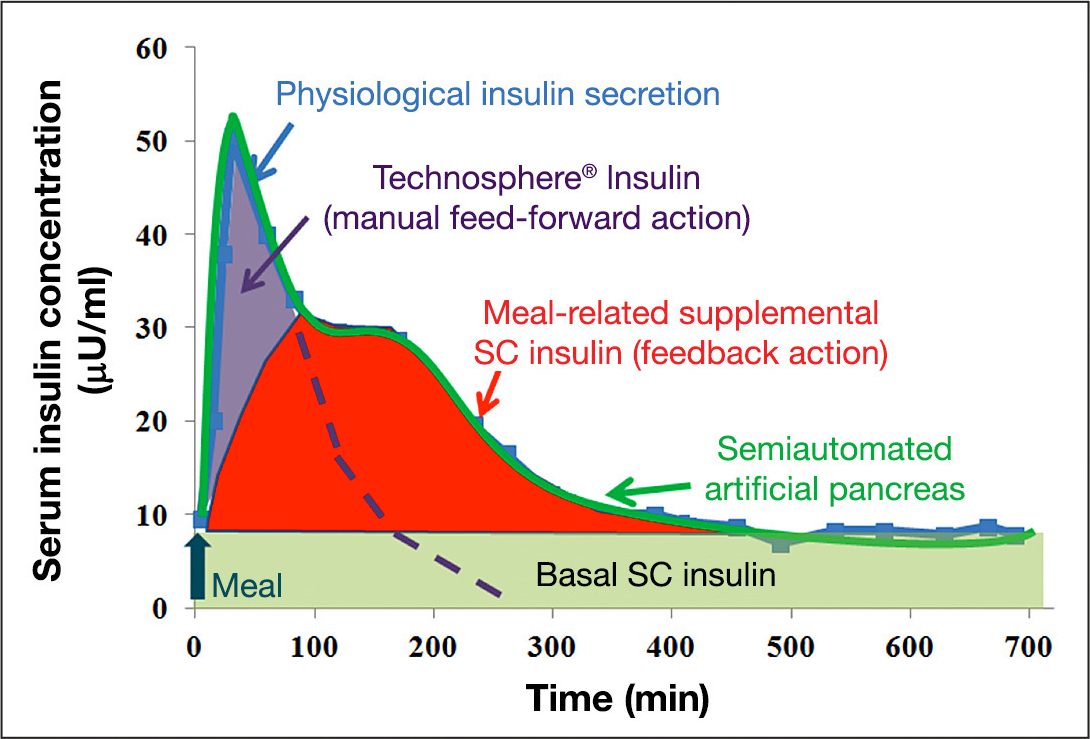
Schematic description of the semiautomated AP with TI. Rapid-acting TI mimics the cephalic and first-phase insulin secretion at mealtime, and zone-MPC-controlled CSII delivers meal-related supplemental insulin and basal insulin. The physiological insulin-secretion profile was modified from a figure in Polonsky and coauthors. 34
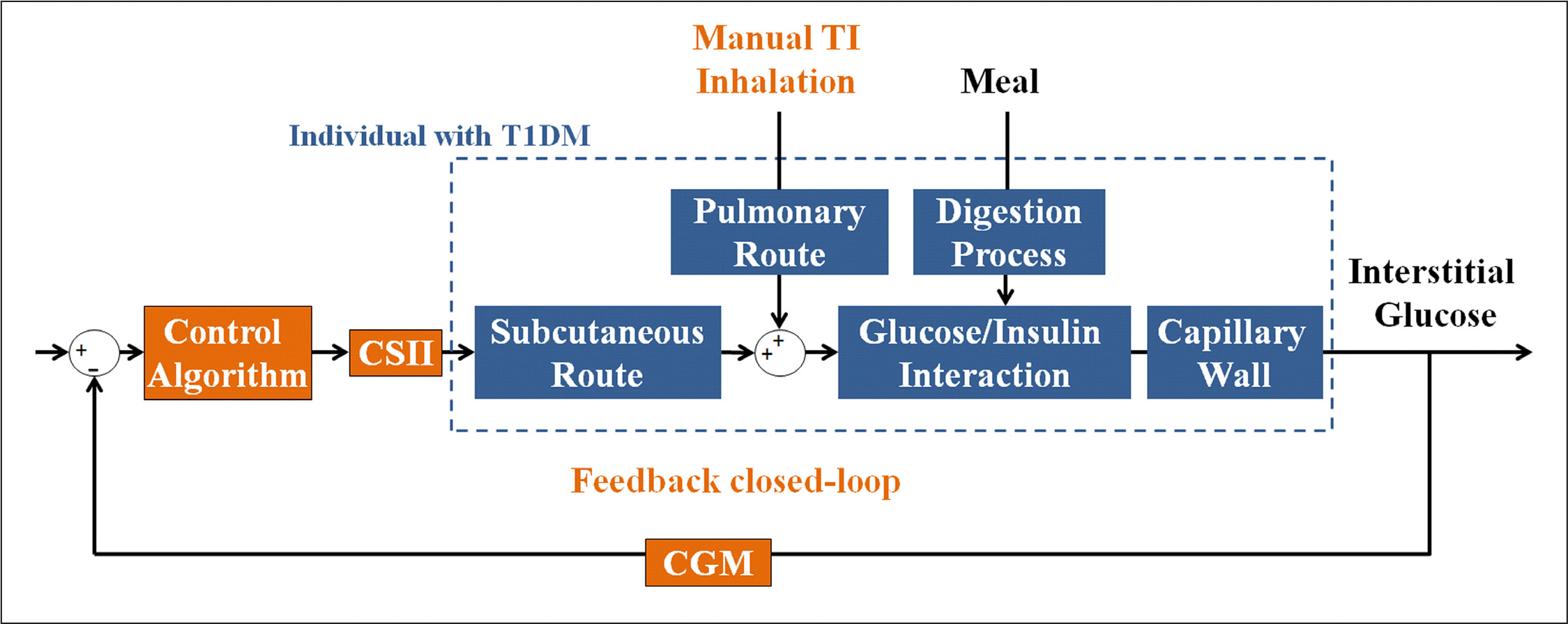
Block diagram of the semiautomated AP with TI. Inhalation of TI (manual feed-forward action) blunts the meal response, and feedback closed-loop control (CGM, control algorithm, and CSII) delivers supplemental insulin and basal insulin.
Zone Model Predictive Control
Zone-MPC is a model predictive algorithm that was designed to control predicted output within a suitable range, or zone, of values, rather than to a set point of a single value. As discussed in the work of Grosman and coauthors, 32 high-frequency measurement errors in glucose measurements and model–subject mismatches in the current AP make the use of a specific set point for BG regulation irrelevant or impractical. 32 Also, normoglycemia itself is defined as a zone. Thus defining the control objective as a zone is a natural strategy for the algorithm of an AP. 32
The essence of the zone-MPC algorithm is well represented by its cost function. As given in
where ŷ(k + i|k), u(k + i|k), yub, ylb, P, M, and e are the predicted output, the predicted input, the upper boundary (140 mg/dl), the lower boundary (80 mg/dl), the prediction horizon, the control horizon, and the error, respectively.
The error is defined in such a way that the predicted outputs are penalized only when the values are outside of the zone. 32 The prediction horizon, P, was set to be nine steps (45 min), because the quality of prediction decreased beyond 45 min. The control horizon, M, and the Q-to-R ratio were tuning parameters of the zone-MPC. Thus the specific values (M of five steps, 25 min, and Q-to-R ratio of 1 to 15) of the parameters were chosen because the values resulted in the best BG regulation, which was illustrated by a fast meal disturbance rejection with minimal hypoglycemia risk. The sampling rate of the algorithm is chosen to be 5 min due to the sampling rate of the CGM device.
