Abstract
Importance
Sudden sensorineural hearing loss (SSNHL) has heterogeneous etiologies and prognoses. Early-imaging evaluation may help identify patients at higher risk of poor hearing recovery.
Objective
To investigate the relationship between labyrinthine gadolinium-enhanced MRI (Gd-MRI) findings and clinical characteristics in patients with SSNHL and to assess the clinical value of MRI-based subtyping for early evaluation and stratified treatment.
Design
Retrospective observational study.
Setting
Single tertiary referral center.
Participants
Ninety-six patients with unilateral SSNHL treated between January 2014 and May 2020 were included.
Exposure or Intervention
All patients underwent gadolinium-enhanced inner ear MRI. According to imaging features, they were categorized into an MRI-negative group, an endolymphatic hydrops (EH) group, and a labyrinthine signal abnormalities (LSA) group.
Main Outcome Measures
Clinical symptoms, audiological data, inner ear subunit involvement, and short-term hearing gain were compared among groups. Independent predictors of hearing improvement were analyzed using multivariate linear regression.
Results
Among 96 patients, 55 were MRI-negative, 20 were EH, and 21 were LSA. The LSA group had the highest rates of vertigo and tinnitus (100% and 95.2%, respectively; P = .001, P = .038) and more extensive inner ear involvement (≥2 subunits, 71.4%; P = .003). Pretreatment hearing thresholds were comparable between groups, but the LSA group had a significantly-poorer pure-tone average after treatment. Multivariate regression identified age and MRI classification as independent predictors of low-frequency hearing gain (P < .05).
Conclusion
Gadolinium-enhanced inner ear MRI effectively stratifies SSNHL by identifying distinct imaging phenotypes correlated with clinical severity and recovery potential. Labyrinthine signal abnormalities, indicating more extensive inner ear involvement, are associated with poorer hearing recovery.
Relevance
MRI-based subtyping can facilitate early evaluation and individualized management of SSNHL, supporting its integration into routine diagnostic assessment. These preliminary findings require confirmation through larger prospective studies with long-term follow-up.

Keywords
Key Messages
Gd-enhanced inner ear MRI stratifies sudden sensorineural hearing loss into MRI-negative, endolymphatic hydrops, and labyrinthine signal abnormalities (LSA).
LSA affects more inner ear subunits, suggesting greater structural damage.
Age and MRI classification independently predict low-frequency hearing gain, serving as valuable markers for early prognosis and personalized management.
Introduction
Sudden sensorineural hearing loss refers to hearing loss of ≥30 dB HL in at least 3 adjacent frequencies occurring within a 72 hour period. Among most patients, sudden sensorineural hearing loss is idiopathic (ISSNHL) 1 and may be multifactorial. The most common causes include viral infections, 2 vascular factors, 3 autoimmunity,4,5 and hydrops of the membranous labyrinth6,7. Hearing outcomes are highly dependent on the early identification of the underlying cause, which helps assess the severity of the disease and provides a key basis for developing individualized intervention plans.1,8,9
Conventional MR imaging in patients with SSNHL is primarily used to rule out brain lesions or to identify retrocochlear lesions such as vestibular schwannomas. 10 In recent years, newer MR sequences, such as three-dimensional fluid-attenuated inversion recovery (3D-FLAIR) combined with gadolinium contrast enhancement, have been developed to assess abnormal signals in the inner ear labyrinth, including methemoglobin deposition or protein accumulation11 -17 and to identify endolymphatic hydrops (EH) by volumetric measurement. 18 Although endolymphatic hydrops is the primary pathological feature of Meniere’s disease, some patients may present with sudden deafness as the initial manifestation, necessitating follow-up for a definitive diagnosis. 19
During the early evaluation of patients with SSNHL, gadolinium-enhanced inner ear MRI not only helps exclude organic causes but also reveals different inner ear pathological states. These imaging abnormalities may be associated with clinical symptoms and the potential for hearing recovery, but their prognostic value remains controversial.13,16 The present study aimed to summarize and categorize 3D-FLAIR imaging sequences in patients with SSNHL to better understand the pathological mechanisms of sudden hearing loss and provide a basis for clinical stratification and treatment.