The model of glucose/SC insulin interaction that is used in this study is presented by Van Heusden and coauthors. 22 The model utilizes a priori knowledge of a subject (i.e., individual correction factor) to account for the intersubject variability of insulin sensitivity and incorporates an adjustable safety factor to improve system robustness due to human errors in the estimation of the correction factor. 22 The health monitoring system (HMS) developed by Harvey and coauthors35,36 and Dassau and coauthors 37 is implemented in parallel to the control loop to provide a safety layer and further reduce hypoglycemia risk.35–37 It was designed to receive and process CGM data and make a prediction of glucose trends. The HMS automatically sends SMS and MMS (short and multimedia message services) to a predefined contact list to alert the receiver of imminent safety hazards.35–37
List of Experiments
A 24 h protocol including two meals and two snacks
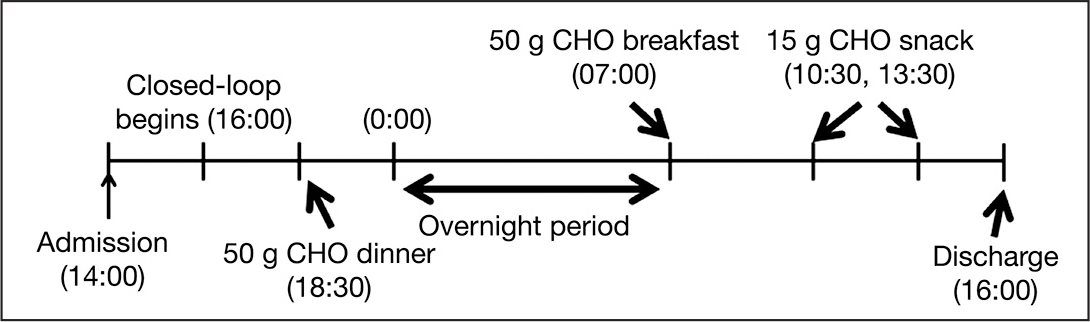
A 24 h protocol consisting of two meals (50 g CHO each), an overnight period, and two snacks (15 g CHO each).
Summary of Artificial Pancreas Designs (Degrees of Automations)

Manual action by the user is required.
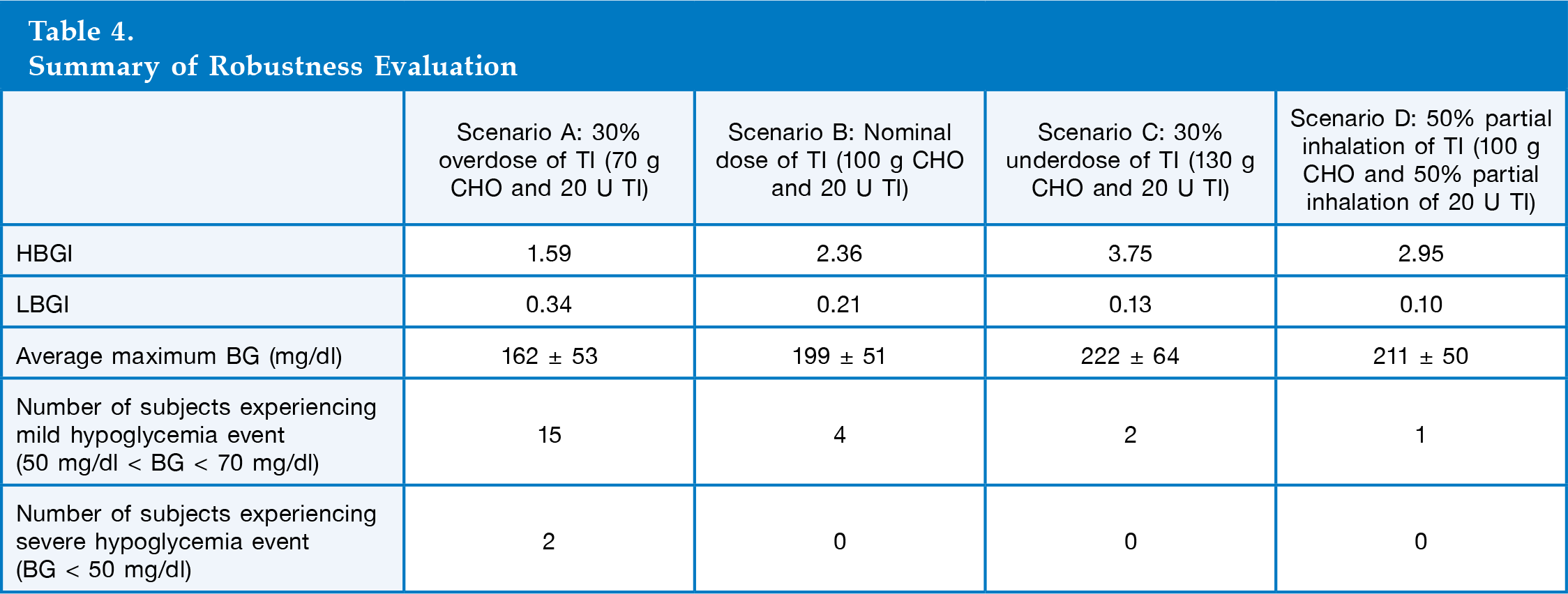
Robustness Evaluation
We evaluated the robustness of the AP design by using a single-meal protocol with four scenarios: (A) 30% overdose of TI (70 g CHO meal + 20 U TI), (B) nominal dose of TI (100 g CHO meal + 20 U TI), (C) 30% underdose of TI (130 g CHO meal + 20 U TI), and (D) 50% partial inhalation of TI (100 g CHO meal + 20 U TI, but only 50%, or 10 U, is inhaled). In the protocol, closed-loop starts 2 h after a subject's admission, and a single meal (70, 100, or 130 g CHO) is served 9 h after the closed-loop starts. At mealtime, a subject inhales 20 U TI, and the control algorithm is not given this information. The simulation was run until 6 h after the meal to observe the full effect of the TI inhalation.