Materials and Methods
Patients
A total of 96 patients with unilateral sudden deafness were recruited during their initial visit to the Department of Otolaryngology Head and Neck Surgery between January 2014 to May 2020. Patients were screened according to strict inclusion and exclusion criteria, and clinical data on pretreatment and posttreatment audiological characteristics and gadolinium-based magnetic resonance imaging examinations were subsequently collected. Inclusion and exclusion criteria included: ① meeting the 2019 American Clinical Practice Guideline: Sudden Hearing Loss criteria for sudden deafness 1 : ≥30 dB sensorineural hearing loss in at least 3 consecutive frequencies occurring within 72 hours; ② having complete clinical records and undergone pure-tone audiometry and gadolinium-based MRI examinations; ③ age ≥18 years, unilateral deafness, first onset; ④ excluding those with clear deafness-related factors, such as trauma, genetics, and other central and peripheral pathogenic factors; ⑤ excluding those with middle ear and inner ear space-occupying lesions as determined by specialist examinations and imaging examinations; ⑥ not receiving standard medication such as hormones or medications to improve inner ear microcirculation; and ⑦ excluding pregnant women and those with contraindications to the use of glucocorticoids.
Audiological Indicators
Classification of hearing curves in sudden deafness. The extent of hearing loss was calculated using the pure-tone average (PTA) of hearing thresholds at 500 Hz, 1 kHz, 2 kHz, and 4 kHz. Patients were subsequently classified into 4 subtypes20 -22: ① Low-frequency decline subtype: Hearing loss at frequencies of 1000 Hz (inclusive) and below, with at least a 20 dB HL hearing loss at 250 and 500 Hz. ② High-frequency decline subtype: Hearing loss at frequencies of 2000 Hz (inclusive) and above, with at least a 20 dB HL hearing loss at 4000 and 8000 Hz. ③ Flat decline subtype: Hearing loss at all frequencies, with an average hearing threshold of ≤80 dB HL. ④ Total deafness subtype: Hearing loss at all frequencies, with an average hearing threshold of ≥81 dB HL at 250, 500, 1000, 2000, 3000, 4000, and 8000 Hz.
Early-efficacy assessment. All patients received standard treatment according to the Clinical Practice Guideline, 23 including uniform intravenous steroid therapy and improved circulatory drug therapy. No intratympanic steroid injections were administered. The treatment duration lasted until hearing recovery or until pure-tone audiometry retesting 10 days after the initiation of treatment. Responses to therapy were classified according to Siegel’s criteria 24 : Complete recovery: final hearing threshold ≤25 dB HL; partial improvement: hearing gain >15 dB HL with a final threshold between 25 and 45 dB HL; slight improvement: hearing gain >15 dB HL with a final threshold >45 dB HL; no response: hearing gain <15 dB HL and final threshold >75 dB HL. Both “Complete recovery” and “Partial improvement” were considered “effective.”