Results
We developed an ARX model that transforms a square pulse inhalation signal into a serum insulin concentration profile based on the pharmacokinetics of TI. We incorporated the developed model into the FDA-accepted University of Virginia/Padova metabolic simulator via the IV port to reproduce the insulin action of TI. The glucose infusion rate (GIR; pharmacodynamics) was measured to evaluate the TI module. The following two subsections describe the evaluation procedures in detail.
Pharmacokinetic Model of Technosphere Insulin
The experimental data of the serum insulin concentration profile after a TI inhalation from the work of Rave and coauthors
31
were used to develop an ARX model that reproduces the pharmacokinetics of TI.
31
The pharmacokinetics of TI model, given in 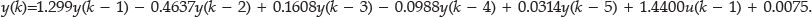
As a validation of the pharmacokinetics of TI model, the predicted serum insulin concentrations of the model after 50 or 100 U TI inhalations were compared with the corresponding experimental data.
31
As shown in
Identified pharmacokinetics of TI model and its evaluation on experimental data. The red dashed, green dashed–dot, and blue solid curves represent the model predictions for 100, 50, and 25 U, respectively, and the red × marks, green × marks, and blue × marks represent the corresponding experimental data of 100, 50, and 25 U from Rave and coauthors, respectively. 31
Pharmacodynamics of the Technosphere Insulin Module
The developed pharmacokinetics of the TI model were incorporated into the metabolic simulator via the IV port to reproduce the pharmacodynamics of a TI inhalation. To validate the pharmacodynamics of the TI module (pharmacokinetics of TI model + IV port), the average GIR of 10 in silico subjects after an inhalation of 30 U TI was obtained by an in silico clamp test and compared with experimental data obtained from MannKind Corporation (Valencia, CA; unpublished data). The procedure of the clamp test is described in the work of Heinemann and Ampudia-Blasco
39
and Bequette.
40
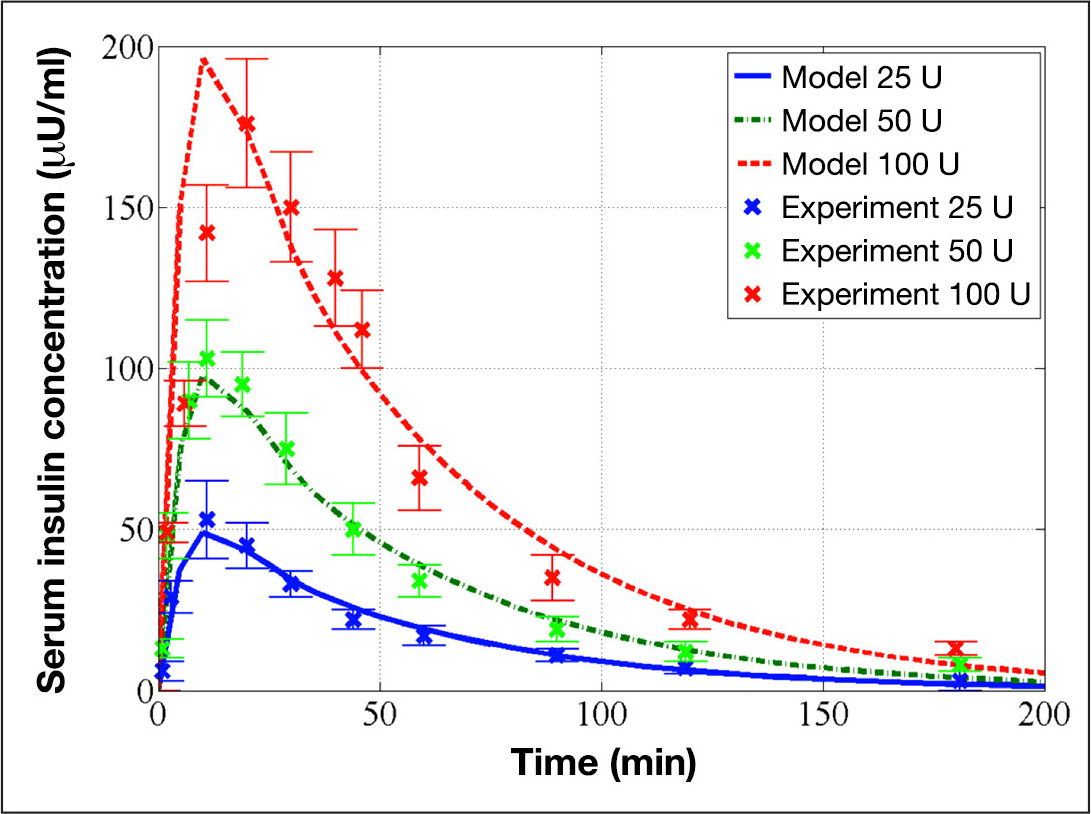
As shown in
Comparison of average GIR of the in silico subjects and average GIR of the experimental participants, which validates the pharmacodynamics of the TI module. The red circles represent the average GIR of the experimental participants, and the red bars represent one standard deviation away from the average (unpublished data). The blue triangles represent the average GIR of in silico subjects, and the blue-shaded region represents one standard deviation away from the average model.
Evaluation of Semiautomated Artificial Pancreas with Technosphere Insulin
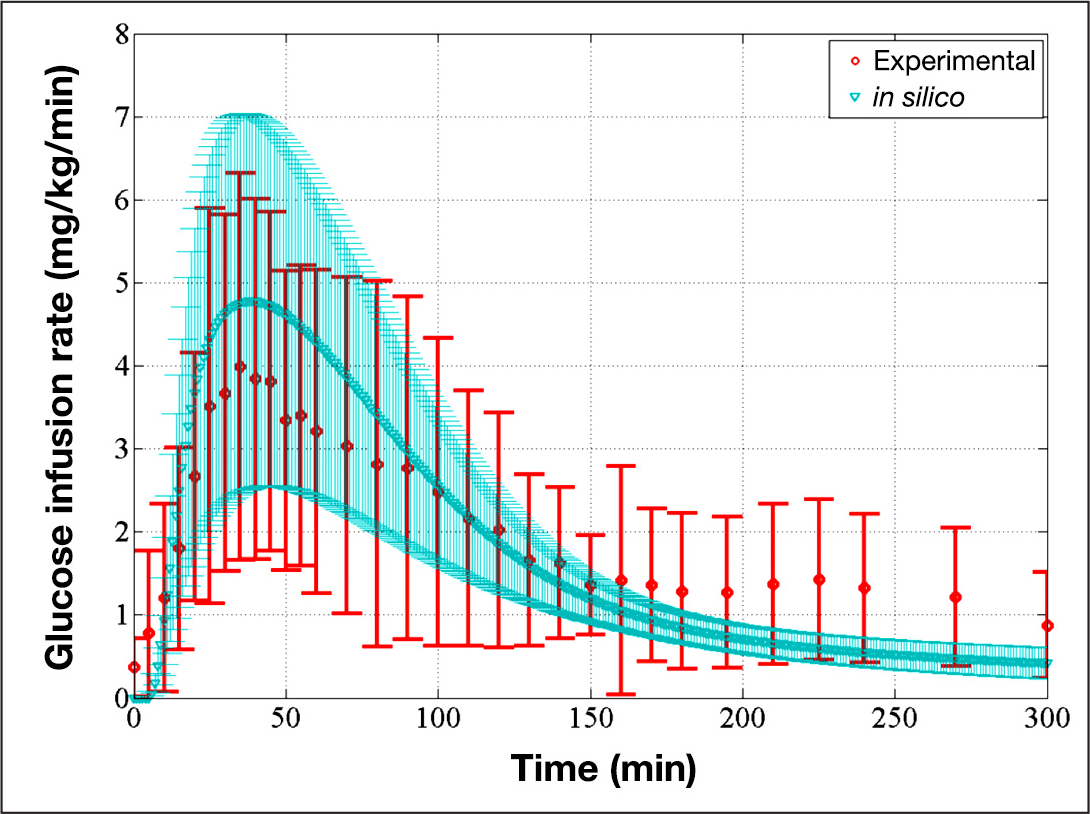