Imaging Acquisition
All 96 patients provided written informed consent prior to the procedure. Before the intervention, patients were fully informed of the options for unilateral or bilateral intratympanic injection and were allowed to choose voluntarily. All patients ultimately included in this study elected to undergo bilateral intratympanic administration. Gadopentetate dimeglumine (Gd-DTPA) was diluted eightfold with normal saline prior to use. The contrast agent was injected into the middle ear cavity through the anteroinferior quadrant of the tympanic membrane, with an administered volume of approximately 0.4 to 0.5 mL per ear. Bilateral injections were performed simultaneously during the same session. After injection, patients remained seated with the head tilted backward for 1 hour and were instructed to minimize speaking, swallowing, and drinking to reduce variability in contrast distribution. After a 24 hour delay, 3D-FLAIR MRI was acquired on a 3.0 T Verio scanner (Siemens, Erlangen, Germany) equipped with a 16-channel head coil. Imaging parameters included repetition time (TR) of 6000 ms, echo time (TE) of 132 ms, and isotropic voxel resolution of 0.5 × 0.5 × 0.5 mm, with a total acquisition time of 4 minutes 16 seconds. In addition, 3D sampling perfection with application-optimized contrasts using different flip angle evolution (3D-SPACE) inversion recovery was applied for fluid signal suppression (TR = 6000 ms; TE = 388 ms; inversion time [TI] = 2100 ms; voxel size = 0.7 × 0.7 × 0.7 mm; scan time = 5 minutes 32 seconds). 25
MRI-Based Classification
Based on MRI findings, patients were categorized into 3 groups: MRI-negative, EH, and labyrinthine signal abnormality (LSA). EH was defined as visible enlargement of the endolymphatic space relative to the perilymphatic space on delayed contrast-enhanced 3D-FLAIR images. 25 LSA was defined as abnormally-increased signal intensity involving the cochlea, vestibule, and/or semicircular canals on MRI, including either hyperintensity on pre-contrast images or increased signal intensity on post-contrast images compared with the contralateral side.13,26 The MRI-negative group was defined as the absence of abnormal MRI findings, with no evidence of EH or LSA. In this cohort, no patients demonstrated concomitant EH and LSA; therefore, the 3 groups were mutually exclusive. All MR images were independently reviewed by one radiologist and one senior otolaryngologist, each with over 5 years of experience in image interpretation. Both reviewers were blinded to the clinical data and the affected side. 25 Any discrepancies were resolved by consensus discussion. Figure 1 shows example images of inner ear gadolinium imaging results for the 3 groups.

Representative MRI findings of SSNHL subtypes. (A) MRI-negative group: 3D-FLAIR sequence shows no abnormal signal, with symmetric appearance between the affected and contralateral sides. (B) Endolymphatic hydrops group: hydrops in left cochlea (white solid-line arrow) and vestibule (white dotted arrow); the right side is normal. (C) LSA group (pre-contrast): slight signal increase in left cochlea and vestibule (white arrowhead) compared with the right side. (D) LSA group (post-contrast): marked enhancement in left cochlea and vestibule (white arrowhead), with no enhancement on the right side. LSA, labyrinthine signal abnormality; SSNHL, sudden sensorineural hearing loss; 3D-FLAIR, three-dimensional fluid-attenuated inversion recovery.
Statistical Analysis
Statistical analyses were performed using R version 4.2.1. Normally-distributed data are expressed as mean ± standard deviation (SD) and were compared using one-way ANOVA; non-normally-distributed data are presented as median with interquartile range (IQR) and were compared using the Kruskal-Wallis test. Categorical variables were compared using the chi-squared test or Fisher’s exact test. When a significant overall difference was observed, post hoc pairwise comparisons were performed with the Bonferroni correction. After univariate analysis, significant variables were included in the multivariate linear regression model. All tests were 2-sided, and differences were considered statistically significant when P < .05.
Results
Demographics
A total of 96 patients were enrolled and classified into 3 groups based on MRI findings: MRI-negative (n = 55), EH (n = 20), and LSA (n = 21). The mean ages of the MRI-negative, EH, and LSA groups were 43.2 ± 12.7, 47.3 ± 14.2, and 48.1 ± 13.9 years, respectively (P = .262). The incidence of vertigo was 52.7%, 50.0%, and 100.0% (P = .001), with a higher incidence in the LSA group than in the MRI-negative group and the EH group (Bonferroni correction, adj. P < .05). Aural fullness was reported in 54.5%, 40.0%, and 28.6% of the patients (P = .107), and tinnitus was reported in 94.5%, 75.0%, and 95.2% of the patients (P = .038). The incidence of underlying diseases (hypertension, hyperlipidemia, or diabetes) was 21.8%, 25.0%, and 19.0%, respectively (P = .891). Baseline data are shown in Table 1.