The semiautomated AP with TI that combines pulmonary and SC insulin was evaluated using the metabolic simulator and compared with pure feedback AP and AP with manual feed-forward SC bolus. The average and 1 SD envelope of 100 subjects over time is illustrated in Summary of BG trace and insulin delivery of 100 in silico subjects in all three cases. Left panels: Average and SD envelope of BG trace in Percentages of Time in the Clinically Accepted Blood Glucose Region (70–180 mg/dl) and Time in the Controller Target Zone (80–140 mg/dl)
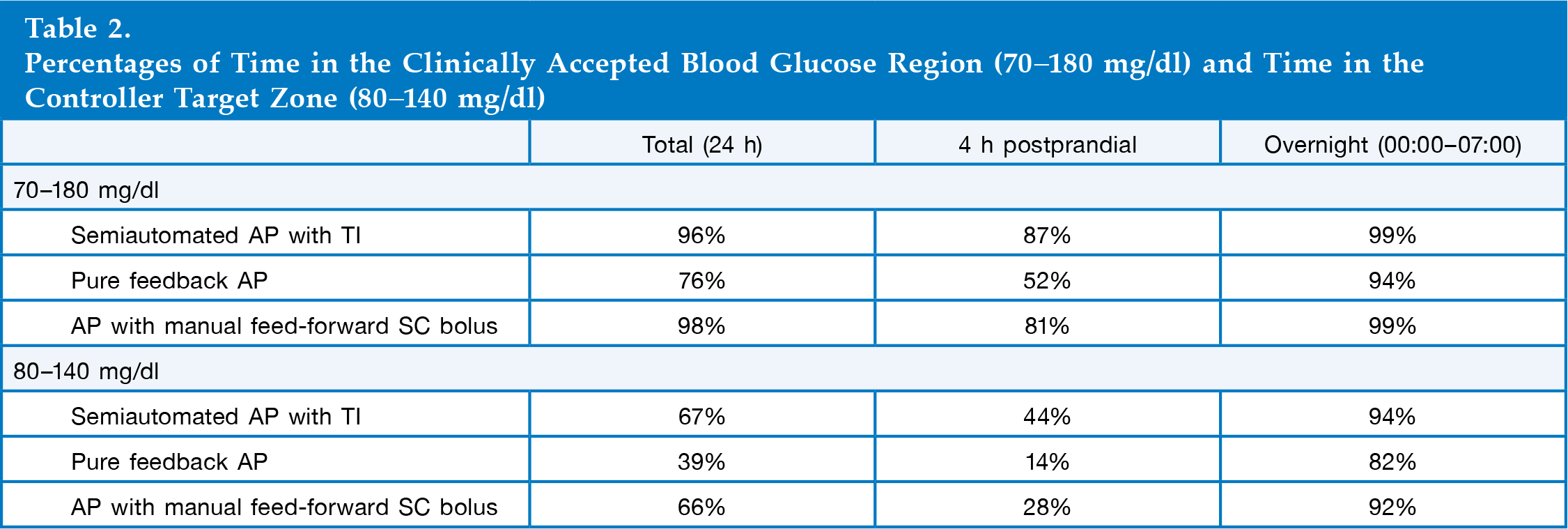

where tindividual and n are the individual's percentage of time in a desired zone during a period and the number of subjects, 100.