Demographic and Clinical Characteristics of Patients by MRI Findings.
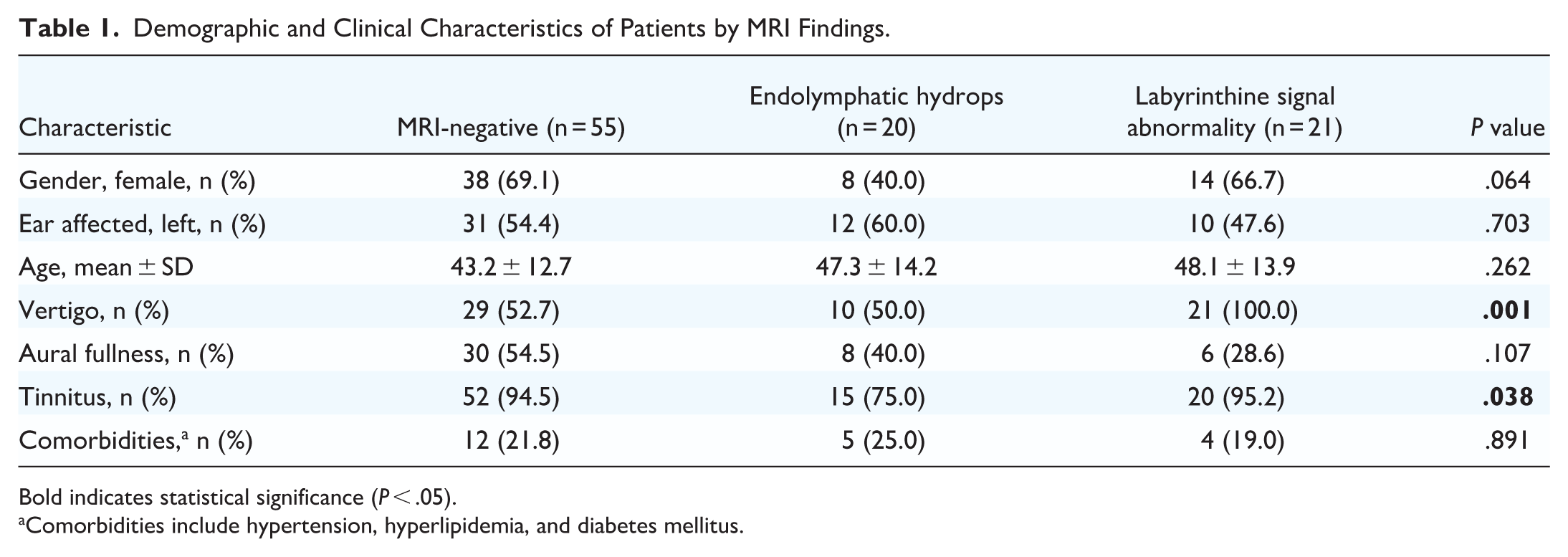
Bold indicates statistical significance (P < .05).
Comorbidities include hypertension, hyperlipidemia, and diabetes mellitus.
MRI
Table 2 shows the distribution of inner ear subunit involvement in the EH and LSA groups. The cochlear involvement rates were 30% and 9.5% in the EH and LSA groups, respectively (P > .05); the vestibular involvement rates were 45% and 19% (P > .05). Involvement of two or more subunits was observed in 25% of the EH group and 71.4% of the LSA group (χ² = 8.838, P = .003), indicating that the LSA group exhibited more extensive inner ear involvement.
Inner Ear Subunit Involvement in Patients With Endolymphatic Hydrops and Labyrinthine Signal Abnormality.

Bold indicates statistical significance (P < .05).
P-value could not be calculated due to zero events in both groups.
Clinical Symptoms
Audiological outcomes were compared among the 3 groups (Table 3, Figure 2A). Pretreatment PTA across the 3 groups was 71.3, 68.1, and 90.0 dB HL, respectively (Kruskal-Wallis test, P = .076). After treatment, these values decreased to 47.5, 68.1, and 81.3 dB HL, respectively (P = .013). Posttreatment low-frequency PTA was 35.0, 60.0, and 77.3 dB HL, respectively (P = .006), mid-frequency PTA was 45.0, 66.7, and 77.3 dB HL, respectively (P = .025), and high-frequency PTA was 62.5, 77.5, and 85.0 dB HL, respectively (P = .010). Multiple comparisons indicated that the LSA group had higher posttreatment PTA and subgroup frequency PTA than the MRI-negative group (Figure 2A; adj. P = .013, .008, .024, and .013, respectively). Analysis of low-frequency ΔPTA revealed significant differences among the 3 groups (Kruskal-Wallis test, P = .028), with post hoc tests confirming significantly poorer improvement in the LSA group than in the MRI-negative group (adj. P < .05). The distribution of the 4 hearing curve types (low-frequency decline, high-frequency decline, flat, and total deafness) was not statistically different among the groups (P = .119). Paired analysis (Wilcoxon signed-rank test, Figure 2B-D) indicated that the MRI-negative group observed significant hearing improvement after treatment (P < .001), while the EH group and the LSA group showed significantly less improvement. Overall, the MRI-negative group showed the greatest hearing improvement after treatment, particularly in PTA and low-frequency, while the LSA group showed the worst recovery.
Audiometric Thresholds and Curve Types Stratified by MRI Findings.

Data are presented as median (IQR) for PTA. ΔPTA represents posttreatment minus pretreatment thresholds. Bold indicates statistical significance (P < .05).
Abbreviation: PTA, pure-tone average.

Changes in hearing thresholds before and after treatment among MRI-defined groups. (A) Posttreatment full-frequency and frequency-specific PTA in MRI-negative, endolymphatic hydrops, and labyrinthine enhancement groups. Group comparisons were performed using the Kruskal-Wallis test with the Bonferroni correction. LF, low frequency; MF, mid-frequency; HF, high frequency. (B-D) Pretreatment and posttreatment full-frequency PTA in each group. Within-group differences were evaluated using the Wilcoxon signed-rank test. PTA, pure-tone average.
Significant differences were observed among the 3 groups in Siegel’s criteria distribution (Fisher’s exact test, P = .032, Table 4) and recovery rate (χ² = 8.19, P = .017). The MRI-negative group demonstrated higher rates of complete recovery and overall recovery than the LSA group (adj. P < .05).
Hearing Recovery Based on Siegel’s Criteria.

Bold indicates statistical significance (P < .05).
Regression Analysis
In the univariate analysis of full-frequency ΔPTA, age (P = .004), MRI classification (P = .028), vertigo (P = .097), and tinnitus (P = .070) met the predefined inclusion criterion (P < .10). Based on baseline comparisons shown in Table 1, sex (P = .064), vertigo (P = .001), and tinnitus (P = .038) differed across MRI groups and were therefore considered potential confounding factors. Accordingly, sex, age, vertigo, tinnitus, and MRI classification were included in the multivariable linear regression model. In the multivariable analysis, only age remained independently associated with full-frequency ΔPTA (B = −0.312, SE = 0.107, β = −.288, P = .004), indicating that older age was associated with smaller hearing gain. MRI classification yielded a P value of .067 in the multivariable model.