Comparing average BG profiles from
Summary Results of Experiments
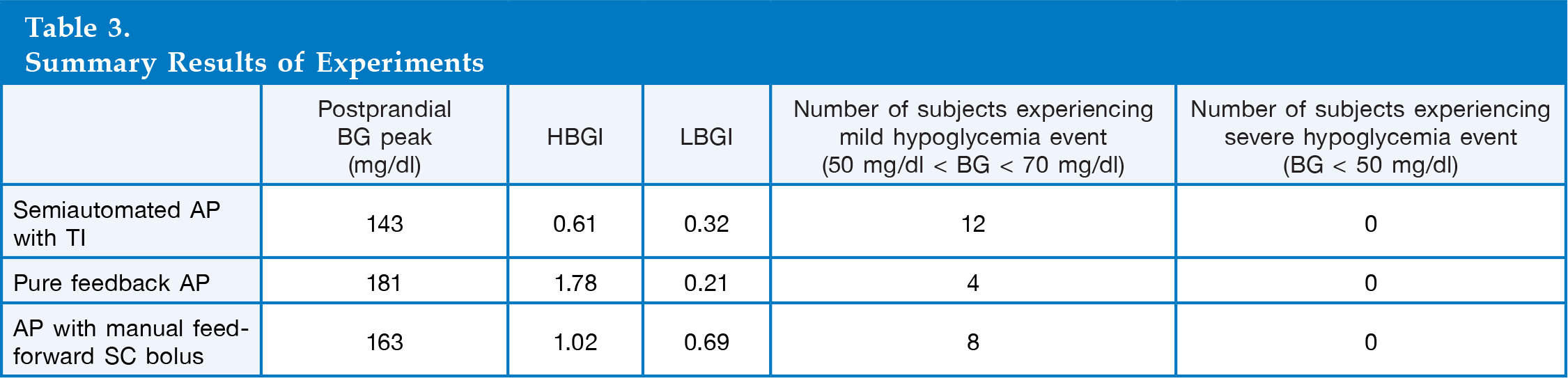
To demonstrate the differences in the closed-loop control actions of three AP designs,
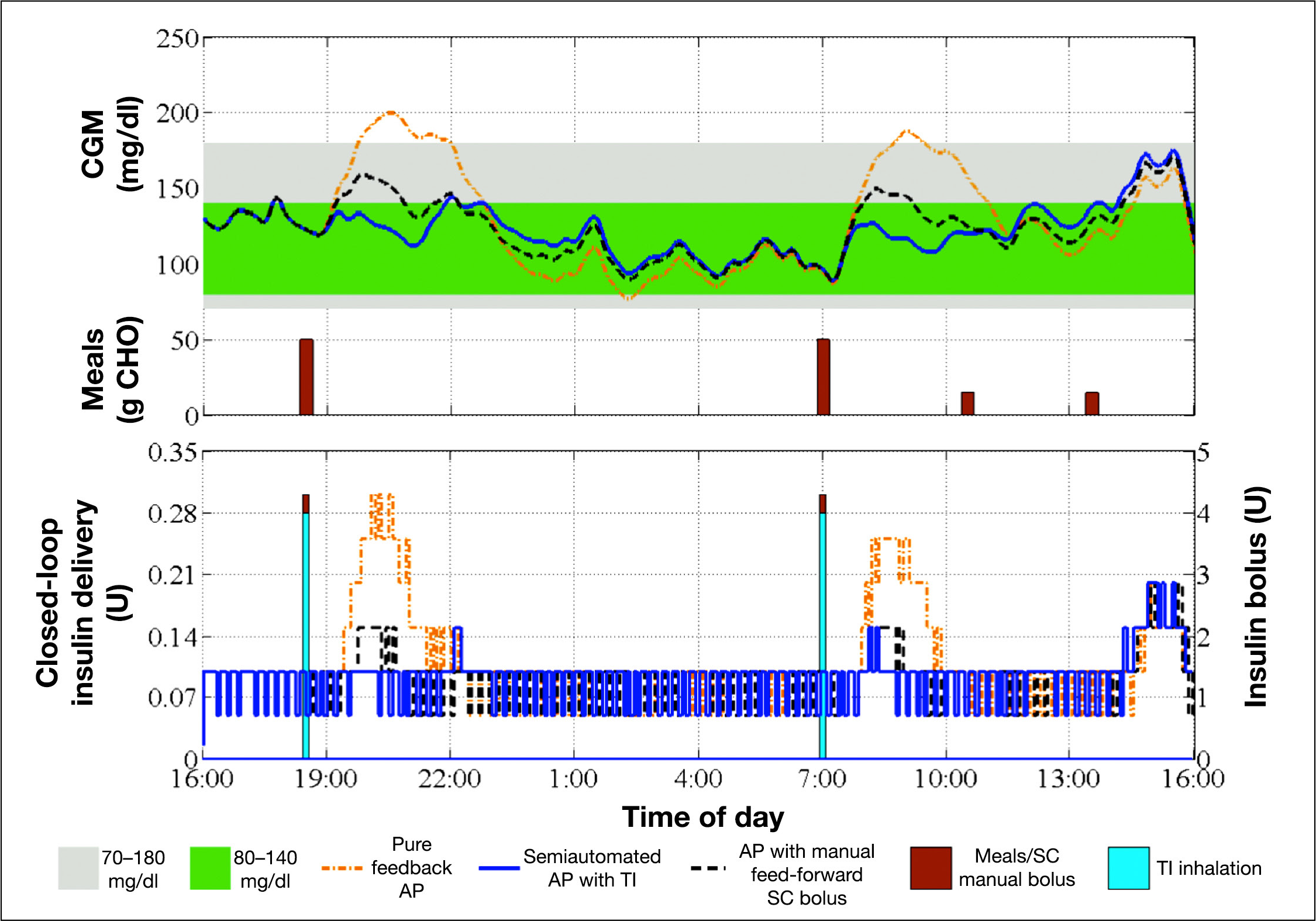
Continuous glucose monitor trace and insulin delivery of a representative subject in all three cases. Top panel: CGM trace of an in silico subject for the pure feedback AP (orange dash–dot curve), the semiautomated AP with TI (blue solid curve), or the AP with manual feed-forward SC bolus (black dashed curve). Sizes of the meals are also indicated (brown bars). Bottom panel: Insulin delivered via CSII pump in the case of the pure feedback AP (orange dashed–dot curve), the semiautomated AP with TI (blue solid curve), or the AP with manual feed-forward SC bolus (black dashed curve). Also, the average size of SC manual bolus (brown bars) and the TI inhalation dosage (blue bars) are represented.
Robustness Analysis
Summary of Robustness Evaluation
Discussion
Establishing normoglycemia without interrupting the regular daily routine of an individual with T1DM is a challenging task.1–4 A pure feedback AP based on SC insulin requires minimal user interaction, but postprandial hyperglycemia is nearly unavoidable using the pure feedback AP without a meal announcement with ingestion of a large meal because of the slow action of SC insulin. Alternatively, an AP with a perfect estimation of the SC bolus can nearly achieve normoglycemia, but it requires meticulous calculation. Thus an AP design that can blunt a meal response with minimal user interaction may achieve superior BG regulation. The semiautomated AP with TI was designed so that it can anticipate a meal response with TI inhalation and manage BG within the normoglycemia region by zone-MPC-controlled CSII. Based on in silico evaluation results following the 24 h protocol, the semiautomated AP with TI showed better performance (i.e., lowest postprandial BG peaks and longest time in the clinically accepted BG region) than either the AP with perfect SC bolus or the pure feedback AP. Also, the robustness analysis showed that TI inhalation does not impose significant risk of hypoglycemia to users.
Conclusions
The semiautomated AP with TI has flexibility in its operation due to an optional TI inhalation and provides proper BG regulation for different users in different situations. This novel design of the AP provides superior performance over other AP designs, such as those with fully automated or manual SC bolus for meals. Therefore, a semiautomated AP with TI will reduce the likelihood of long- or short-term complications related to T1DM and improve the quality of life of people with T1DM.
Footnotes
Abbreviations:
This work was supported by the Juvenile Diabetes Research Foundation Grant #17-2010-765.
Howard Zisser and Lois Jovanovič have received research support from and are consultants to MannKind Corporation
Acknowledgments:
MannKind Corporation provided Technosphere pharmacokinetics and pharmacodynamics data.