Further analyses were performed for frequency-specific ΔPTA. In the univariable linear regression analysis of low-frequency ΔPTA, age (B = −0.387, SE = 0.135, P = .005) and MRI classification (B = −5.958, SE = 2.205, P = .008) were significantly associated with low-frequency hearing gain, while vertigo (P = .079) met the predefined inclusion criterion (P < .10). Other variables showed no significant associations (Table S1). Based on the univariable results and baseline differences, relevant variables were entered into the multivariable linear regression model. In the multivariable analysis (Table 5), both age (B = −0.337, SE = 0.134, P = .014) and MRI classification (B = −5.074, SE = 2.174, P = .022) remained independently associated with low-frequency ΔPTA, with older age and LSA phenotype predicting smaller hearing gain. No significant associations between MRI classification and mid- or high-frequency ΔPTA were observed in the regression analyses.
Multivariable Linear Regression Analysis of Factors Associated With Low-Frequency ΔPTA.

Bold indicates statistical significance (P < .05).
Abbreviation: PTA, pure-tone average.
Discussion
SSNHL is commonly manifested as the first symptom of various inner ear diseases. The etiology of SSNHL is complex, and its clinical manifestations are highly heterogeneous. Early-objective evaluation and classification have been challenging to achieve in clinical practice. In recent years, gadolinium-enhanced inner ear MRI of the inner ear has been integrated for the study of inner ear pathology. MRI can be used to evaluate inner ear fluid dynamics, blood-labyrinth barrier function, and lesion extent.27 -29 In contrast to the limitations of clinical symptoms, MRI can provide direct pathophysiological information. Building on this advancement, we classified SSNHL into 3 imaging-based subtypes: MRI-negative, endolymphatic hydrops, and labyrinthine signal abnormality. Subsequently, the association between imaging classification and clinical phenotype and prognosis was evaluated.
Previous studies have shown that the unaffected ear of SSNHL patients typically lacks hyperintensity on 3D-FLAIR sequences,27,30 a feature also consistently observed in healthy individuals. 13 Therefore, 3D-FLAIR inner ear hyperintensity is considered an abnormal or even pathological phenomenon, which we collectively term LSA. Theoretically, these signal abnormalities can be further subdivided based on underlying mechanisms, such as intralabyrinthine hemorrhage, inflammatory response, or blood-labyrinthine barrier disruption. 13 However, due to the limited sample size in this study, reliable subcategorization at this level was not feasible. Consequently, all cases exhibiting 3D-FLAIR hyperintensity were analyzed collectively as LSA.
The results of this study indicate that there are significant differences in the clinical phenotypes among patients with different MRI classifications. Patients with LSA exhibited distinct clinical features: 71.4% had involvement of ≥2 inner ear subunits, with vertigo in 100% and tinnitus in 95.2% of cases. Posttreatment hearing thresholds were significantly higher in this group, and functional recovery rates were the lowest, consistent with previous reports.31,32 We speculate that inner ear hyperintensity reflects severe blood-labyrinth barrier disruption and irreversible outer hair cell damage, 33 which may impede drug delivery and explain the poor prognosis. Combined with standard clinical treatments, more aggressive early interventions may be warranted for these patients, such as intratympanic steroid injections, while the use of defibrination or anticoagulant drugs should be approached with caution to improve therapeutic outcomes.
EH represents a distinct imaging subtype with important prognostic implications. Longitudinal studies have shown that more than half of SSNHL patients with EH eventually develop Menière’s disease. 34 This progression suggests that EH, which is the primary pathological manifestation of MD, 35 may not be a coincidental imaging finding in SSNHL but rather identifies an early-stage or a shared pathological substrate between the 2 conditions. In our cohort, the EH group exhibited moderate clinical involvement: vertigo in 50% and tinnitus in 75% of cases, with most patients (75%) having a single affected inner ear subunit (cochlear: 30%; vestibular: 45%). Audiologically, the full-frequency, low-frequency, and mid-frequency hearing thresholds of patients in the EH group were at the intermediate level among the 3 groups before and after treatment. The ΔPTA gains were limited, and the hearing curves were predominantly flat or showed low-frequency decline. The recovery rate of the EH group was also in the middle among all 3 groups (15.0%). Although the initial hearing impairment in EH-related SSNHL may not be the most severe, the prognosis for recovery warrants caution as it may represent a chronic, evolving pathology rather than a monophasic event.
Multivariable regression analysis identified age as an independent predictor of hearing gain, with older patients showing less improvement, consistent with previous reports.34 -37 This may reflect age-related declines in outer hair cell plasticity and inner ear microcirculation, limiting recovery capacity. For low-frequency hearing gain, both age and MRI classification emerged as independent predictors, indicating that the prognostic value of imaging subtypes is frequency-specific. Specifically, the LSA group showed poorer low-frequency recovery, whereas the MRI-negative group had relatively-better outcomes at low frequencies; the EH group showed intermediate recovery. This may be explained by several factors. First, the LSA group exhibited more extensive inner ear involvement, which is associated with more severe impairment of auditory and vestibular function. 38 Second, EH is characterized by fluctuating low-frequency hearing loss, 39 and recovery in SSNHL cases with low-frequency involvement or ascending audiograms is generally better than in those with high-frequency involvement or descending audiograms.37,40 -42 Therefore, these observations collectively explain the association between MRI classification and low-frequency hearing gain. However, given the retrospective design and limited sample size, these findings warrant validation in larger prospective studies.
This study has several limitations. The relatively-small sample size, particularly in the EH and LSA subgroups, may have limited statistical power to detect group differences, necessitating validation in larger prospective cohorts. In addition, while intratympanic gadolinium administration was chosen for its superior inner ear enhancement via direct diffusion across the round and oval window membranes,43,44 this technique has inherent methodological limitations, including variability in round window permeability, potential contrast loss, and the logistical burden of delayed imaging. Well-designed comparative studies are needed to clarify the relative advantages of intratympanic versus intravenous approaches and to inform optimal imaging strategies for SSNHL. Finally, as this study focused primarily on early-treatment response during hospitalization, delayed recovery cannot be excluded. Therefore, follow-up evaluations at the 3rd and 6th month, when hearing thresholds typically stabilize, are warranted. Overall, the present findings are more applicable to the prediction of early-treatment sensitivity, and the predictive value of imaging classification for long-term prognosis requires validation in larger prospective cohorts.
Conclusion
This study demonstrates that gadolinium-enhanced inner ear MRI can identify distinct imaging features in patients with SSNHL, which are associated with differences in clinical presentation and hearing outcomes. Labyrinthine signal abnormalities were associated with more extensive inner ear involvement and poorer hearing recovery, while older age was independently associated with reduced hearing gain. These findings suggest that MRI-based classification may provide additional information for early assessment of SSNHL. Given the retrospective design, limited subgroup sample size, and short-term follow-up, these findings should be interpreted cautiously. Larger prospective studies with long-term follow-up are needed to confirm the clinical applicability of MRI-based classification in SSNHL.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216261440784 – Supplemental material for Gadolinium-Enhanced MRI Classification of Sudden Sensorineural Hearing Loss and Its Clinical Correlates
Supplemental material, sj-docx-1-ohn-10.1177_19160216261440784 for Gadolinium-Enhanced MRI Classification of Sudden Sensorineural Hearing Loss and Its Clinical Correlates by Xiaojing Guo, Xiaoying Ke, Shengnan Ye and Heng Xiao in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
The authors thank all the patients who participated in this study.
Author Contributions
Xiaojing Guo and Xiaoying Ke designed and coordinated the study. Xiaoying Ke conducted data collection. Xiaojing Guo analyzed the data and drafted the manuscript. Heng Xiao and Shengnan Ye interpreted the findings and critically revised the manuscript. All authors contributed to the study and approved the final version for submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Fujian Province (No. 2024J01557), Joint Funds for the Innovation of Science and Technology, Fujian Province (No. 2024Y9190) and Startup Fund for Scientific Research of Fujian Medical University (No. 2024QH1085).
Supplemental Material
Additional supporting information is available in the online version of the article.
